2.10: Acids and Bases - The Lewis Definition
- Page ID
- 31393
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- state the Lewis definition of an acid and a base.
- identify a given compound as being a Lewis acid or Lewis base, given its Lewis structure or its Kekulé structure.
- identify an organic molecule or ion in a reaction as either an electrophile or nucleophile.
Make certain that you can define, and use in context, the key terms below.
- Lewis acid
- Lewis base
- Electrophile
- Nucleophile
The Lewis concept of acidity and basicity will be of great use to you when you study reaction mechanisms. The realization that an ion such as
is electron deficient, and is therefore a Lewis acid, should help you understand why this ion reacts with substances which are Lewis bases (e.g., H2O).
Introduction to Lewis Acid-Base Theory
The Brønsted acid-base theory has been used throughout the history of acid and base chemistry. However, this theory is very restrictive and focuses primarily on acids and bases acting as proton donors and acceptors. Sometimes conditions arise where the theory doesn't necessarily fit, such as in solids and gases. In 1923, G.N. Lewis from UC Berkeley proposed an alternate theory to describe acids and bases. His theory gave a generalized explanation of acids and bases based on structure and bonding. Through the use of the Lewis definition of acids and bases, chemists are now able to predict a wider variety of acid-base reactions. Lewis' theory used electrons instead of proton transfer and specifically stated that an acid is a species that accepts an electron pair while a base donates an electron pair.
Example of Lewis base (oxygen atom from carbonyl) reacting with Lewis Acid (Mg2+ ion).
The reaction of a Lewis acid and a Lewis base will produce a coordinate covalent bond. A coordinate covalent bond is just a type of covalent bond in which one reactant donates both electrons to form the bond. In this case the Lewis base donates its electrons to form a bond to the Lewis acid. The resulting product is called an addition compound, or more commonly a complex. The electron-pair flow from Lewis base to Lewis acid is shown using curved arrows much like those used for resonance structures in Section 2.5. Curved arrows always mean that an electron pair moves from the atom at the tail of the arrow to the atom at the head of the arrow. In this case the lone pair on the Lewis base attacks the Lewis acid forming a bond. This new type of electron pair movement, along with those described in Section 2.5 will be used throughout this text to describe electron flow during reactions.
- Lewis Acid: a species that accepts an electron pair and will typically either have vacant orbitals or a polar bond involving hydrogen such that it can donate H+ (which has an empty 1s orbital)
- Lewis Base: a species that donates an electron pair and will have lone-pair electrons of pi bonding electrons.
Lewis Acids
Neutral compounds of boron, aluminum, and the other Group 13 elements, (BF3, AlCl3), which possess only six valence electrons, have a very strong tendency to gain an additional electron pair. Because these compounds are only surrounded by three electron groups, they are sp2 hybridized, contained a vacant p orbital, and are potent Lewis acids. Trimethylamine's lone pair elections are contained in an sp3 hybrid orbital making it a Lewis base. These two orbitals overlap, creating a covalent bond in a boron trifluoride-trimethylamine complex. The movement of electrons during this interaction is show by by an arrow.
Positive ions are often Lewis acids because they have an electrostatic attraction for electron donors. Examples include alkali and alkaline earth metals in the group IA and IIA columns. K+, Mg2+ and Ca2+ are sometimes seen as Lewis acidic sites in biology, for example. These ions are very stable forms of these elements because of their low electron ionization potentials. However, their positive charges do attract electron donors.The interaction between a magnesium cation (Mg+2) and a carbonyl oxygen is a common example of a Lewis acid-base reaction. The carbonyl oxygen (the Lewis base) donates a pair of electrons to the magnesium cation (the Lewis acid). As we will see in Chapter 19 when we begin the study of reactions involving carbonyl groups, this interaction has the very important effect of increasing the polarity of the carbon-oxygen double bond.
The eight-electron rule does not hold throughout the periodic table. In order to obtain noble gas configurations, some atoms may need eighteen electrons in their valence shell. Transition metals such as titanium, iron and nickel may have up to eighteen electrons and can frequently accept electron pairs from Lewis bases. Transition metals are often Lewis acids. For example, titanium has four valence electrons and can form four bonds in compounds such as titanium tetrakis (isopropoxide), below, or titanium tetrachloride, TiCl4. However, the titanium atom in that compound has only eight valence electrons, not eighteen. It can easily accept electrons from donors.
For example, when THF and TiCl4 are combined, a Lewis acid-base complex is formed, TiCl4(THF)2. TiCl4(THF)2 is a yellow solid at room temperature.
The Proton as a Common Lewis Acid
Perhaps the most common example of a Lewis acid is also the simplest. It is the hydrogen cation (H+) or proton. It is called a proton because, in most hydrogen atoms, the only particle in the nucleus is a proton. If an electron is removed to make a cation, a proton is all that is left. A proton is a Lewis acid for a number of reasons. It has a positive charge, and so it will attract electrons, which are negative. Also, it lacks the electron configuration of its noble gas neighbor, helium. Helium has two electrons. If a Lewis base or nucleophile donates a pair of electrons to a proton, the proton will obtain a helium noble gas configuration.
There is something about hydrogen cations that is not so simple, however. They are actually not so common. Instead, protons are generally always bound to a Lewis base. Hydrogen is almost always covalently (or coordinately) bonded to another atom. Many of the other elements commonly found in compounds with hydrogen are more electronegative than hydrogen. As a result, hydrogen often has a partial positive charge making is still act as a Lewis Acid.
