1.8: Acids, Bases, and Solvents - Choosing a Solvent
- Page ID
- 212611
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The choice of solvent is an important parameter for any chemical reaction. The following provides a guide to some of the consideration to be made in choosing a solvent to ensure the desired reaction occurs.
Solvation
Solvation may be defined as the interaction between the solvent and the solute, however, two general classes of solvation have different consequences to the stability of either reagents or products in a chemical reaction, and hence the potential of a reaction to occur
- Specific solvation is where the solvent interacts with one of the ions (or molecules) in solution via a covalent interaction. Furthermore, there will be a specific number of solvent molecules bound to each ion (or molecule), e.g., [Cu(NH3)4]2+ and [Mg(H2O)6]2+ (Figure \(\PageIndex{1}\).60a).
- Non-specific solvation is as a result of van der Waals or dipole-dipole forces between the solvent and an ion (or molecule). There will be no defined number of interactions and the solvent...ion interaction will be highly fluxional, e.g., while water solvates the chloride ion (Figure \(\PageIndex{1}\).60b) the number of water molecules around each anion is not fixed
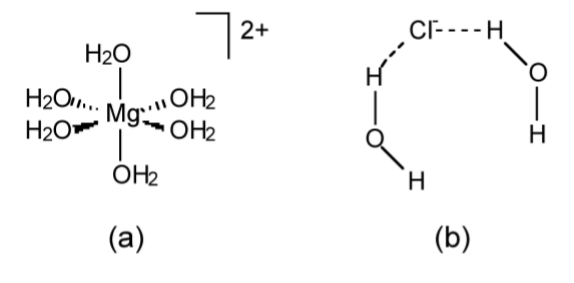
Table \(\PageIndex{1}\).10 shows the ability of three solvents to act with specific and non-specific solvation. The relative solvation ability of each solvent results in three different products from the dissolution of iron(III) chloride (FeCl3).
| Solvation | DMSO (Me2SO) | Pyridine (C5H5N) | Acetonitrile (MeCN) |
|---|---|---|---|
| Specific | Good | Very good | Poor |
| Non-specific | Good | Poor | Moderately good |
Dissolution of FeCl3 in DMSO results in the dissociation of a chloride ligand, (1.8.1), due to both the specific solvation of the FeCl2+ cation and the non-specific solvation of the Cl- anion. In fact, the good solvation properties of DMSO means that depending on the concentration (and temperature) a series of dissociations may occur, (1.8.2).
\[ FeCl_3 \overset{DMSO}{\rightleftharpoons} [FeCl_2(DMSO)_4]^+ Cl^-_{(solv)} \]
\[[FeCl_2(DMSO)_4]^+ \xleftrightarrow[-Cl^-_{(solv)}]{+DMSO}[FeCl(DMSO)_5]^{2+} \xleftrightarrow[-Cl^-_{(solv)}]{+ DMSO} [Fe(DMSO)_6]^{3+}\]
In contrast, if FeCl3 is dissolved in pyridine (py) the neutral Lewis acid-base complex is formed, (1.8.3), because while pyridine is a very good at specific solvation (Table \(\PageIndex{1}\).10), it is poor at solvating the chloride anion.
\[FeCl_3 + py \rightarrow FeCl_3(py)\]
In a similar manner, FeCl3(MeCN)3 will be formed by the dissolution in acetonitrile, because although it is not good at specific solvation, it is not sufficiently good at non-specific solvation to stabilize the chloride anion. However, since the FeCl4- anion has a lower charge density that Cl-, it can be supported by the non-specific solvation of acetonitrile and thus a disproportionation reaction occurs, (1.8.4).
\[ 2 FeCl_3(MeCN)_3 \rightleftharpoons [FeCl_2(MeCN)_4]^+ + FeCl_4^-\]
Interference by the solvent
Rather than solvating a molecule or ion, the solvent can take an active and detrimental role in the synthesis of a desired compound.
