1.9: Chemical Reactivity - The Basics of Combustion
- Page ID
- 212612
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)When discussing the combustion of a compound it is ordinarily referring to the reaction of an organic compound (hydrocarbon) with oxygen, in which the carbon is converted to carbon dioxide (CO2) and the hydrogen forms water (H2O) as a vapor, e.g., (1.9.1).
\[CH_4 + 2 O_2 \rightarrow CO_2 + 2 H_2O \]
However, this only a narrow view of combustion, and a more general denition should be that combustion or burning is the sequence of exothermic chemical reactions between a fuel and an oxidant accompanied by the production of heat and conversion of chemical species. Based upon this definition methane can combust in the presence of fluorine (F2) as strong oxidant, (1.9.2).
\[CH_4 + 4 F_2 \rightarrow CF_4 + 4 HF\]
In considering the combustion of any flammable compound, for example gasoline, it should be noted that the compounds that make up gasoline are quiet stable in the absence of a source of oxygen (usually from the air). Furthermore, some form of energy input (heat, flame, or spark) must be provided. Thus, the combustion of gasoline provides the archetypal example of the three component explosive system typical of a traditional chemical explosive: fuel (something that will burn), an oxidizer (usually a source of oxygen), and energy (ignition). In this regard, combustion also includes the exothermic reactions of many metals with oxygen, (1.9.3).
\[6Al + 3 O_2 \rightarrow 2 Al_2O_3\]
It is not just reactive metals that can be used as the fuel component of combustion, but many of their compounds as well. The formation of water from the hydrogen in organic compounds, in combination with an oxygen source, releases significant energy. It stands to reason therefore that any compound comprising of hydrogen and an element can be a potential fuel: a compound of hydrogen and another element is known as a hydride. This is especially true for the hydrides of reactive metals such as aluminum and sodium (and metalloids such as boron, (1.9.4)), but is also true for the hydrides of silicon and phosphorus. These hydride compounds react with an oxidizer in a manner analogous to that of a hydrocarbon, as may be seen by a comparison of (1.9.5) and (1.9.1).
\[2BH_3 + 3 O_2 \rightarrow B_2O_3 + 3 H_2O \]
\[SiH_4 + 2 O_2 \rightarrow SiO_2 + 2 H_2O \]
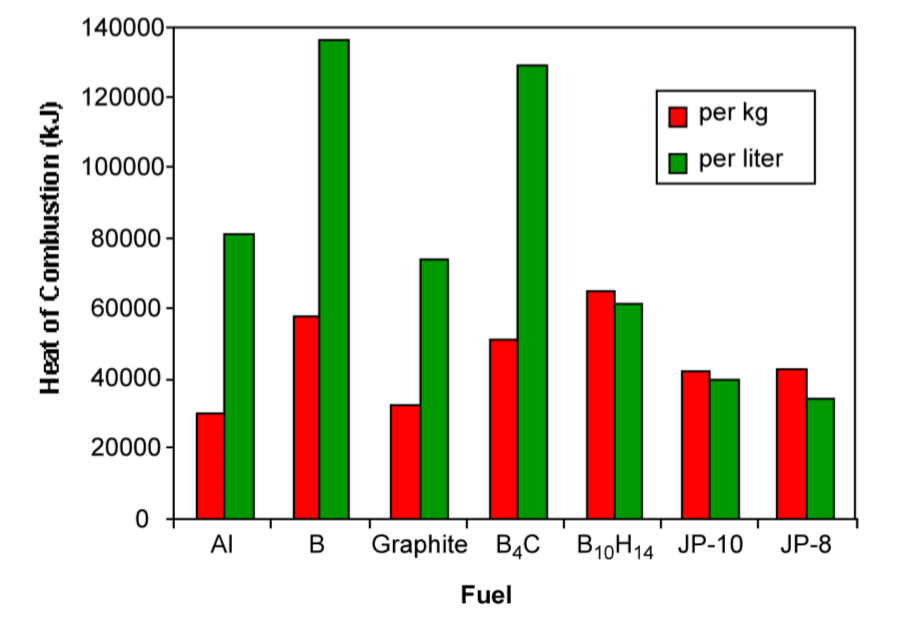
Figure \(\PageIndex{1}\).64 shows a comparison of the heat of combustion of various inorganic fuels with commonly used jet fuels (JP-10 and JP-8). Hydrocarbons heat of combustion is limited by the C:H ratio, while graphite is the limiting case. Boron and boron compounds have greater volumetric and gravimetric energy density than hydrocarbons, and therefore were studied as potential high-energy fuels.
Note
Ammonia has been proposed as a practical alternative to fossil fuel for internal combustion engines. The energy value of ammonia is 22.5 MJ/kg, which is about half that of diesel. In a normal engine, in which the water vapor is not condensed, the caloric value of ammonia will be about 21% less than this value; however, it can be used in existing engines with only minor modifications to carburetors/fuel injectors.
