9.3: Multiple Bonds
- Page ID
- 360635
- Describe multiple covalent bonding in terms of atomic orbital overlap
- Relate the concept of resonance to π-bonding and electron delocalization
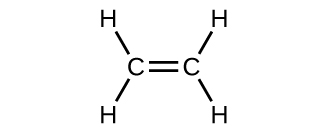
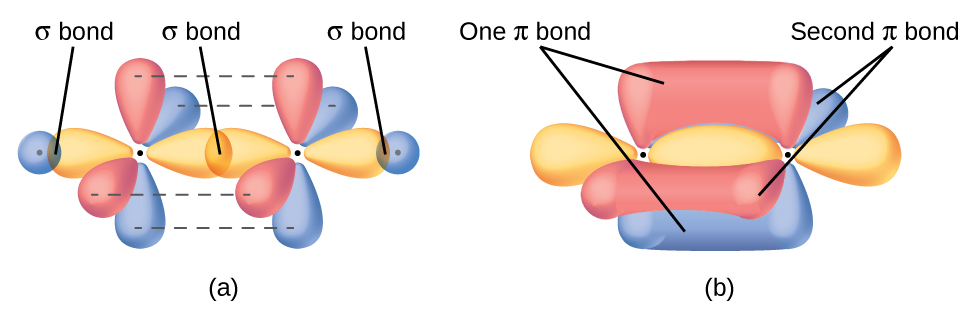
The hybrid orbital model appears to account well for the geometry of molecules involving single covalent bonds. Is it also capable of describing molecules containing double and triple bonds? We have already discussed that multiple bonds consist of σ and π bonds. Next we can consider how we visualize these components and how they relate to hybrid orbitals. The Lewis structure of ethene, C2H4, shows us that each carbon atom is surrounded by one other carbon atom and two hydrogen atoms. As a result each carbon atom has trigonal planar geometry - according to VSEPR - and is sp2 hybridized. Figure \(\PageIndex{1}\) shows the atomic orbitals and the hybrid orbitals. The sp2 hybrid orbitals overlap to make σ bonds to the hydrogen atoms and between the carbon atoms.
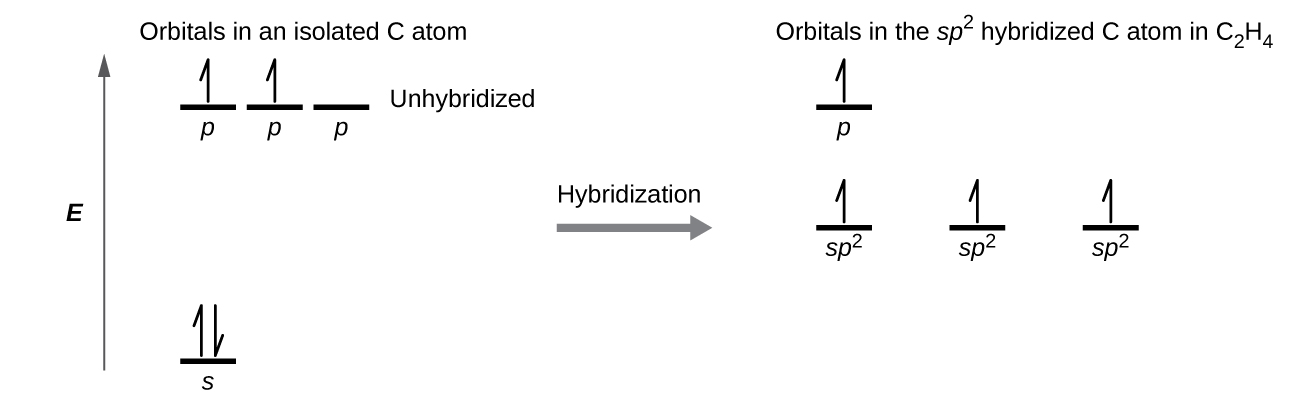
The π bond in the C=C double bond results from the overlap of the third (unused) 2p orbital on each carbon atom that is not involved in hybridization. This unhybridized p orbital (lobes shown in red and blue in Figure \(\PageIndex{2}\)) is perpendicular to the plane of the sp2 hybrid orbitals. Thus the unhybridized 2p orbitals overlap in a side-by-side fashion, above and below the internuclear axis and form a π bond.