Solvolysis
The archetypal solvolysis reaction is the reaction with water, i.e., hydrolysis, (\ref{1.8.5}). However, solvolysis is a general reaction, involving bond breaking by the solvent. Thus, the reaction with ammonia is ammonolysis, (\ref{1.8.6}), the reaction with acetic acid is acetolysis, (\ref{1.8.7}), and the reaction with an alcohol is alcoholysis, (\ref{1.8.8}) where Et = \(\ce{C2H5}\). In each case the same general reaction takes place yielding the cation associated with the solvent.
\[\ce{SO_2Cl_2 + 4 H_2O \rightarrow SO_2(OH)_2 + 2 H_3O^+ + 2 Cl^-} \label{1.8.5}\]
\[\ce{SO_2Cl_2 + 4 NH_3 \rightarrow SO_2(NH_2)_2 + 2 NH_4^+ + 2 Cl^-} \label{1.8.6}\]
\[\ce{ SO_2Cl_2 + 4 MeCO_2H \rightarrow SO_2(O_2CMe)_2 + 2 MeCO_2H_2^+ + 2 Cl^-} \label{1.8.7}\]
\[\ce{SO_2Cl_2 + 4 EtOH \rightarrow SO_2(OEt)_2 + 2 EtOH_2^+ + 2 Cl^-} \label{1.8.8}\]
Competition reactions
Where more than one reaction could occur the reaction involving the solvent can often compete with the desired reaction.
If it is desired to synthesis the Lewis acid-base complex between diethylether (Et2O) and boron triuoride (BF3) it is important to choose a solvent that will not compete with the complex formation. For example, pyridine is a poor choice because the nitrogen donor is a stronger Lewis base than the diethylether, (1.8.9), and thus no reaction would occur between diethylether and boron trifluoride. In contrast, since acetonitrile (MeCN) is a poor Lewis base, then the reaction will occur.
\[BF_3(Et_2O) + py \rightarrow BF_3(py) + Et_2O\]
If the synthesis of GeCl62- from germanium tetrachloride (GeCl4) and a source of chloride anion, then water would be a poor choice of solvent since hydrolysis of GeCl4 would result. Liquid hydrogen chloride would be equally poor solvent since strong Cl-...H-Cl hydrogen bonding would stabilize the chloride anion and preclude reaction. In contrast, nitromethane (CH3NO2) would be a polar enough solvent to solvate the GeCl4, but it will be displaced by the chloride anion, which would be only weakly solvated.
Salt formation
The formation of a salt via a double displacement reaction, (1.8.10), can be promoted by the choice of solvent by shifting the equilibrium by stabilization of one or more reagent/product.
\[MX + M'X' \rightleftharpoons MX' + M'X \]
Salt stabilization through relative acidity
The attempted formation of nitronium perchlorate from nitric acid and perchloric acid, (1.8.11), in water will result in the decomposition of the NO2+ cation, (1.24). However, if the reaction is carried out in a stronger acid, i.e., sulfuric acid, the NO2+ cation is stable, and the resulting salt can be recystallized, (1.8.12).
\[HNO_3 + HClO_4 \rightarrow NO_2^+ + ClO_4^- + H_2O \]
\[NO_2^+ + 2 H_2O \rightarrow HNO_3 + H_3O^+ \]
In a similar manner, the weak basic character of water means the equilibrium reaction, (1.25), has a very small equilibrium constant, K. However, if the reaction is carried out in a strongly basic solvent such as ammonia the uride anion is stabilized, (1.26), and can be precipitated by cation exchange.
Salt stabilization through solvation
The following observations may be explained by a consideration of the solvation ability of the solvent.
- The reaction of silver nitrate with barium chloride in water yields silver chloride and barium nitrate, (1.8.13).
- The reaction of barium nitrate with silver chloride in ammonia yields barium chloride and silver nitrate, (1.8.14).
\[ 2 AgNO_3 + BaCl_2 \xrightarrow{H_2O} 2 AgCl\downarrow + Ba^{2+} + 2 NO_3^-\]
\[ 2 AgCl + Ba(NO_3)_2 \xrightarrow{NH_2} BaCl_2\downarrow + 2 Ag^+ + 2 NO_3^-\]
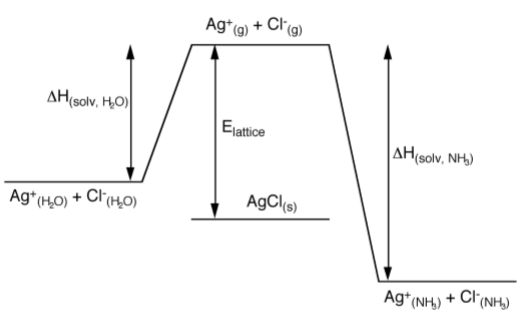
Since silver nitrate and barium nitrate are soluble in both solvents, the differences must be due to differences in the solubility of the chlorides in each solvent. A consideration of the relative stability of solid silver chloride versus the solvated species (Figure \(\PageIndex{1}\).61) shows that the enthalpy of solvation in water is less than the lattice energy. Thus, if silver chloride were present as Ag+ and Cl- in water it would spontaneously precipitate. In contrast, the enthalpy of solvation in ammonia is greater than the lattice energy, thus solid AgCl will dissolve readily in liquid ammonia. The reason for the extra stabilization from the specific solvation of the silver cation by the ammonia, i.e., the formation of the covalent complex [Ag(NH3)2]+.