Oxygen (O2) from the air does not have to be the source of oxidizer. Alternatives such as hydrogen peroxide (H2O2), nitrous oxide (N2O), and nitrates (e.g., ammonium nitrate, NH4(NO3)) are all sources of oxygen for combustion or explosions. However, this does not mean that every compound containing oxygen can be an oxidizer. For example, alcohols such as methanol will burn (in the presence of additional oxygen), but they will not act as an oxidizer. The oxygen within any compound must be reactive. By this we mean that it must be able to be released, preferentially as the more reactive oxygen atom (O) rather than O2, or be attached (chemically bonded to an element that wants to get rid of the oxygen (i.e., an element that is readily reduced). The most likely element in this case is nitrogen, with sulfur and phosphorus also potential candidates. Almost all compounds containing nitrogen bonded to oxygen can act as an oxidizer. Generally, the more oxygen atoms attached to nitrogen the more reactive the compound.
The efficiency of a chemical reaction such as combustion is dependent on how well the fuel and oxidizer are mixed at the molecular scale. Obviously the best situation is if both components are in the same molecule. Self-oxidizers are compounds containing oxygen in a reactive form as well as a suitable fuel (carbon or hydrogen). The most common self-oxidizers are organic nitrates. It should be pointed out that in spite of the presence of reactive oxygen, self-oxidizers may still require an external source of oxygen to ensure complete reaction, and some form of energy input (ignition) is still required.
Oxygen balance
While many compounds contain oxygen that does not mean they will combust efficiently in the absence of an external oxidizer, or whether they have sufficient oxygen to completely self-combust, or whether they can act as an oxidizer for other compounds. The simplest test for a compound's potential to fulfill these roles is its oxygen balance. The oxygen balance for a chemical is the amount of oxygen needed or produced to ensure the complete oxidation of all the carbon, hydrogen, or other elements.
Compounds such as trinitrotoluene (TNT, Figure \(\PageIndex{1}\).65a) have a negative oxygen balance since extra oxygen is needed for complete formation of all the CO2 and H2O possible, (1.9.6). Thus, despite its reputation as an explosive, TNT is only efficient in the presence of an external oxidant, which may be air or another compound that provides a positive oxygen balance.
\[2 C_7H_5N_3O_6 + 21 "O" \rightarrow 14 CO_2 + 5 H_2O + 3N_2\]
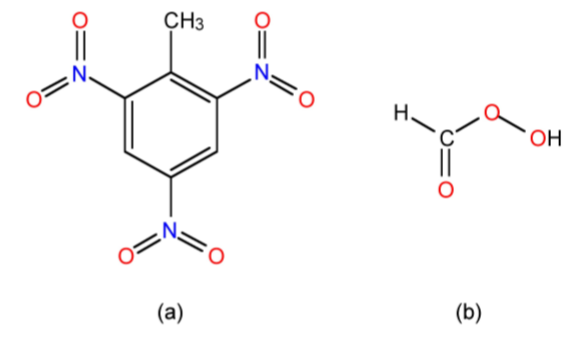
In contrast to TNT, performic acid (Figure \(\PageIndex{1}\).65b) is an example of a compound with a zero oxygen balance: it has all the oxygen it needs for complete combustion, (1.9.7), and hence only requires energy to detonate making at a much more dangerous compound per se than TNT.
\[2 CH_2O_3 \rightarrow CO_2 + H_2O \]
A positive oxygen balance means that the compound liberates oxygen surplus to its own needs, for example the decomposition of ammonium nitrate provides one atom of oxygen per molecule, (1.9.8). Clearly, any compound with a positive oxygen balance makes a good oxidizer and is highly incompatible with combustible chemicals.
\[NH_4(NO_3) \rightarrow 2 H_2O + N_2 + "O" \]
Quantification of oxygen balance
A quantification of oxygen balance allows for the determination of approximate ratio of reagents to optimize combustion/explosion. In this regard oxygen balance is defined as the number of moles of oxygen (excess or deficient) for 100 g of a compound of a known molecular weight (Mw), (1.8.9) where x = number of atoms of carbon, y = number of atoms of hydrogen, z = number of atoms of oxygen, and m = number of atoms of metal oxide produced.
\[OB\% = \dfrac{-1600}{M_w} \times (2x + \dfrac{y}{2} + m - z)\]
Example \(\PageIndex{1}\)
In the case of TNT, the Mw = 227.1 g/mol, the number of carbon atoms (x) = 7, the number of hydrogen atoms (y) = 5, the number of oxygen atoms (z) = 6, and the number of atoms of metal oxide produced (m) = 0. Therefore:
\[OB\% = \dfrac{-1600}{227.1} \times (14 + 2.5 + 0 - 6) = -74\%\]
A summary of selected oxygen balance values is given in Table \(\PageIndex{1}\).11.
| Element or Compound | Oxygen balance (%) |
|---|---|
| Carbon | -266.7 |
| Sulfur | -100 |
| Aluminum powder | -89 |
| Trinitrotoluene | -74 |
| Nitroglycerine | +3.5 |
| Ammonium nitrate | +20 |
| Ammonium perchlorate | +27 |
| Potassium chlorate | +26 |
| Sodium chlorate | +45 |
| Sodium nitrate | +37 |
| Tetranitromethane | +57 |
| Lithium perchlorate | +45 |


