Concepts & Vocabulary
20.0 Chapter Objectives and Introduction to Carboxylic Acids
- Carboxylic acids are defined as molecules that have a hydroxyl group bonded to a carbonyl group (RCOOH).
- Fatty acids are naturally occurring carboxylic acids that are important components of lipids.
- Fatty acids can either be saturated (all carbon-carbon bonds are single bonds) and unsaturated (one or more carbon-carbon double bonds).
- Several other functional groups can be prepared from carboxylic acids and are considered derivatives (acyl halides, anhydrides, esters, amides and nitriles).
20.1 Naming Carboxylic Acids and Nitriles
- Carboxylic acids are named following IUPAC rules with ending -oic acid.
- When carboxylic acids are named as an attachment to a ring, add -carboxylic acid to the name of the cyclic compound.
- Nitriles are named following IUPAC rules with the ending -nitrile or -carbonitrile.
20.2 Structure and Properties of Carboxylic Acids
- Carboxylic acids have trigonal planar shape around the carbonyl carbon and due to resonance structures, all the atoms of the functional group are co-planar.
- Conjugation in carboxylic acids increases acidity of the hydroxyl hydrogen compared to alcohols. The conjugate base (carboxylate) is stabilized by resonance.
- Carboxylic acids form hydrogen bonded dimers yielding higher boiling points than for other molecules of similar size.
- Small carboxylic acids are often soluble in water due to hydrogen bonding. Larger carboxylic acids are often slightly soluble in water, though when converted to a carboxylate salt by treating with base, become water soluble.
20.3 Biological Acids and the Henderson-Hasselbalch Equation
- Carboxylic acids are fully protonated in acidic solutions.
- Carboxylic acids are fully dissociated in basic solutions.
- The Henderson-Hasselbalch equation can be used to relate pH, pKa and ratio of concentrations of acid and conjugate base.
20.4 Substituent Effects on Acidity
- Withdrawing groups (through inductive or resonance effects) can stabilize carboxylates, increasing acidity of carboxylic acids.
- Donating groups (through inductive or resonance effects) destabilize carboxylates, decreasing acidity of carboxylic acids.
- Nearly all substituents in ortho positions increase acid strength whether donating or withdrawing, this is called the ortho-effect.
20.5 Preparing Carboxylic Acids
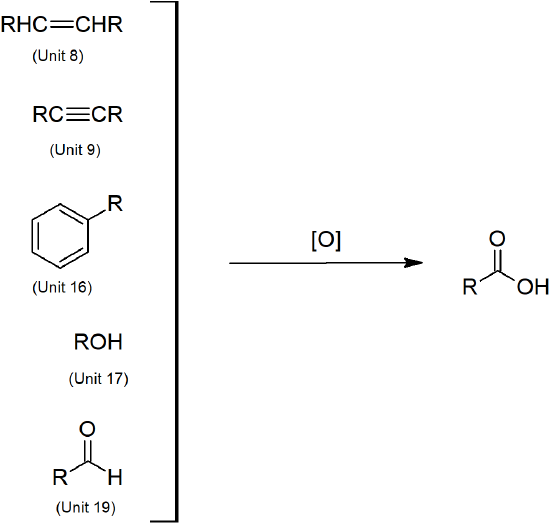
- Carboxylic acids can be prepared through oxidation of several functional groups including: 1o alcohols, aldehydes, alkyl arene side chains, and alkenes.
