Periodic Trends
- Page ID
- 618
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties. These trends exist because of the similar atomic structure of the elements within their respective group families or periods, and because of the periodic nature of the elements.
Electronegativity Trends
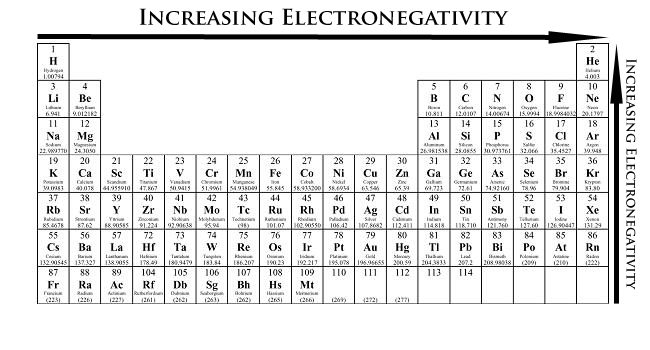
Electronegativity can be understood as a chemical property describing an atom's ability to attract and bind with electrons. Because electronegativity is a qualitative property, there is no standardized method for calculating electronegativity. However, the most common scale for quantifying electronegativity is the Pauling scale (Table A2), named after the chemist Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due to the qualitative nature of electronegativity. Electronegativity values for each element can be found on certain periodic tables. An example is provided below.
Electronegativity measures an atom's tendency to attract and form bonds with electrons. This property exists due to the electronic configuration of atoms. Most atoms follow the octet rule (having the valence, or outer, shell comprise of 8 electrons). Because elements on the left side of the periodic table have less than a half-full valence shell, the energy required to gain electrons is significantly higher compared with the energy required to lose electrons. As a result, the elements on the left side of the periodic table generally lose electrons when forming bonds. Conversely, elements on the right side of the periodic table are more energy-efficient in gaining electrons to create a complete valence shell of 8 electrons. The nature of electronegativity is effectively described thus: the more inclined an atom is to gain electrons, the more likely that atom will pull electrons toward itself.
- From left to right across a period of elements, electronegativity increases. If the valence shell of an atom is less than half full, it requires less energy to lose an electron than to gain one. Conversely, if the valence shell is more than half full, it is easier to pull an electron into the valence shell than to donate one.
- From top to bottom down a group, electronegativity decreases. This is because atomic number increases down a group, and thus there is an increased distance between the valence electrons and nucleus, or a greater atomic radius.
- Important exceptions of the above rules include the noble gases, lanthanides, and actinides. The noble gases possess a complete valence shell and do not usually attract electrons. The lanthanides and actinides possess more complicated chemistry that does not generally follow any trends. Therefore, noble gases, lanthanides, and actinides do not have electronegativity values.
- As for the transition metals, although they have electronegativity values, there is little variance among them across the period and up and down a group. This is because their metallic properties affect their ability to attract electrons as easily as the other elements.
According to these two general trends, the most electronegative element is fluorine, with 3.98 Pauling units.
Ionization Energy Trends
Ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. Conceptually, ionization energy is the opposite of electronegativity. The lower this energy is, the more readily the atom becomes a cation. Therefore, the higher this energy is, the more unlikely it is the atom becomes a cation. Generally, elements on the right side of the periodic table have a higher ionization energy because their valence shell is nearly filled. Elements on the left side of the periodic table have low ionization energies because of their willingness to lose electrons and become cations. Thus, ionization energy increases from left to right on the periodic table.
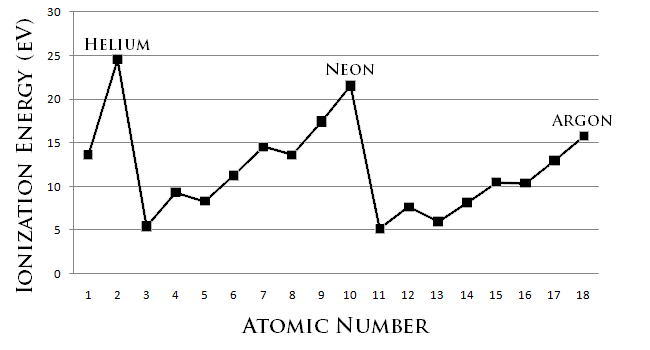
Another factor that affects ionization energy is electron shielding. Electron shielding describes the ability of an atom's inner electrons to shield its positively-charged nucleus from its valence electrons. When moving to the right of a period, the number of electrons increases and the strength of shielding increases. As a result, it is easier for valence shell electrons to ionize, and thus the ionization energy decreases down a group. Electron shielding is also known as screening.