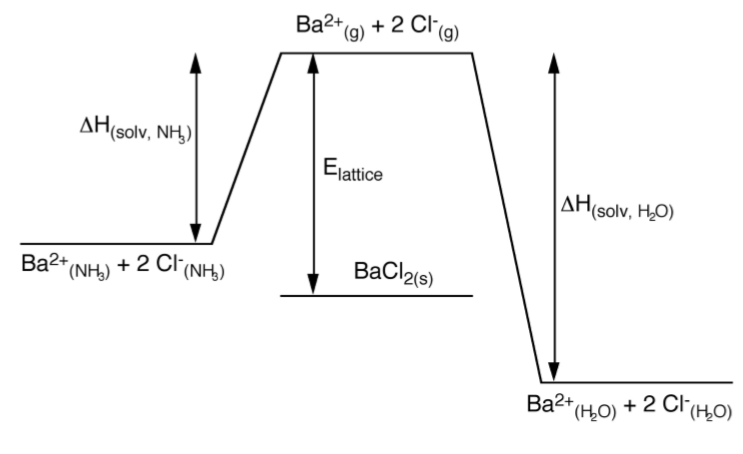
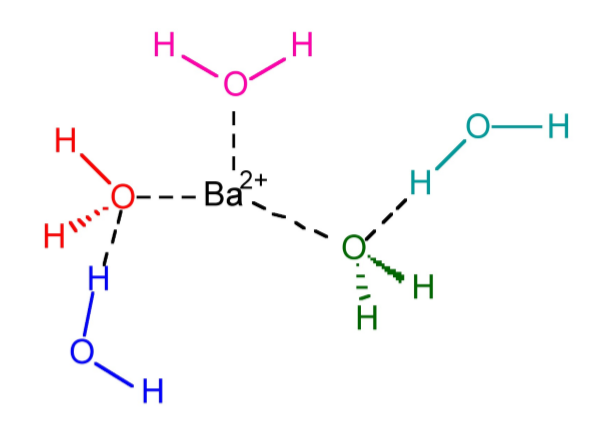
As may be seen from Figure \(\PageIndex{1}\).62, the opposite effect occurs for barium chloride. Here the enthalpy of solvation in ammonia is less than the lattice energy. Thus, if barium chloride were present as Ba2+ and Cl- in ammonia it would spontaneously precipitate. In contrast, the enthalpy of solvation in water is greater than the lattice energy, thus solid BaCl2 will dissolve readily in water. The stabilization of Ba2+(aq) occurs because water will have a larger sphere of non-specific solvation as a consequence of having two lone pairs, allowing interaction with the Ba2+ as well as other water molecules (Figure \(\PageIndex{1}\).63).
Electron transfer reaction
A consideration of the oxidation, (1.8.15), and reduction, (1.8.16), reactions that occur for pure water at neutral pH (where [H+] = 10-7) would suggest that water will not tolerate oxidants whose E0 is greater than 0.82 V nor tolerate reductants whose E0 is less than -0.41 V.
\[2 H^+ + \dfrac{1}{2} O_2 + 2 e^- \rightarrow H_2O \space\space\space\space\space\space E_0=+0.82V\]
\[H^+ + e^- \rightarrow \dfrac{1}{2} H_2 \space\space\space\space\space\space E_0 = -0.41V\]
Thus, while water has a fair range to support redox reactions it is not very good at the extremes with strong reducing agents or strong oxidizing agents. Liquid ammonia is an excellent solvent for very strong reducing agents because of the stabilization of solvated electrons, i.e., [e-(NH3)6]. In contrast, hydrochloric acid is a good solvent for reactions involving very strong oxidizing agents.