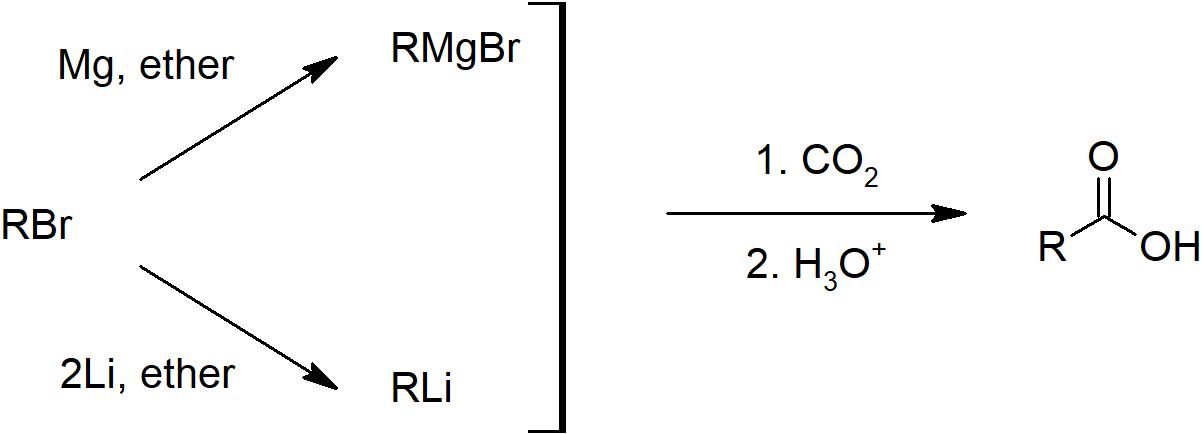
- Carboxylic acids can also be prepared by carboxylation of Grignard reagents and hydrolysis of nitriles.
20.6 Reactions of Carboxylic Acids: An Overview
- There are four main categories of carboxylic acid reactions:
- Acid/base reaction (salt formation)
- Nucleophilic acyl substitution
- Reduction
- Substitution alpha to the carbonyl
20.7 Chemistry of Nitriles
- Nitriles have linear geometry due to the sp hybrid orbitals on carbon and nitrogen.
- Nitriles are less basic than amines.
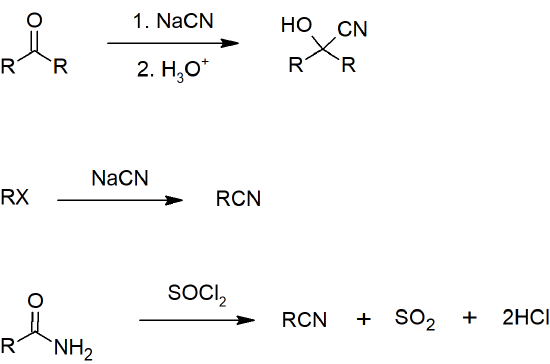
- Nitriles can be prepared from aldehydes, ketones, or amides.
- Nitriles react similarly to carbonyl compounds where nucleophiles will attack the carbon end of the nitrile.
- Nitriles can be hydrolyzed to carboxylic acids under basic or acidic conditions.
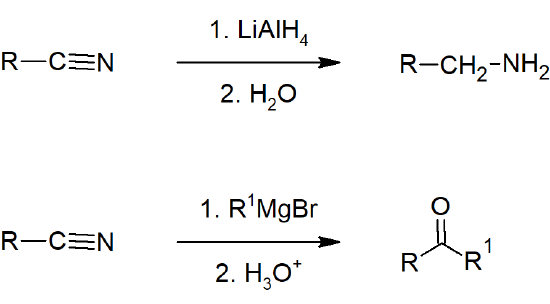
- Nitriles can be reduced to amines by hydride reduction.
- Nitriles can be converted to aldehyde by reaction with DIBALH.
- Nitriles can be converted to ketones by reaction with organometallic compounds.
20.8 Spectroscopy of Carboxylic Acids and Nitriles
- IR of carboxylic acids typically show a very strong and broad OH stretch from about 2500 to 3300 cm-1 as well as a strong carbonyl stretch around 1710 cm-1.
- 1H NMR of carboxylic acids show the OH proton between 10-12 ppm as well as hydrogens on carbon adjacent to the carbonyl around 2-3 ppm.
- 13C NMR of carboxylic acids show the carbonyl carbon at 160-180 ppm.
- Mass spectra of carboxylic acids often have fragments for OH (loss of 17) and COOH (loss of 45).
- The most identifying characteristic of IR of nitriles is the CN triple bond stretch which appears near 2250 cm-1.
- 1H NMR of nitriles show protons on the carbon next to the nitrile at around 2-3 ppm.
- 13C NMR of nitriles show the nitrile carbon in the 115-120 ppm range.
- Mass spectra of nitriles will often not have a visible molecular ion, but instead exhibit a strong M-1 peak.