21.10: Spectroscopy of Carboxylic Acid Derivatives
- Page ID
- 36407
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- identify the region of the infrared spectrum in which absorptions resulting from the carbonyl group of carboxylic acid derivatives occur.
- identify the characteristic features of the infrared spectra of acid anhydrides and nitriles that allow us to use infrared spectroscopy to distinguish between these compounds and the others discussed in this unit.
- use a table of characteristic infrared absorptions to interpret the infrared spectra of carbonyl‑containing compounds.
- use infrared spectroscopy and 1H NMR spectroscopy in the deduction of the structure of an unknown compound.
Note: You should recognize that protons on carbon atoms next to carbonyl groups are slightly deshielded, and absorb near 2 δ in the 1H NMR spectrum.
The influence of heteroatom substituents on the reactivity of carbonyl functions toward nucleophiles was discussed earlier with respect to carboxylic acid derivatives. A useful relationship exists between the reactivity of these derivatives and their carbonyl stretching frequencies. Thus, the very reactive acyl halides and anhydrides absorb at frequencies significantly higher than ketones, whereas the relatively unreactive amides absorb at lower frequencies. These characteristics are listed below.
Infrared spectra of many carboxylic acid derivatives will be displayed in the figure below the table by clicking the appropriate buttons presented there.
| Carbonyl Derivative | Carbonyl Absorption | Comments |
|---|---|---|
|
Acyl Halides (RCOX) |
C=O stretch |
Conjugation lowers the C=O frequencies reported here, as with aldehydes & ketones. In acyl chlorides a lower intensity shoulder or peak near 1740 cm-1 is due to an overtone interaction. |
|
Acid Anhydride, (RCO)2O |
C=O stretch (2 bands) |
Conjugation lowers the C=O frequencies reported here, as with aldehydes & ketones. The two stretching bands are separated by 60 ± 30 cm-1, and for acyclic anhydrides the higher frequency (asymmetric stretching) band is stronger than the lower frequency (symmetric) absorption. Cyclic anhydrides also display two carbonyl stretching absorptions, but the lower frequency band is the strongest. One or two -CO-O-CO- stretching bands are observed in the 1000 to 1300 cm-1 region. |
|
Esters & Lactones (RCOOR') |
C=O stretch |
Conjugation lowers the C=O frequencies reported here, as with aldehydes & ketones Strong CO-O stretching absorptions (one ot two) are found from 1150 to 1250 cm-1 |
|
Amides & Lactams (RCONR2) |
C=O bands |
The effect of conjugation is much less than for aldehydes & ketones. The higher frequency absorption (1665± 30) is called the Amide I band. The lower frequency Amide II band (1620± 30 in 1° amides & 1530± 30 in 2° amides) is largely due to N-H bending trans to the carbonyl oxygen. In concentrated samples this absorption is often obscured by the stronger amide I absorption. Hydrogen bonded association shifts some of these absorptions, as well as the prominent N-H stretching absorptions. N-H stretch: 3170 to 3500 cm-1. Two bands for 1°-amides, one for 2°-amides. |
