13.3: Radiochemistry
- Page ID
- 151884
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Atoms that have the same number of protons but a different number of neutrons are isotopes. To identify an isotope we use the notation \({}_Z^A E\), where E is the element’s atomic symbol, Z is the element’s atomic number, and A is the element’s atomic mass number. Although an element’s different isotopes have the same chemical properties, their nuclear properties are not identical. The most important difference between isotopes is their stability. The nuclear configuration of a stable isotope remains constant with time. Unstable isotopes, however, disintegrate spontaneously, emitting radioactive particles as they transform into a more stable form.
An element’s atomic number, Z, is equal to the number of protons and its atomic mass, A, is equal to the sum of the number of protons and neutrons. We represent an isotope of carbon-13 as \(_{6}^{13} \text{C}\) because carbon has six protons and seven neutrons. Sometimes we omit Z from this notation—identifying the element and the atomic number is repetitive because all isotopes of carbon have six protons and any atom that has six protons is an isotope of carbon. Thus, 13C and C–13 are alternative notations for this isotope of carbon.
The most important types of radioactive particles are alpha particles, beta particles, gamma rays, and X-rays. An alpha particle, \(\alpha\), is equivalent to a helium nucleus, \({}_2^4 \text{He}\). When an atom emits an alpha particle, the product in a new atom whose atomic number and atomic mass number are, respectively, 2 and 4 less than its unstable parent. The decay of uranium to thorium is one example of alpha emission.
\[_{92}^{238} \text{U} \longrightarrow _{90}^{234} \text{Th}+\alpha \nonumber\]
A beta particle, \(\beta\), comes in one of two forms. A negatron, \(_{-1}^0 \beta\), is produced when a neutron changes into a proton, increasing the atomic number by one, as shown here for lead.
\[_{82}^{214} \mathrm{Pb} \longrightarrow_{83}^{214} \mathrm{Bi} + _{-1}^{0} \beta \nonumber\]
The conversion of a proton to a neutron results in the emission of a positron, \(_{1}^0 \beta\).
\[_{15}^{30} \mathrm{P} \longrightarrow_{14}^{30} \mathrm{Si} + _{1}^{0} \beta \nonumber\]
A negatron, which is the more common type of beta particle, is equivalent to an electron.
The emission of an alpha or a beta particle often produces an isotope in an unstable, high energy state. This excess energy is released as a gamma ray, \(\gamma\), or as an X-ray. Gamma ray and X-ray emission may also occur without the release of an alpha particle or a beta particle.
Theory and Practice
A radioactive isotope’s rate of decay, or activity, follows first-order kinetics
\[A=-\frac{d N}{d t}=\lambda N \label{13.1}\]
where A is the isotope’s activity, N is the number of radioactive atoms present in the sample at time t, and \(\lambda\) is the isotope’s decay constant. Activity is expressed as the number of disintegrations per unit time.
As with any first-order process, we can rewrite Equation \ref{13.1} in an integrated form.
\[N_{t}=N_{0} e^{-\lambda t} \label{13.2}\]
Substituting Equation \ref{13.2} into Equation \ref{13.2} gives
\[A=\lambda N_{0} e^{-\lambda t}=A_{0} e^{-\lambda t} \label{13.3}\]
If we measure a sample’s activity at time t we can determine the sample’s initial activity, A0, or the number of radioactive atoms originally present in the sample, N0.
An important characteristic property of a radioactive isotope is its half-life, t1/2, which is the amount of time required for half of the radioactive atoms to disintegrate. For first-order kinetics the half-life is
\[t_{1 / 2}=\frac{0.693}{\lambda} \label{13.4}\]
Because the half-life is independent of the number of radioactive atoms, it remains constant throughout the decay process. For example, if 50% of the radioactive atoms remain after one half-life, then 25% remain after two half-lives, and 12.5% remain after three half-lives.
Suppose we begin with an N0 of 1200 atoms During the first half-life, 600 atoms disintegrate and 600 remain. During the second half-life, 300 of the 600 remaining atoms disintegrate, leaving 300 atoms or 25% of the original 1200 atoms. Of the 300 remaining atoms, only 150 remain after the third half-life, or 12.5% of the original 1200 atoms.