- The ionization energy of the elements within a period generally increases from left to right. This is due to valence shell stability.
- The ionization energy of the elements within a group generally decreases from top to bottom. This is due to electron shielding.
- The noble gases possess very high ionization energies because of their full valence shells as indicated in the graph. Note that helium has the highest ionization energy of all the elements.
Some elements have several ionization energies; these varying energies are referred to as the first ionization energy, the second ionization energy, third ionization energy, etc. The first ionization energy is the energy requiredto remove the outermost, or highest, energy electron, the second ionization energy is the energy required to remove any subsequent high-energy electron from a gaseous cation, etc. Below are the chemical equations describing the first and second ionization energies:
First Ionization Energy:
\[ X_{(g)} \rightarrow X^+_{(g)} + e^- \nonumber \]
Second Ionization Energy:
\[ X^+_{(g)} \rightarrow X^{2+}_{(g)} + e^- \nonumber \]
Generally, any subsequent ionization energies (2nd, 3rd, etc.) follow the same periodic trend as the first ionization energy.

Ionization energies decrease as atomic radii increase. This observation is affected by \(n\) (the principal quantum number) and \(Z_{eff}\) (based on the atomic number and shows how many protons are seen in the atom) on the ionization energy (I). The relationship is given by the following equation:
\[ I = \dfrac{R_H Z^2_{eff}}{n^2} \nonumber \]
- Across a period, \(Z_{eff}\) increases and n (principal quantum number) remains the same, so the ionization energy increases.
- Down a group, \(n\) increases and \(Z_{eff}\) increases slightly; the ionization energy decreases.
Electron Affinity Trends
As the name suggests, electron affinity is the ability of an atom to accept an electron. Unlike electronegativity, electron affinity is a quantitative measurement of the energy change that occurs when an electron is added to a neutral gas atom. The more negative the electron affinity value, the higher an atom's affinity for electrons.
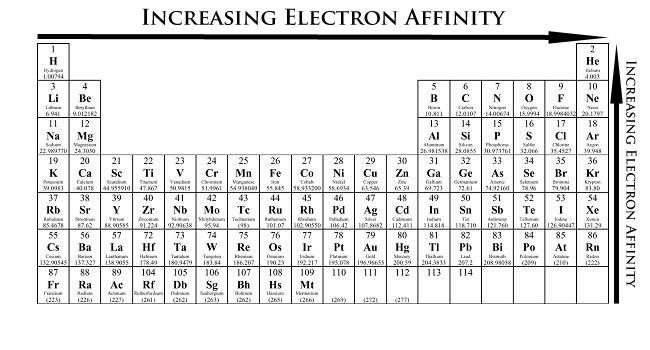
Electron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below). This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. Therefore, electron affinity decreases. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period.
- Electron affinity increases from left to right within a period. This is caused by the decrease in atomic radius.
- Electron affinity decreases from top to bottom within a group. This is caused by the increase in atomic radius.
Atomic Radius Trends
The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. The covalent radii of these molecules are often referred to as atomic radii. This distance is measured in picometers. Atomic radius patterns are observed throughout the periodic table.
Atomic size gradually decreases from left to right across a period of elements. This is because, within a period or family of elements, all electrons are added to the same shell. However, at the same time, protons are being added to the nucleus, making it more positively charged. The effect of increasing proton number is greater than that of the increasing electron number; therefore, there is a greater nuclear attraction. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The valence electrons are held closer towards the nucleus of the atom. As a result, the atomic radius decreases.