A acid-base reaction involving protons might better be expressed as:
The Lewis acid-base interactions we have looked at so far are slightly different here. Instead of two compounds coming together and forming a bond, we have one Lewis base replacing another at a proton. Two specific movements of electrons are shown in the reaction both of which are show by arrows. Lone pair electrons on oxygen attack the hydrogen to form an O-H bond in the product. Also, the electrons of the H-Cl bond move to become a lone pair on chlorine as the H-Cl bond breaks. These two arrows together are said to represent the mechanism of this acid-base reaction.
Lewis Bases
What makes a molecule (or an atom or ion) a Lewis base? It must have a pair of electrons available to share with another atom to form a bond. The most readily available electrons are those that are not already in bonds. Bonding electrons are low in energy. Non-bonding electrons are higher in energy and may be stabilized when they are delocalized in a new bond. Lewis bases usually have non-bonding electrons or lone pairs this makes oxygen and nitrogen compounds common Lewis bases. Lewis bases may be anionic or neutral. The basic requirement is that they have a pair of electrons to donate.
Ammonia, NH3, has a lone pair and is a Lewis base. It can donate to compounds that will accept electrons.
Ammonia donating to an electron acceptor or Lewis acid.
Not all compounds can act as a Lewis base. For example, methane, CH4, has all of its valence electrons in bonding pairs. These bonding pairs are too stable to donate under normal conditions therefore methane is not a Lewis base. Neutral boron compounds also have all electrons in bonding pairs. For example, borane, BH3 has no lone pairs; all its valence electrons are in bonds. Boron compounds are not typically Lewis bases.
Which of the following compounds would you expect to be Lewis bases?
a) SiH4 b) AlH3 c) PH3 d) SH2 e) -SH
- Answer
-
a) No, silicon has 4 valence electrons (like carbon) and all 4 are involved in sigma bonds
b) No, aluminum has 3 valence electrons and all 3 are involved in sigma bonds
c) Yes, phosphorus has 5 valence electrons, so there is one lone pair available
d) Yes, sulfur has 6 valence electrons, so there are two lone pairs available
e) Yes, this ion has 3 lone pairs available
Lewis Acid-Base Complexes
What happens when a Lewis base donates a pair of electrons to a Lewis acid? The electron pushing formalism (arrows) we have been using to illustrate the behavior of Lewis acids and Lewis bases is meant to show the direction of electron movement from the donor to the acceptor. However, given that a bond can be thought of as a pair of electrons that are shared between two atoms (in this case, between the donor and the acceptor), these arrows also show where bonds are forming.
The electrons donated from a Lewis base to a Lewis acid form a new bond. A new, larger compound is formed from the smaller Lewis acid and Lewis base. This compound is called a Lewis acid-base complex. A simple example of Lewis acid-base complexation involves ammonia and boron trifluoride. The nitrogen atom has a lone pair and is an electron donor. The boron has no octet and is an electron acceptor. The two compounds can form a Lewis acid-base complex or a coordination complex together.
When the nitrogen donates a pair of electrons to share with the boron, the bond that forms is sometimes called a coordinate bond. A coordinate bond is any covalent bond that arose because one atom brought a pair of its electrons and donated them to another.
In organic chemistry terminology, the electron donor is called a nucleophile and the electron acceptor is called an electrophile. Ammonia is a nucleophile and boron trifluoride is an electrophile.
- Because Lewis bases are attracted to electron-deficient atoms, and because positive charge is generally associated with the nucleus of an atom, Lewis bases are sometimes refered to as "nucleophiles". Nucleophile means nucleus-loving.
- Because Lewis acids attract electron pairs, Lewis acids are sometimes called "electrophiles". Electrophile means electron-loving.
For the following reaction, add curved arrows (electron pushing formalism) to indicate the electron flow.
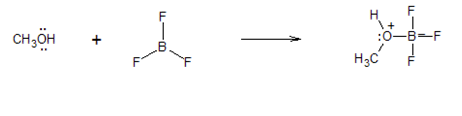
- Answer
-
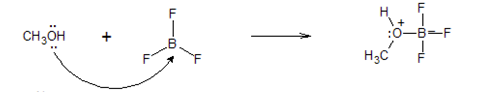
A Lewis acid-base complex is formed between THF (tetrahyrofuran) and borane, BH3.
a) Which compound is the Lewis acid? Which one is the Lewis base?
b) Which atom in the Lewis acid is the acidic site? Why?
c) Which atom in the Lewis base is the basic site? Why?
d) How many donors would be needed to satisfy the acidic site?
e) Show, using arrow notation, the reaction to form a Lewis acid-base complex.
f) Borane is highly pyrophoric; it reacts violently with air, bursting into flames. Show, using arrow notation, what might be happening when borane contacts the air.
g) Borane-THF complex is much less pyrophoric than borane. Why do you suppose that is so?
Add exercises text here.
- Answer
-
a) Borane is the Lewis acid. THF has lone pair electrons so it is the Lewis base.
b) The Boron atom has an unfilled octet so it has an empty p orbital that can accept electrons.
c) The oxygen atom in THF has lone pair electrons contained in a sp3 hybridized orbital.
d) The boron in borane has six electrons around it so it would only need one lone pair donor to reach an octet.
e) Show, using arrow notation, the reaction to form a Lewis acid-base complex.
f) The Borane initially reacts with water in air.
g) After the Borane-THF complex is formed, the boron atom has a complete octet making it less reactive.