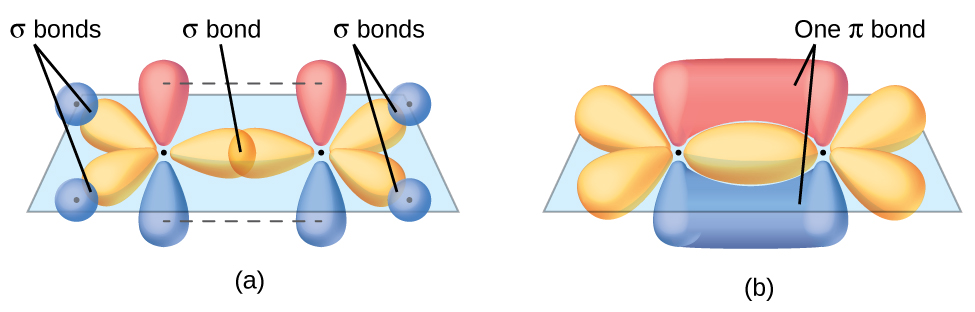
In an ethene molecule, the four hydrogen atoms and the two carbon atoms are all in the same plane. If the two planes of sp2 hybrid orbitals tilted relative to each other, the p orbitals would not be oriented to overlap efficiently to create the π bond. The planar configuration for the ethene molecule occurs because it is the most stable bonding arrangement. This is a significant difference between σ and π bonds; rotation around single (σ) bonds occurs easily because the end-to-end orbital overlap does not depend on the relative orientation of the orbitals on each atom in the bond. In other words, rotation around the internuclear axis does not change the extent to which the σ bonding orbitals overlap because the bonding electron density is symmetric about the axis. Rotation about the internuclear axis is much more difficult for multiple bonds because it would change the overlap of the π bonding orbitals and require breaking the π bond.
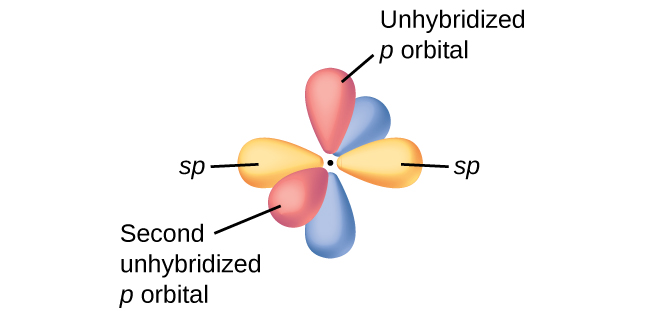
In molecules with sp hybrid orbitals there are two unhybridized p orbitals, (Figure \(\PageIndex{3}\)). Acetylene, H−C≡C−H, is a linear molecule with this arragement. The sp hybrid orbitals of the two carbon atoms overlap end to end to form a σ bond between the carbon atoms (Figure \(\PageIndex{4}\)). The remaining sp orbitals form σ bonds with hydrogen atoms. This leaves two unhybridized p orbitals on each carbon atom that can overlap toform two π bonds. The two carbon atoms of acetylene are bound by one σ bond and two π bonds, giving a triple bond.
Hybridization involves only σ bonds, lone pairs of electrons, and single unpaired electrons (radicals). Structures that account for these features describe the correct hybridization of the atoms. However, many structures also include resonance forms. Remember that resonance forms occur when various arrangements of π bonds are possible. Since the arrangement of π bonds involves only the unhybridized orbitals, resonance does not influence the assignment of hybridization.
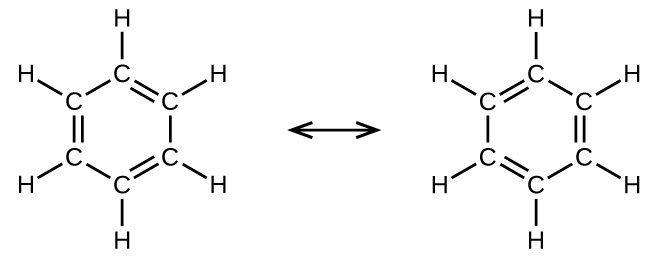
For example, benzene has two resonance forms (Figure \(\PageIndex{5}\)). Either of these forms show each carbon atom is bonded to three other atoms with no lone pairs, so the hybridization is sp2. The electrons in the unhybridized p orbitals form π bonds but there are two possible configurations. Neither resonance structure completely describes the electrons in the π bonds. They are not located in one position or the other, but are in both configurations at the same time.
One component of acid rain is produced by the reaction of sulfur dioxide with atmospheric water vapor to form sulfuric acid. Sulfur dioxide, \(\ce{SO2}\), is a major component of volcanic gases and a product of the combustion of sulfur-containing coal. What is the hybridization of the \(S\) atom in \(\ce{SO2}\)?
Solution
The resonance structures of \(\ce{SO2}\) are

The sulfur atom is surrounded by two bonds and one lone pair of electrons in either resonance structure. Therefore, the electron-pair geometry is trigonal planar, and the hybridization of the sulfur atom is sp2.
Another acid in acid rain is nitric acid, HNO3, which is produced by the reaction of nitrogen dioxide, NO2, with atmospheric water vapor. What is the hybridization of the nitrogen atom in NO2? (Note: the lone electron on nitrogen occupies a hybridized orbital just as a lone pair would.)
- Answer
-
sp2
Summary
Multiple bonds consist of a σ bond located along the axis between two atoms and one or two π bonds. The σ bonds are usually formed by the overlap of hybridized atomic orbitals, while the π bonds are formed by the side-by-side overlap of unhybridized orbitals. Resonance occurs when there are multiple unhybridized orbitals with the appropriate alignment to overlap, so the placement of π bonds can vary.


