Chlorides of Group 4 Elements
- Page ID
- 3692
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page briefly examines the tetrachlorides of carbon, silicon, and lead, as well as lead(II) chloride. It considers the compounds' structures, stability, and reactions with water.
Structures
Carbon, silicon and lead tetrachlorides
Each of these compounds has the formula XCl4. They are simple covalent molecules with a typical tetrahedral shape. They are liquids at room temperature (although at room temperature, lead(IV) chloride will tend to decompose to give lead(II) chloride and chlorine gas—see the discussion below).
Lead(II) chloride, PbCl2
Lead(II) chloride is a white solid, melting at 501°C. It is slightly soluble in cold water, but its solubility increases with temperature. Lead(II) chloride is essentially ionic in character.
Stability
At the top of Group 4, the most stable oxidation state is +4. This is the oxidation state of carbon and silicon in CCl4 and SiCl4. These compounds have no tendency to break down into dichlorides. However, the relative stability of the +4 oxidation state decreases down the group, and the +2 oxidation state becomes the most stable for lead and below. Lead(IV) chloride decomposes at room temperature to form the more stable lead(II) chloride and chlorine gas.
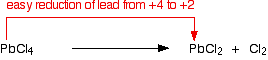
Reaction with water (hydrolysis)
Carbon tetrachloride (tetrachloromethane)
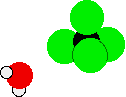
Carbon tetrachloride has no reaction with water. When added to water, it forms a separate layer underneath the layer of water. If a water molecule were to react with carbon tetrachloride, the oxygen atom in the water molecule would need to attach itself to the carbon atom via the oxygen's lone pair. A chlorine atom would be displaced the process. There are two problems with this idea.
First, chlorine atoms are so bulky and the carbon atom so small that the oxygen atom is sterically hindered from attacking the carbon atom.
Even if this were possible, there would be considerable cluttering around that carbon atom before the chlorine atom breaks away completely, causing a lot of repulsion between the various lone pairs on all the atoms surrounding the carbon, as shown below:
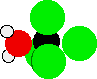
This repulsion makes the transition state very unstable. An unstable transition state indicates a high activation energy for the reaction.
The other problem is that there is no appropriate empty carbon orbital the oxygen lone pair can occupy.
If it attaches before the chlorine starts to break away, there would be an advantage. Forming a bond releases energy, and that energy would be readily available for breaking a carbon-chlorine bond. In the case of a carbon atom, however, this is impossible.
Silicon tetrachloride
The situation is different with silicon tetrachloride. Silicon is larger, so there is more room for the water molecule to attack; the transition is less cluttered. Silicon has an additional advantage: there are empty 3d orbitals available to accept a lone pair from the water molecule. Carbon lacks this advantage because there are no empty 2-level orbitals available.
The oxygen atom can therefore bond to silicon before a silicon-chlorine bond breaks, makes the whole process easier energetically. In practice, silicon tetrachloride therefore reacts violently with water, forming white solid silicon dioxide and HCl gas.
\[SiCl_4 + 2H_2O \rightarrow SiO_2 + 4HCl \nonumber \]
Liquid SiCl4 fumes in moist air for this reason—it reacts with water vapor in the air.
Lead tetrachloride (lead(IV) chloride)
The reaction of lead(IV) chloride with water is just like that of silicon tetrachloride. Lead(IV) oxide is produced as a brown solid, and fumes of hydrogen chloride given off (this can be confused with the decomposition of the lead(IV) chloride, which gives lead(II) chloride and chlorine gas as mentioned above).
\[ PbCl_4 + 2H_2O \rightarrow PbO_2 + 4HCl \nonumber \]
Lead(II) chloride
Unlike the tetrachlorides, lead(II) chloride can be considered ionic in nature. It is slightly soluble in cold water, but more soluble in hot water. Water solubility involves disruption of the ionic lattice and hydration of the lead(II) and chloride ions to give Pb2+(aq) and Cl-(aq).
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