Kinetic information about a radioactive isotope usually is given in terms of its half-life because it provides a more intuitive sense of the isotope’s stability. Knowing, for example, that the decay constant for \(_{38}^{90}\text{Sr}\) is 0.0247 yr–1 does not give an immediate sense of how fast it disintegrates. On the other hand, knowing that its half-life is 28.1 yr makes it clear that the concentration of \(_{38}^{90}\text{Sr}\) in a sample remains essentially constant over a short period of time.
Instrumentation
Alpha particles, beta particles, gamma rays, and X-rays are measured by using the particle’s energy to produce an amplified pulse of electrical current in a detector. These pulses are counted to give the rate of disintegration. There are three common types of detectors: gas-filled detectors, scintillation counters, and semiconductor detectors. A gas-filled detector consists of a tube that contains an inert gas, such as Ar. When a radioactive particle enters the tube it ionizes the inert gas, producing an Ar+/e– ion-pair. Movement of the electron toward the anode and of the Ar+ toward the cathode generates a measurable electrical current. A Geiger counter is one example of a gas-filled detector. A scintillation counter uses a fluorescent material to convert radioactive particles into easy to measure photons. For example, one solid-state scintillation counter consists of a NaI crystal that contains 0.2% TlI, which produces several thousand photons for each radioactive particle. Finally, in a semiconductor detector, adsorption of a single radioactive particle promotes thousands of electrons to the semiconductor’s conduction band, increasing conductivity.
Quantitative Applications
In this section we consider three common quantitative radiochemical methods of analysis: the direct analysis of a radioactive isotope by measuring its rate of disintegration, neutron activation, and isotope dilution.
Direct Analysis of Radioactive Analytes
The concentration of a long-lived radioactive isotope remains essentially constant during the period of analysis. As shown in Example 13.3.1 , we can use the sample’s activity to calculate the number of radioactive particles in the sample.
The activity in a 10.00-mL sample of wastewater that contains \(_{38}^{90}\text{Sr}\) is \(9.07 \times 10^6\) disintegrations/s. What is the molar concentration of \(_{38}^{90}\text{Sr}\) in the sample? The half-life for \(_{38}^{90}\text{Sr}\) is 28.1 yr.
Solution
Solving Equation \ref{13.4} for \(\lambda\), substituting into Equation \ref{13.1}, and solving for N gives
\[N=\frac{A \times t_{1 / 2}}{0.693} \nonumber\]
Before we can determine the number of atoms of \(_{38}^{90}\text{Sr}\) in the sample we must express its activity and its half-life using the same units. Converting the half-life to seconds gives t1/2 as \(8.86 \times 10^8\) s; thus, there are
\[\frac{\left(9.07 \times 10^{6} \text { disintegrations/s }\right)\left(8.86 \times 10^{8} \text{ s}\right)}{0.693} = 1.16 \times 10^{16} \text{ atoms} _{38}^{90}\text{Sr} \nonumber\]
The concentration of \(_{38}^{90}\text{Sr}\) in the sample is
\[\frac{1.16 \times 10^{16} \text { atoms } _{38}^{90} \text{Sr}}{\left(6.022 \times 10^{23} \text { atoms/mol }\right)(0.01000 \mathrm{L})} = 1.93 \times 10^{-6} \text{ M } _{38}^{90}\text{Sr} \nonumber\]
The direct analysis of a short-lived radioactive isotope using the method outlined in Example 13.3.1 is less useful because it provides only a transient measure of the isotope’s concentration. Instead, we can measure its activity after an elapsed time, t, and use Equation \ref{13.3} to calculate N0.
Neutron Activation Analysis
Few analytes are naturally radioactive. For many analytes, however, we can induce radioactivity by irradiating the sample with neutrons in a process called neutron activation analysis (NAA). The radioactive element formed by neutron activation decays to a stable isotope by emitting a gamma ray, and, possibly, other nuclear particles. The rate of gamma-ray emission is proportional to the analyte’s initial concentration in the sample. For example, if we place a sample containing non-radioactive \(_{13}^{27}\text{Al}\) in a nuclear reactor and irradiate it with neutrons, the following nuclear reaction takes place.
\[_{13}^{27} \mathrm{Al}+_{0}^{1} \mathrm{n} \longrightarrow_{13}^{28} \mathrm{Al} \nonumber\]
The radioactive isotope of 13Al has a characteristic decay process that includes the release of a beta particle and a gamma ray.
\[_{13}^{28} \mathrm{Al} \longrightarrow_{14}^{28} \mathrm{Al}+_{-1}^{0} \beta + \gamma \nonumber\]
When irradiation is complete, we remove the sample from the nuclear reactor, allow any short-lived radioactive interferences to decay into the background, and measure the rate of gamma-ray emission.
