18.S: Ethers and Epoxides; Thiols and Sulfides (Summary)
- Page ID
- 207107
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Concepts & Vocabulary
- Ethers are molecules containing oxygen which is bonded to two carbon groups.
- Thiols are sulfur analogues of alcohols with an SH group instead of OH.
- Sulfides are sulfur analogues of ethers with sulfur bonded to two carbon groups instead of oxygen.
18.1 Names and Properties of Ethers
- Ether groups are named as alkoxy groups.
- Ethers do not have intramolecular hydrogen bonding (unlike alcohols), therefore ethers have significantly reduced intermolecular forces causing boiling points that are much lower than similar sized alcohols.
- Alkoxymercuration can be used to prepare an ether from an alkene.
18.3 Reactions of Ethers: Acidic Cleavage
- The carbon-oxygen bonds of ethers can be cleaved with strong acids through either nucleophilic substitution or elimination reactions.
18.4 Reactions of Ethers: Claisen Rearrangement
- The Claisen rearrangement is a [3, 3] sigmatropic rearrangement reaction that converts aryl or enol ethers into carbonyl compounds (though the aromatic version rearranges into a phenol to re-establish aromaticity.
- Epoxides, also called oxiranes, have a three-membered ring structure with one oxygen and two carbon atoms.
- Epoxides can be formed from alkenes by reaction with peroxy acids (MCPBA for example).
- Epoxides can be formed from halohydrin molecules by reaction with a base, which causes an intramolecular Williamson ether synthesis.
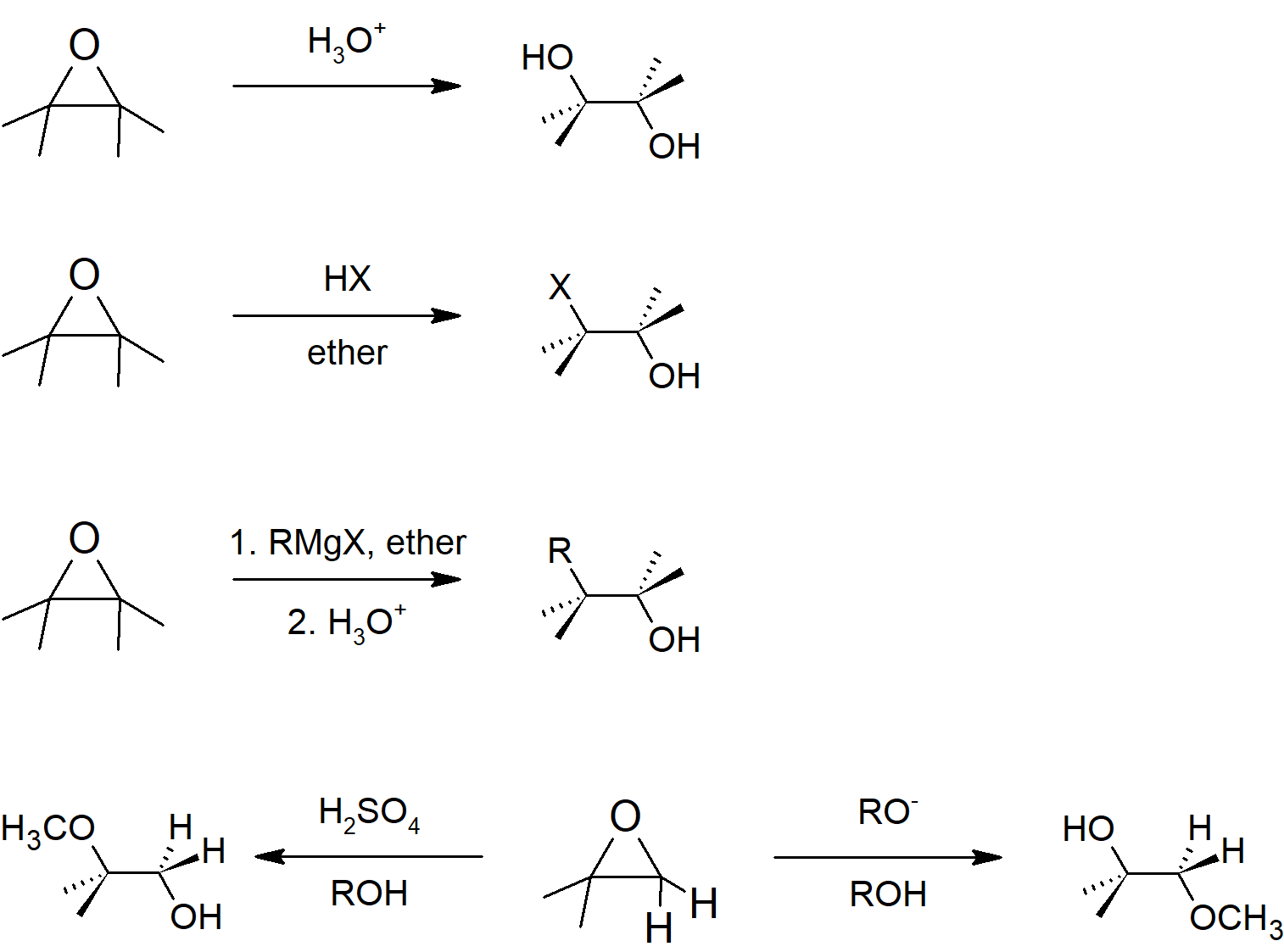
18.6 Reactions of Epoxides: Ring Opening
- When epoxides are ring opened under basic conditions, they follow SN2 mechanism leading to the nucleophile adding to the less substituted side of the epoxide.
- When epoxides are ring opened under acidic conditions, they follow SN1 mechanism leading to the nucleophile adding to the more substituted side of the epoxide.
- When epoxides are ring opened in aqueous reactions, the result is an anti-diol.
- Halo acids can be added to epoxides to form anti-halohydrins.
- Crown ethers are cyclic ethers containing several oxygen atoms.
- Crown ethers are named by the number of total atoms in the ring, followed by the word crown and finally the number of oxygen atoms (18-crown-6 for example).
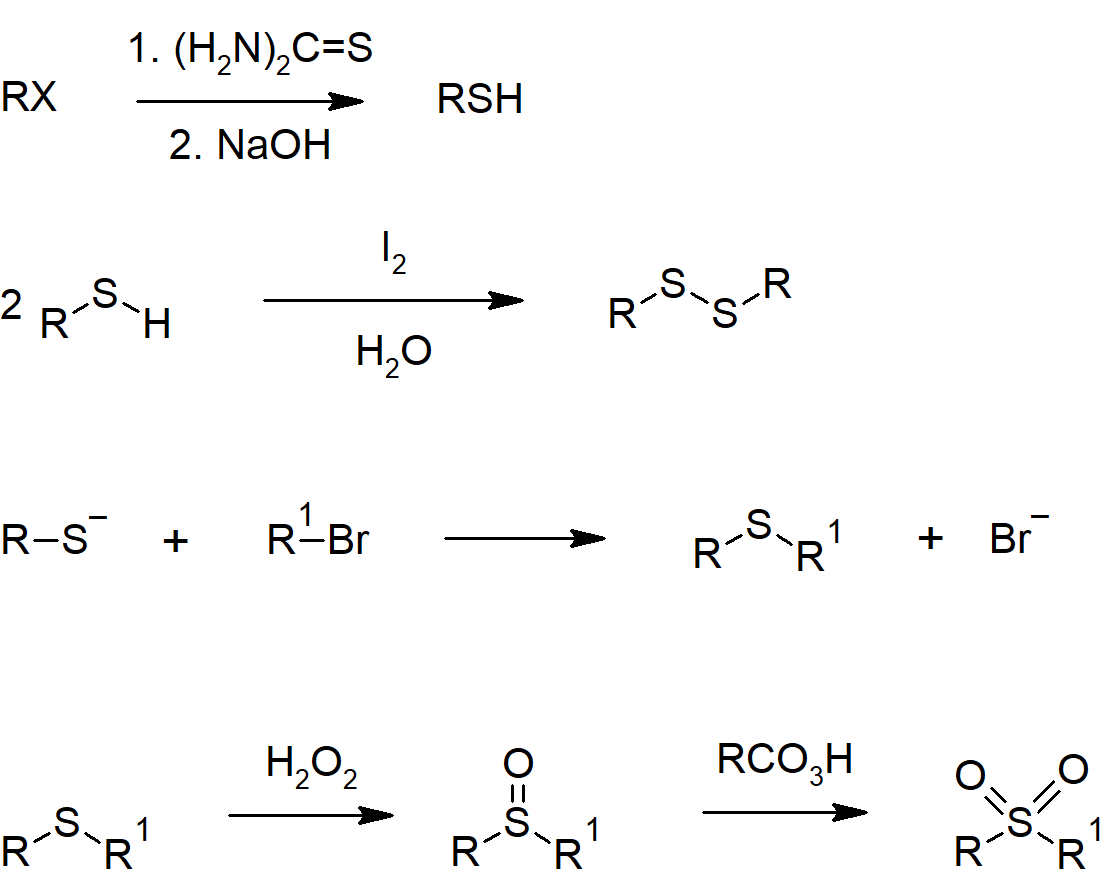
- Thiols can be prepared from alkyl halides through reaction with hydrosulfide ion (SH-) or through a more complicated series of reactions including thiourea.