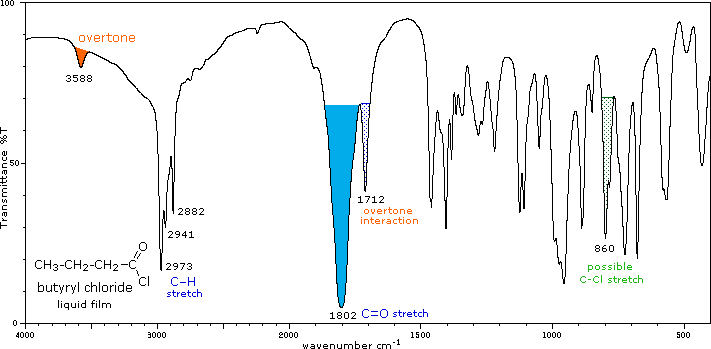
IR - Carbonyl peak of an acid halide
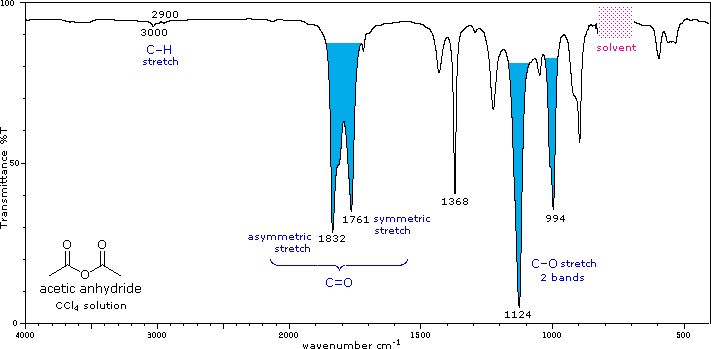
IR - Carbonyl and carbon-oxygen single bond peaks of an acid anhydride
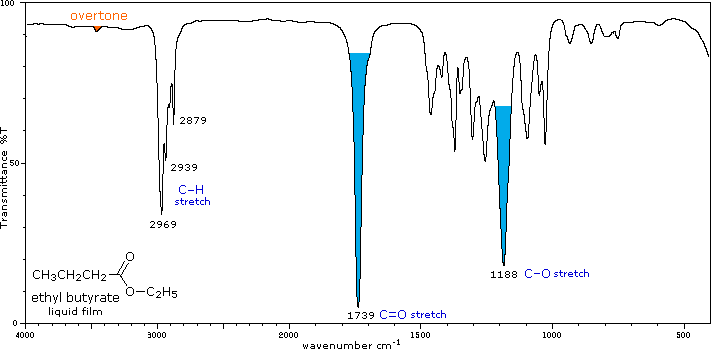
IR - Carbonyl and carbon-oxygen single bond peaks of an ester
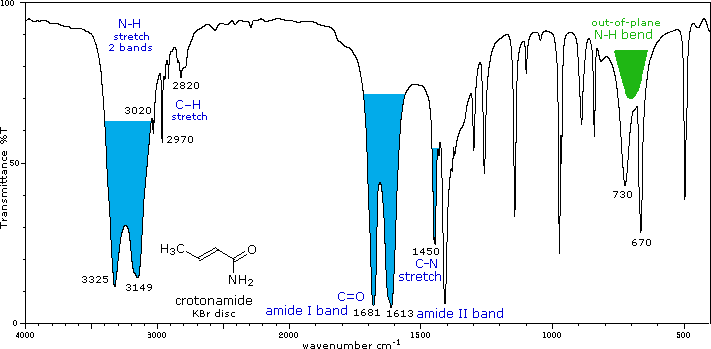
IR - Carbonyl, carbon-nitrogen and nitrogen-hydrogen bond peaks of an amide
1H Nuclear Magnetic Spectroscopy
Alpha protons of carboxylic acid derivatives, due to the presence of a carbonyl, resonate in the 2.0-3.0 ppm region of a 1H NMR spectra. The downfield shift occurs from deshielding due to higher electronegativity of the sp2 hybridized carbonyl carbon relative to the sp3 hybridized alpha carbons. This effect is reduced for protons which are further from the carbonyl group as seen in the figure below.
Acid halides
The X groups of acid halides do not contain additional protons and thus do not provide any distinctive 1H NMR peaks. The protons on carbon adjacent to the carbonyl appear at similar chemical shift to other carbonyl containing compounds (2-3 ppm).
Acid anhydrides
Many anhydrides used in organic chemistry are symmetrical, therefore the alkyl chains on either side of the carbonyls produce equivalent protons.
Esters and Thioesters
The Y groups of esters, and thioesters have alkyl groups attached to their heteroatom which provide additional 1H NMR peaks. Protons on carbons attached to the alkoxide oxygen in esters show up in the 3.5-4.5 ppm region while those attached to the sulfur in thioesters show up in the 2.0-3.0 ppm region. The downfield shift in these protons is attributable to the deshielding caused by the electronegativity of oxygen and sulfur.
Amides
Amides have the possibility of alkyl groups or hydrogens attached to their Y group both of which provide distinctive 1H NMR peaks. Resonances from protons directly attached to the carbon bonded to nitrogen in an amide tend to resonate in the 2.0-3.0 ppm region of an NMR spectra. The NH protons of primary and secondary amides resonate in the 7.5-8.5 ppm regions. Like other hydrogen bonding protons, these NH proton peaks appear broad and can be washed out by the hydrogen-deuterium exchange created by D2O addition.
An interesting facet of amide structure is shown when looking at the 1H NMR spectra of N,N-dimethylacetamide. The two N-methyl groups are in fact non-equivalent and appear as two singlets with difference chemical shifts. Resonance in the amide bond provides a significant amount of double bond character reducing the rate of rotation about the OC-N bond. The rotation is slow enough that on the time scale of an NMR experiment the two methyl appears to be frozen as if it is part of a double bond. This causes the N-methyl groups to have different chemical shifts because one appears to be cis to the carbonyl oxygen and the other trans during an NMR experiment.
