5.1.1: Biology- Applications of Electronic Structure
- Page ID
- 50001
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The Electronic Structure of Atoms in Biology
back to The Electronic Structure of Atoms
Since Rutherford had discovered that the nucleus occupies only 10-12% (a millionth of a millionth of a percent) of an atom's volume, it became clear that the nucleus was essentially a tiny point, and that the shape and size of atoms is entirely due to a very sparse "electron cloud" of some kind. This realization was critical to biology, because the shape and size of molecules is critical to understanding their biological function, and the "electron cloud" of constituent atoms determines their shape and size.
This is sometimes very subtle. For example, there are two different forms of the carvone molecule, C10H14O (like right and left hands):
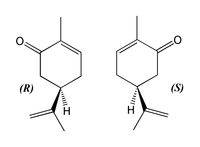

The two forms of carvone[1]
The carvone on the left (R-(-)-Carvone) smells like spearmint, and is used for aromatherapy, alternative medicine, and room fresheners. The carvone on the right, (S-(+)-carvone) smells like caraway. The difference in smell is an indication of the difference in biological activity that comes from very small differences in shape, a principal that is encountered again and again in the drug industry.
But it would also be impossible to understand photosynthesis and vision, and indeed, the color of leaves and flowers without understanding the electronic structure of atoms, because there would be no light (or color or photochemical reactions) if it were not for electrons and their ability to absorb or release energy.
Naturally, all chemists, biologists, and physicists became interested in the electronic structure of atoms—the chemists because they wanted to explain valence, chemical properties and bonding, the physicists because they wanted to explain the color and spectra of atoms (the light emitted when gaseous atoms were raised to a high temperature or bombarded by electrons) and biologists for the reasons above. The chief contributors to fundamental understanding of electronic structure, which developed mainly during the 15 years between 1910 and 1925, were the U.S. chemist Gilbert Newton Lewis (1875 to 1946), and the Danish physicist Niels Bohr (1885 to 1962).
Ideas about the electronic structures of atoms progressed during the first half of the twentieth century. The periodic repetition of chemical properties discovered by Mendeleev led G. N. Lewis to the conclusion that atoms must have a shell structure. This was confirmed by wave mechanics, which developed very beautiful wave patterns which replaced the circular or elliptical shells of earlier models.
The energy of each electron in an atom depends on how strongly the electron is attracted by the positive charge on the nucleus and on how much it is repelled by other electrons. Although each electron cannot be assigned a precise trajectory or orbit in an atom, its wave pattern allows us to determine the probability that it will be within a certain location. From this the energy of each electron and the order of filling orbitals can be obtained. Thus we can determine the electron configuration for an atom of any element. Such electron configurations correlate with the periodic table.
Because electrons in inner orbitals screen outer electrons from nuclear charge, the fourth and higher shells begin to fill before d (and sometimes f) subshells in previous shells are occupied. This overlap in energies of shells explains why Lewis’ ideas are less useful for elements in the fourth and subsequent rows of the periodic table, like the cobalt in vitamin B12 or iron in hemoglobin. It also accounts for the steady variation in properties of transition metals, from titanium to zinc, across the middle of the table, and for the nearly identical characteristics of inner transition elements as opposed to the large differences from one group of representative elements to the next.
Although some added complication arises from the wave-mechanical picture, it does confirm Lewis’ basic postulate that valence electrons determine chemical properties and influence the bonding of one atom to another. In other pages you will see how rearrangement of valence electrons can hold atoms together, and how different kinds of bonds result in different macroscopic properties.