The initial activity at the end of irradiation depends on the number of atoms that are present. This, in turn, is a equal to the difference between the rate of formation for \(_{13}^{28}\text{Al}\) and its rate of disintegration
\[\frac {dN_{_{13}^{28} \text{Al}}} {dt} = \Phi \sigma N_{_{13}^{27} \text{Al}} - \lambda N_{_{13}^{28} \text{Al}} \label{13.5}\]
where \(\Phi\) is the neutron flux and \(\sigma\) is the reaction cross-section, or probability that a \(_{13}^{27}\text{Al}\) nucleus captures a neutron. Integrating Equation \ref{13.5} over the time of irradiation, ti, and multiplying by \(\lambda\) gives the initial activity, A0, at the end of irradiation as
\[A_0 = \lambda N_{_{13}^{28}\text{Al}} = \Phi \sigma N_{_{13}^{27}\text{Al}} (1-e^{-kt}) \nonumber\]
If we know the values for A0, \(\Phi\), \(\sigma\), \(\lambda\), and ti, then we can calculate the number of atoms of \(_{13}^{27}\text{Al}\) initially present in the sample.
A simpler approach is to use one or more external standards. Letting \((A_0)_x\) and \((A_0)_s\) represent the analyte’s initial activity in an unknown and in an external standard, and letting \(w_x\) and \(w_s\) represent the analyte’s weight in the unknown and in the external standard, we obtain the following pair of equations
\[\left(A_{0}\right)_{x}=k w_{x} \label{13.6}\]
\[\left(A_{0}\right)_{s}=k w_{s} \label{13.7}\]
that we can solve to determine the analyte’s mass in the sample.
As noted earlier, gamma ray emission is measured following a period during which we allow short-lived interferents to decay into the background. As shown in Figure 13.3.1 , we determine the sample’s or the standard’s initial activity by extrapolating a curve of activity versus time back to t = 0. Alternatively, if we irradiate the sample and the standard at the same time, and if we measure their activities at the same time, then we can substitute these activities for (A0)x and (A0)s. This is the strategy used in the following example.
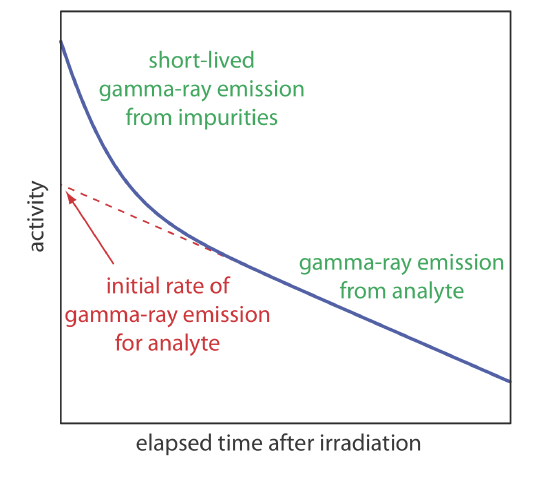
The concentration of Mn in steel is determined by a neutron activation analysis using the method of external standards. A 1.000-g sample of an unknown steel sample and a 0.950-g sample of a standard steel known to contain 0.463% w/w Mn are irradiated with neutrons for 10 h in a nuclear reactor. After a 40-min delay the gamma ray emission is 2542 cpm (counts per minute) for the unknown and 1984 cpm for the external standard. What is the %w/w Mn in the unknown steel sample?
Solution
Combining equations \ref{13.6} and \ref{13.7} gives
\[w_{x}=\frac{A_{x}}{A_{s}} \times w_{s} \nonumber\]
The weight of Mn in the external standard is
\[w_{s}=\frac{0.00463 \text{ g } \text{Mn}}{\text{ g } \text { steel }} \times 0.950 \text{ g} \text { steel }=0.00440 \text{ g} \text{ Mn} \nonumber\]
Substituting into the above equation gives
\[w_{x}=\frac{2542 \text{ cpm}}{1984 \text{ cpm}} \times 0.00440 \text{ g} \text{ Mn}=0.00564 \text{ g} \text{ Mn} \nonumber\]
Because the original mass of steel is 1.000 g, the %w/w Mn is 0.564%.