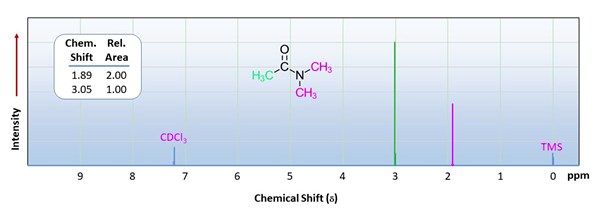
13C Nuclear Magnetic Spectroscopy
The carbonyl carbons of carboxylic acid derivatives are strongly deshielded (160-180 ppm) due to the close presence (due to the double bond) of a highly electronegative oxygen. However, they are not as deshielded as the carbonyl carbon of an aldehyde or ketone (180-220 ppm) due to the electron donating effects of the Y group. Carbons attached to the alkoxide oxygen of esters appear in the range of 50-90 ppm. Carbons attached to the nitrogen of an amide appear in the 20-65 ppm range and those attached to the sulfur of thioesters appear at 20-45 ppm.
Acid halide 13C NMR chemical shifts (ppm)
Anhydride 13C NMR chemical shifts (ppm)
Ester 13C NMR chemical shifts (ppm)
Amide 13C NMR chemical shifts (ppm)
UV/Vis Spectroscopy
Carboxylic acid derivatives show the n→π* absorption typical for carbonyl containing compounds. The Y group of acid derivatives give a hypsochromic shift (wavelength lowering) to the absorption due to the inductive effect of the electronegative oxygen, nitrogen, and halogens present. Electrons are inductively removed from the carbonyl carbon which causes the lone pair electrons on the carbonyl oxygen to be held more firmly. This causes them to require higher energy photons to transition which corresponds to a lower wavelength λmax when compared to other carbonyls such as a ketone.
UV-Visible absorption of carbonyl compounds (showing λmax in nm)
Mass Spectrometry
In the mass spectra of most carboxylic acid derivatives, the base peak is due to the cleavage of C-Y bond to form an acylium ion (R-CO+). Other than the molecular ion peak, the acylium ion is one of the only major peaks present in the mass spectra of acid halides and thioesters. Amide and ester often show additional fragmentations.
Acid halides
The molecular ion peak of acid chlorides is often not seen. When they do occur, acid chlorides and acid bromides would have an important M + 2 peak due to the two isotopic forms of the halogens. The base peak fragmentation involves the alpha cleavage of C-X bond to from an acylium ion. In butanoyl chloride this peak shows up at m/z = 72.
Amide
Amides, like other compounds containing nitrogen, follow the Nitrogen Rule. This means that a compound with an odd number of nitrogen atoms will also have an odd-numbered molecular weight. This is shown in butanamide (C4H9NO) which has the odd-numbered molecular weight of 87.12 g/mol.
Primary amides show a base peak due to the McLafferty rearrangement which appears at m/z = 59. Secondary and tertiary amides give analogous peaks at higher m/z values.
Amides can also cleave the alkyl group attached to the carbonyl forming an amide stabilized acelium ion. For butanamide, like other primary amines, this peak shows up at m/z = 44.
Ester
Esters have multiple fragmentation pathways which are often dependent on the overall size of the ester. The most important fragmentation appears due to alpha-cleavage bond of the C-O to produce the corresponding acylium ion RCO+. In the mass spectra of ethyl acetate the acylium ion peak appears at 43 m/z and the alkoxy ion peak (RO+) shows up a 45 m/z. Other important fragments include the loss or the alkyl group (R+) from the acyl side and the alkoxy side of the ester. Loss of the alkyl group on the alkyoxy side of ethyl acetate (+CH2CH3) produces a peak at m/z 61.
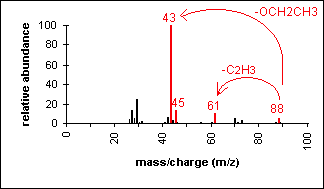
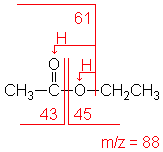
SDBSWeb : https://sdbs.db.aist.go.jp (National Institute of Advanced Industrial Science and Technology, date of access
NIST Chemistry WebBook: https://webbook.nist.gov/chemistry/ (National Insitute of Standards and Technology)

