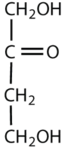6.1: Overview of Carbohydrates
- Page ID
- 341504
Learning Objectives
- To recognize carbohydrates and classify them as mono-, di-, oligo- or polysaccharides.
Carbohydrates are biological molecules composed of carbon, hydrogen and oxygen atoms. The term carbohydrate is generally applied to the group of polyhydroxy aldehydes or polyhydroxy ketones commonly known as sugars.

Figure \(\PageIndex{1}\): Structures of a polyhydroxy aldehyde (left) and polyhydroxy ketone (right).
Examples of carbohydrates include starch, fiber, the sweet-tasting compounds called sugars, and structural materials such as cellulose. The term carbohydrate had its origin in a misinterpretation of the molecular formulas of many of these substances. For example, because its formula is C6H12O6, glucose was once thought to be a “carbon hydrate” with the structure Cn·(H2O)n or C6·6H2O molecular formula.
Example \(\PageIndex{1}\)
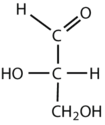
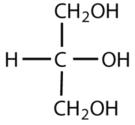
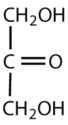
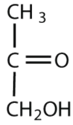
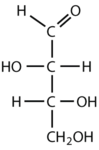
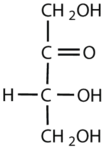
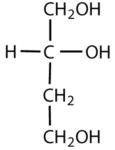
Which compounds would be classified as carbohydrates?
Solution
-
This is a carbohydrate because the molecule contains an aldehyde functional group with OH groups on the other two carbon atoms.
-
This is not a carbohydrate because the molecule does not contain an aldehyde or a ketone functional group.
-
This is a carbohydrate because the molecule contains a ketone functional group with OH groups on the other two carbon atoms.
-
This is not a carbohydrate; although it has a ketone functional group, one of the other carbons atoms does not have an OH group attached.
Exercise \(\PageIndex{1}\)
Which compounds would be classified as carbohydrates?
Green plants are capable of synthesizing glucose (C6H12O6) from carbon dioxide (CO2) and water (H2O) by using solar energy in the process known as photosynthesis:
\[\ce{6CO_2 + 6H_2O} + \text{686 kcal} \rightarrow \ce{C_6H_{12}O_6 + 6O_2} \label{\(\PageIndex{1}\)}\]
(The 686 kcal come from solar energy.) Plants can use the glucose for energy or convert it to larger carbohydrates, such as starch or cellulose. Starch provides energy for later use, perhaps as nourishment for a plant’s seeds, while cellulose is the structural material of plants. We can gather and eat the parts of a plant that store energy—seeds, roots, tubers, and fruits—and use some of that energy ourselves. Carbohydrates are also needed for the synthesis of nucleic acids and many proteins and lipids.
Animals, including humans, cannot synthesize carbohydrates from carbon dioxide and water and are therefore dependent on the plant kingdom to provide these vital compounds. We use carbohydrates not only for food (about 60%–65% by mass of the average diet) but also for clothing (cotton, linen, rayon), shelter (wood), fuel (wood), and paper (wood).
Carbohydrates are classified based on the number of sugar units combined to make the molecule. The simplest carbohydrates—those that cannot be hydrolyzed to produce even smaller carbohydrates—are called monosaccharides. Two or more monosaccharides can link together to form chains that contain from two to several hundred or thousand monosaccharide units. Prefixes are used to indicate the number of such units in the chains. Disaccharides (meaning two sugars) consist of two monosaccharides joined together; oligosaccharides (meaning a few sugars) contain anywhere from three to nine monosaccharides; and polysaccharides (meaning many sugars) are biological polymers containing 10 or more monosaccharide units. All these so-called higher saccharides can be hydrolyzed back to their constituent monosaccharides.
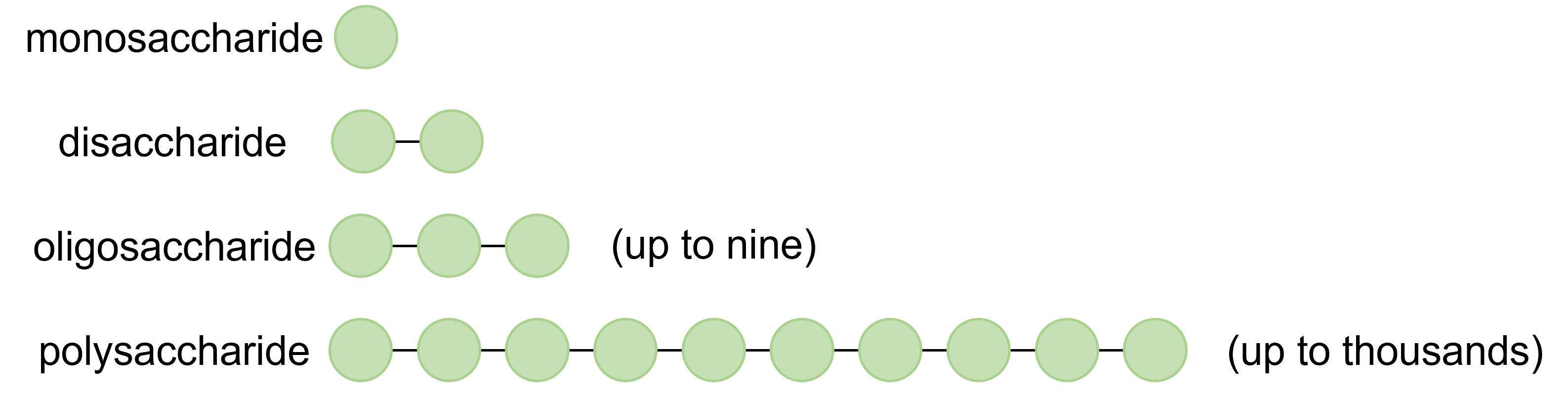
Figure \(\PageIndex{2}\): Types of carbohydrates categorized based on the number of sugar units present.
Compounds that cannot be hydrolyzed will not react with water to form two or more smaller compounds.
Example \(\PageIndex{1}\)
When an aqueous solution of trehalose is hydrolyzed, two molecules of glucose are produced for each molecule of trehalose. Is trehalose a monosaccharide, a disaccharide, an oligosaccharide, or a polysaccharide?
Solution
Trehalose is a disaccharide because it is hydrolyzed into two molecules of glucose (a monosaccharide)
Exercise \(\PageIndex{1}\)
When an aqueous solution of arabinose is hydrolyzed, no other molecules are produced. Is arabinose a monosaccharide, a disaccharide, an oligosaccharide, or a polysaccharide
Summary
- Carbohydrates are an important group of biological molecules that includes sugars and starches.
- Photosynthesis is the process by which plants use energy from sunlight to synthesize carbohydrates.
- Carbohydrates are classified based on the number of sugar units. A monosaccharide is the simplest carbohydrate and cannot be hydrolyzed to produce a smaller carbohydrate molecule. Disaccharides contain two monosaccharide units, oligosaccharides contain between three and nine monosaccharide units, and polysaccharides contain ten or more monosaccharide units.