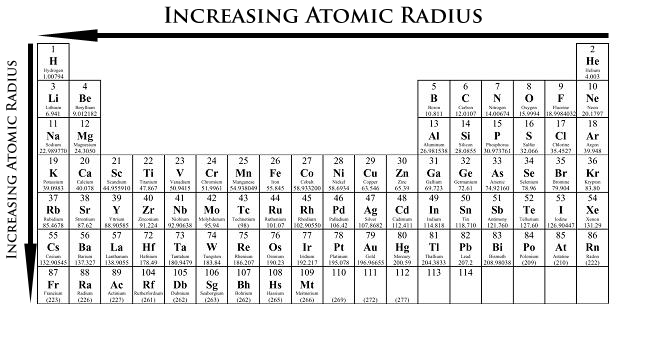
Down a group, atomic radius increases. The valence electrons occupy higher levels due to the increasing quantum number (n). As a result, the valence electrons are further away from the nucleus as ‘n’ increases. Electron shielding prevents these outer electrons from being attracted to the nucleus; thus, they are loosely held, and the resulting atomic radius is large.
- Atomic radius decreases from left to right within a period. This is caused by the increase in the number of protons and electrons across a period. One proton has a greater effect than one electron; thus, electrons are pulled towards the nucleus, resulting in a smaller radius.
- Atomic radius increases from top to bottom within a group. This is caused by electron shielding.
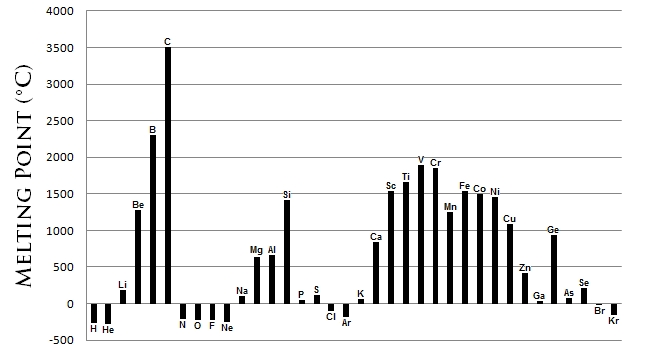
Melting Point Trends
The melting points is the amount of energy required to break a bond(s) to change the solid phase of a substance to a liquid. Generally, the stronger the bond between the atoms of an element, the more energy required to break that bond. Because temperature is directly proportional to energy, a high bond dissociation energy correlates to a high temperature. Melting points are varied and do not generally form a distinguishable trend across the periodic table. However, certain conclusions can be drawn from Figure \(\PageIndex{7}\).
- Metals generally possess a high melting point.
- Most non-metals possess low melting points.
- The non-metal carbon possesses the highest melting point of all the elements. The semi-metal boron also possesses a high melting point.
Metallic Character Trends
The metallic character of an element can be defined as how readily an atom can lose an electron. From right to left across a period, metallic character increases because the attraction between valence electron and the nucleus is weaker, enabling an easier loss of electrons. Metallic character increases as you move down a group because the atomic size is increasing. When the atomic size increases, the outer shells are farther away. The principal quantum number increases and average electron density moves farther from nucleus. The electrons of the valence shell have less attraction to the nucleus and, as a result, can lose electrons more readily. This causes an increase in metallic character.
- Metallic characteristics decrease from left to right across a period. This is caused by the decrease in radius (caused by Zeff, as stated above) of the atom that allows the outer electrons to ionize more readily.
- Metallic characteristics increase down a group. Electron shielding causes the atomic radius to increase thus the outer electrons ionizes more readily than electrons in smaller atoms.
- Metallic character relates to the ability to lose electrons, and nonmetallic character relates to the ability to gain electrons.
Another easier way to remember the trend of metallic character is that moving left and down toward the bottom-left corner of the periodic table, metallic character increases toward Groups 1 and 2, or the alkali and alkaline earth metal groups. Likewise, moving up and to the right to the upper-right corner of the periodic table, metallic character decreases because you are passing by to the right side of the staircase, which indicate the nonmetals. These include the Group 8, the noble gases, and other common gases such as oxygen and nitrogen.
- In other words:
- Move left across period and down the group: increase metallic character (heading towards alkali and alkaline metals)
- Move right across period and up the group: decrease metallic character (heading towards nonmetals like noble gases)

Problems
The following series of problems reviews general understanding of the aforementioned material.