Among the advantages of neutron activation are its applicability to almost all elements in the periodic table and that it is nondestructive to the sample. Consequently, NAA is an important technique for analyzing archeological and forensic samples, as well as works of art.
Isotope Dilution
Another important radiochemical method for the analysis of nonradioactive analytes is isotope dilution. An external source of analyte is prepared in a radioactive form with a known activity, AT, for its radioactive decay—we call this form of the analyte a tracer. To prepare a sample for analysis we add a known mass of the tracer, wT, to a portion of sample that contains an unknown mass, wx , of analyte. After homogenizing the sample and tracer, we isolate wA grams of analyte by using a series of appropriate chemical and physical treatments. Because these chemical and physical treatments cannot distinguish between radioactive and nonradioactive forms of the analyte, the isolated material contains both. Finally, we measure the activity of the isolated sample, AA. If we recover all the analyte—both the radioactive tracer and the nonradioactive analyte—then AA and AT are equal and wx = wA – wT. Normally, we fail to recover all the analyte. In this case AA is less than AT, and
\[A_{A}=A_{T} \times \frac{w_{A}}{w_{x}+w_{T}} \label{13.8}\]
The ratio of weights in Equation \ref{13.8} accounts for any loss of activity that results from our failure to recover all the analyte. Solving Equation \ref{13.8} for wx gives
\[w_{x}=\frac{A_{T}}{A_{A}} w_{A}-w_{T} \label{13.9}\]
How we process the sample depends on the analyte and the sample’s matrix. We might, for example, digest the sample to bring the analyte into solution. After filtering the sample to remove the residual solids, we might precipitate the analyte, isolate it by filtration, dry it in an oven, and obtain its weight.
Given that the goal of an analysis is to determine the amount of nonradioactive analyte in our sample, the realization that we might not recover all the analyte might strike you as unsettling. Recall from Chapter 7.7, that a single liquid–liquid extraction rarely has an extraction efficiency of 100%. One advantage of isotope dilution is that the extraction efficiency for the nonradioactive analyte and for the tracer are the same. If we recover 50% of the tracer, then we also recover 50% of the nonradioactive analyte. Because we know how much tracer we added to the sample, we can determine how much of the nonradioactive analyte is in the sample.
The concentration of insulin in a production vat is determined by isotope dilution. A 1.00-mg sample of insulin labeled with 14C having an activity of 549 cpm is added to a 10.0-mL sample taken from the production vat. After homogenizing the sample, a portion of the insulin is separated and purified, yielding 18.3 mg of pure insulin. The activity for the isolated insulin is measured at 148 cpm. How many mg of insulin are in the original sample?
Solution
Substituting known values into Equation \ref{13.8} gives
\[w_{x}=\frac{549 \text{ cpm}}{148 \text{ cpm}} \times 18.3 \text{ mg}-1.00 \text{ mg}=66.9 \text{ mg} \text { insulin } \nonumber\]
Equation \ref{13.8} and Equation \ref{13.9} are valid only if the tracer’s half-life is considerably longer than the time it takes to conduct the analysis. If this is not the case, then the decrease in activity is due both to the incomplete recovery and the natural decrease in the tracer’s activity. Table 13.3.1 provides a list of several common tracers for isotope dilution.
| isotope | half-life |
|---|---|
| 3H | 12.5 years |
| 14C | 5730 years |
| 32P | 14.3 days |
| 35S | 87.1 days |
| 45Ca | 152 days |
| 55Fe | 2.91 years |
| 60Co | 5.3 years |
| 131I | 8 days |
An important feature of isotope dilution is that it is not necessary to recover all the analyte to determine the amount of analyte present in the original sample. Isotope dilution, therefore, is useful for the analysis of samples with complex matrices, where a complete recovery of the analyte is difficult.
Characterization Applications
One example of a characterization application is the determination of a sample’s age based on the decay of a radioactive isotope naturally present in the sample. The most common example is carbon-14 dating, which is used to determine the age of natural organic materials.