- Thiols can be oxidized with mild oxidizing agents to form disulfides.
- Disulfide bridges link cysteine residues in protein structures.
- Sulfides are sulfur analogues of ether, though are much better nucleophiles with sulfur in place of oxygen.
- Ethers show standard C-H stretches and bends in IR along with a strong C-O stretch around 1000 cm-1.
- In 1H NMR, hydrogens on carbons adjacent to the oxygen typically appear between 3.4-4.5 ppm.
- Hydrogens on carbons of an epoxide ring typically appear between 2.5-3.5 ppm in 1H NMR.
18.10 Interchapter: A Preview of Carbonyl Chemistry
- Carbonyl groups are one of the most important features in organic chemistry and consist of a sp2 carbon double-bonded to oxygen.
- Carbonyl groups are present in ~10 different functional groups.
- Carbonyl groups are polarized with a partial positive charge on carbon and partial negative charge on oxygen. This makes the carbon atom an electrophile, while the oxygen can act as a nucleophile.
- Carbonyl groups can react through several mechanisms including nucleophilic addition and nucleophilic acyl substitution, alpha substitution and condensation.
Skills to Master
- Skill 18.1 Name ethers using common naming and IUPAC.
- Skill 18.2 Write reaction equations for preparation of ethers.
- Skill 18.3 Write mechanisms for reactions of ethers with strong halogen acids.
- Skill 18.4 Draw mechanisms for Cope and Claisen rearrangements.
- Skill 18.5 Draw mechanisms for ring-opening epoxides under acidic and basic conditions.
- Skill 18.6 Draw and name crown ethers.
- Skill 18.7 Explain how disulfide bridges contribute to protein structure.
- Skill 18.8 Give an example of S-adenosyl methionine activity in biological systems.
- Skill 18.9 Use IR and NMR spectra to identify ethers.
Summary of Reactions
Ether and Epoxide Preparation
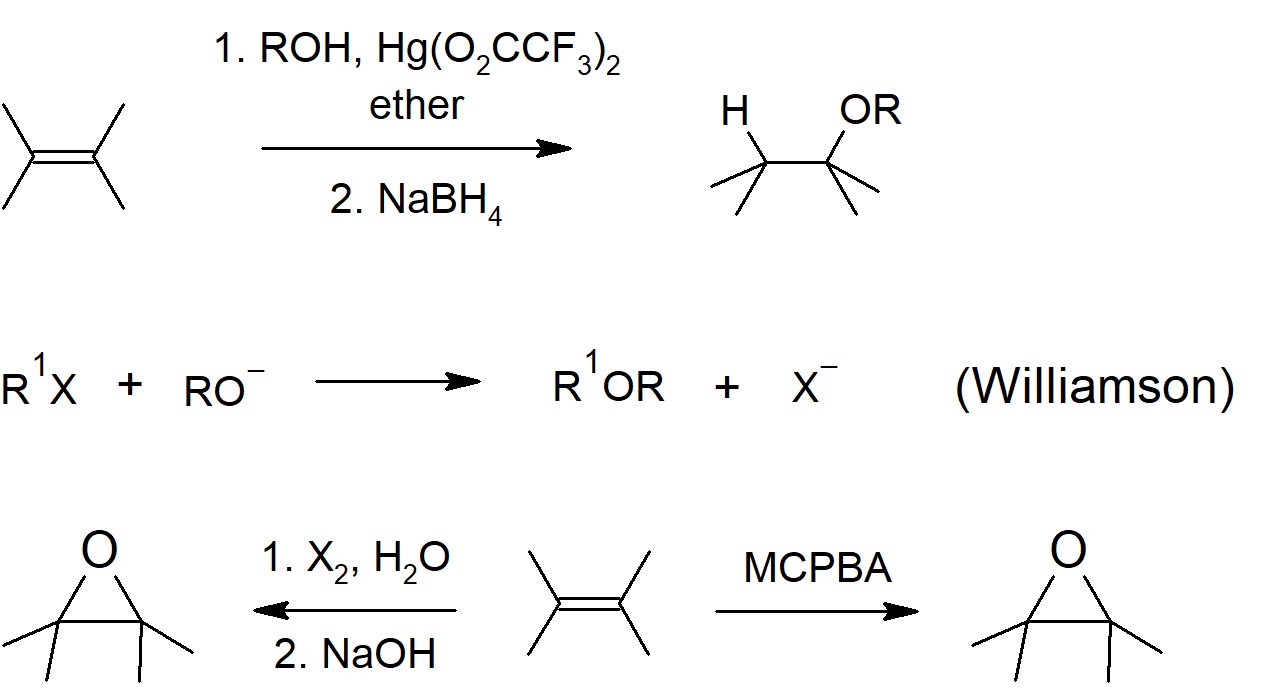
Ether Reactions
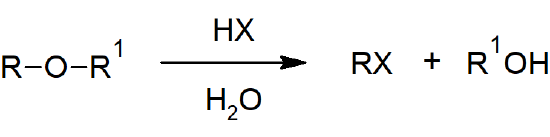
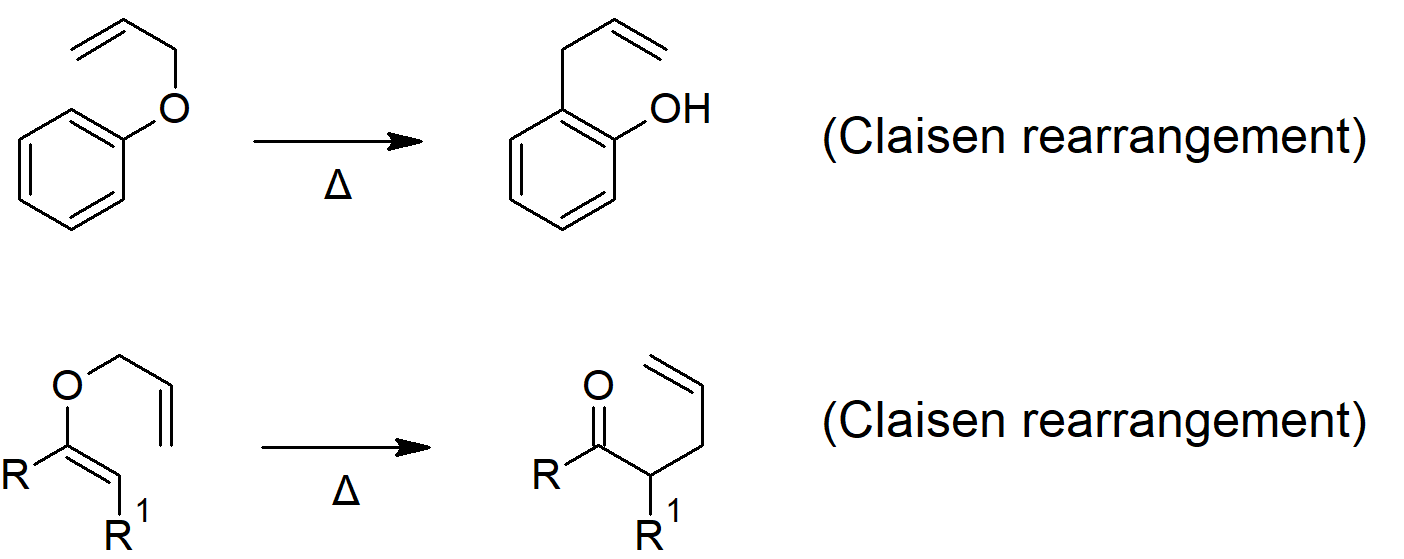
Epoxide Reactions
Sulfur Compound Reactions