1. Based on the periodic trends for ionization energy, which element has the highest ionization energy?
- Fluorine (F)
- Nitrogen (N)
- Helium (He)
2.) Nitrogen has a larger atomic radius than oxygen.
- A.) True
- B.) False
3.) Which has more metallic character, Lead (Pb) or Tin (Sn)?
4.) Which element has a higher melting point: chlorine (Cl) or bromine (Br)?
5.) Which element is more electronegative, sulfur (S) or selenium (Se)?
6) Why is the electronegativity value of most noble gases zero?
7) Arrange these atoms in order of decreasing effective nuclear charge by the valence electrons: Si, Al, Mg, S
8) Rewrite the following list in order of decreasing electron affinity: fluorine (F), phosphorous (P), sulfur (S), boron (B).
9) An atom with an atomic radius smaller than that of sulfur (S) is __________.
- A.) Oxygen (O)
- B.) Chlorine (Cl)
- C.) Calcium (Ca)
- D.) Lithium (Li)
- E.) None of the above
10) A nonmetal has a smaller ionic radius compared with a metal of the same period.
- A.) True B.) False
Solutions
1. Answer: C.) Helium (He)
Explanation: Helium (He) has the highest ionization energy because, like other noble gases, helium's valence shell is full. Therefore, helium is stable and does not readily lose or gain electrons.
2. Answer: A.) True
Explanation: Atomic radius increases from right to left on the periodic table. Therefore, nitrogen is larger than oxygen.
3. Answer: Lead (Pb)
Explanation: Lead and tin share the same column. Metallic character increases down a column. Lead is under tin, so lead has more metallic character.
4. Answer: Bromine (Br)
Explanation: In non-metals, melting point increases down a column. Because chlorine and bromine share the same column, bromine possesses the higher melting point.
5. Answer: Sulfur (S)
Explanation: Note that sulfur and selenium share the same column. Electronegativity increases up a column. This indicates that sulfur is more electronegative than selenium.
6. Answer: Most noble gases have full valence shells.
Explanation: Because of their full valence electron shell, the noble gases are extremely stable and do not readily lose or gain electrons.
7. Answer: S > Si > Al > Mg.
Explanation: The electrons above a closed shell are shielded by the closed shell. S has 6 electrons above a closed shell, so each one feels the pull of 6 protons in the nucleus.
8. Answer: Fluorine (F)>Sulfur (S)>Phosphorous (P)>Boron (B)
Explanation: Electron affinity generally increases from left to right and from bottom to top.
9. Answer: C.) Oxygen (O)
Explanation: Periodic trends indicate that atomic radius increases up a group and from left to right across a period. Therefore, oxygen has a smaller atomic radius sulfur.
10. Answer: B.) False
Explanation: The reasoning behind this lies in the fact that a metal usually loses an electron in becoming an ion while a non-metal gains an electron. This results in a smaller ionic radius for the metal ion and a larger ionic radius for the non-metal ion.
References
- Pinto, Gabriel. "Using Balls of Different Sports To Model the Variation of Atomic Sizes." J. Chem. Educ.1998 75 725.{cke_protected}{C}
- Qureshi, Pushkin M.; Kamoonpuri, S. Iqbal M. "Ion solvation: The ionic radii problem." J. Chem. Educ.1991, 68, 109.
- Smith, Derek W. "Atomization enthalpies of metallic elemental substances using the semi-quantitative theory of ionic solids: A simple model for rationalizing periodic trends." J. Chem. Educ. 1993, 70, 368.
- Russo, Steve, and Mike Silver. Introductory Chemistry. San Francisco: Pearson, 2007.
- Petrucci, Ralph H, et al. General Chemistry: Principles and Modern Applications. 9th Ed. New Jersey: Pearson, 2007.
- Atkins, Peter et. al, Physical Chemistry, 7th Edition, 2002, W.H Freeman and Company, New York, pg. 390.
- Alberty, Robert A. et. al, Physical Chemistry, 3rd Edition, 2001, John Wiley & Sons, Inc, pg. 380.
- Kots, John C. et. al, Chemistry & Chemical Reactivity, 5th Edition, 2003, Thomson Learning Inc, pg. 305-309.