As cosmic rays pass through the upper atmosphere, some \(_7^{14}\text{N}\) atoms in the atmosphere capture high energy neutrons, converting them into \(_6^{14}\text{C}\). The \(_6^{14}\text{C}\) then migrates into the lower atmosphere where it oxidizes to form C-14 labeled CO2. Animals and plants subsequently incorporate this labeled CO2 into their tissues. Because this is a steady-state process, all plants and animals have the same ratio of \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) in their tissues. When an organism dies, the radioactive decay of \(_6^{14}\text{C}\) to \(_7^{14}\text{N}\) by \(_{-1}^0 \beta\) emission (t = 5730 years) leads to predictable reduction in the \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) ratio. We can use the change in this ratio to date samples that are as much as 30000 years old, although the precision of the analysis is best when the sample’s age is less than 7000 years. The accuracy of carbon-14 dating depends upon our assumption that the natural \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) ratio in the atmosphere is constant over time. Some variation in the ratio has occurred as the result of the increased consumption of fossil fuels and the production of \(_6^{14}\text{C}\) during the testing of nuclear weapons. A calibration curve prepared using samples of known age—examples of samples include tree rings, deep ocean sediments, coral samples, and cave deposits—limits this source of uncertainty.
There is no need to prepare a calibration curve for each analysis. Instead, there is a universal calibration curve known as IntCal. The most recent such curve, IntCal13 is described in the following paper: Reimer, P. J., et. al. “IntCal13 and Marine 13 Radiocarbon Age Calibration Curve 0–50,000 Years Cal BP,” Radiocarbon 2013, 55, 1869–1887. This calibration spans 50 000 years before the present (BP).
To determine the age of a fabric sample, the relative ratio of \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) was measured yielding a result of 80.9% of that found in modern fibers. How old is the fabric?
Solution
Equation \ref{13.3} and Equation \ref{13.4} provide us with a method to convert a change in the ratio of \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) to the fabric’s age. Letting A0 be the ratio of \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) in modern fibers, we assign it a value of 1.00. The ratio of \(_6^{14}\text{C}\) to \(_6^{12}\text{C}\) in the sample, A, is 0.809. Solving gives
\[t=\ln \frac{A_{0}}{A} \times \frac{t_{1 / 2}}{0.693}=\ln \frac{1.00}{0.809} \times \frac{5730 \text { yr }}{0.693}=1750 \text { yr } \nonumber\]
Other isotopes can be used to determine a sample’s age. The age of rocks, for example, has been determined from the ratio of the number of \(_{92}^{238}\text{U}\) to the number of stable \(_{82}^{206}\text{Pb}\) atoms produced by radioactive decay. For rocks that do not contain uranium, dating is accomplished by comparing the ratio of radioactive \(_{19}^{40}\text{K}\) to the stable \(_{18}^{40}\text{Ar}\). Another example is the dating of sediments collected from lakes by measuring the amount of \(_{82}^{210}\text{Pb}\) that is present.
Evaluation
Radiochemical methods routinely are used for the analysis of trace analytes in macro and meso samples. The accuracy and precision of radiochemical methods generally are within the range of 1–5%. We can improve the precision—which is limited by the random nature of radioactive decay—by counting the emission of radioactive particles for as long a time as is practical. If the number of counts, M, is reasonably large (M ≥ 100), and the counting period is significantly less than the isotope’s half-life, then the percent relative standard deviation for the activity, \((\sigma_A)_{rel}\), is approximately
\[\left(\sigma_{A}\right)_{\mathrm{rel}}=\frac{1}{\sqrt{M}} \times 100 \nonumber\]
For example, if we determine the activity by counting 10 000 radioactive particles, then the relative standard deviation is 1%. A radiochemical method’s sensitivity is inversely proportional to \((\sigma_A)_{rel}\), which means we can improve the sensitivity by counting more particles.
Selectivity rarely is of concern when using a radiochemical method because most samples have only a single radioactive isotope. When several radioactive isotopes are present, we can determine each isotope’s activity by taking advantage of differences in the energies of their respective radioactive particles or differences in their respective decay rates.
In comparison to most other analytical techniques, radiochemical methods usually are more expensive and require more time to complete an analysis. Radiochemical methods also are subject to significant safety concerns due to the analyst’s potential exposure to high energy radiation and the need to safely dispose of radioactive waste.


