6.2: Stereoisomers
- Page ID
- 341589
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Learning Outcomes
- Determine the isomeric relationship between a pair of molecules.
- Identify the chiral centers in a molecule.
- Describe different types of isomers.
One of the interesting aspects of organic chemistry is that it is three-dimensional. A molecule can have a shape in space that may contribute to its properties. Molecules can differ in the way the atoms are arranged - the same combination of atoms can be assembled in more than one way. These compounds are known as isomers. Isomers are molecules with the same molecular formulas, but different arrangements of atoms. There are several different types of isomers (some previously discussed and some being introduced):

Structural Isomers
Structural isomers are molecules that have the same molecular formula, but the atoms are connected/bonded differently. As a result, these compounds will have different IUPAC names.

Conformational Isomers
Conformational isomers are molecules with the same molecular formula and the same connectivity/bonding between the atoms. However, due to the rotation of one or more single bonds, the molecules look as if they are bonded differently. Conformational isomers will have the same IUPAC name.

Stereoisomers
Stereoisomers also have the same molecular formula and same connectivity/bonding between the atoms. However, these isomers have different three-dimensional arrangements. Stereoisomers can be further categorized based on how the 3-D arrangements differ.
Geometric Isomers
Geometric isomers, also called cis-trans isomers, have different configurations due to the restricted rotation of double bonds or cyclic molecules. The cis isomer occurs when the groups are on the same side of the molecule. The trans isomer occurs when the groups are on the opposite side of the molecule. Geometric isomers will have the same IUPAC name, except for the cis/trans designation at the beginning of the name.
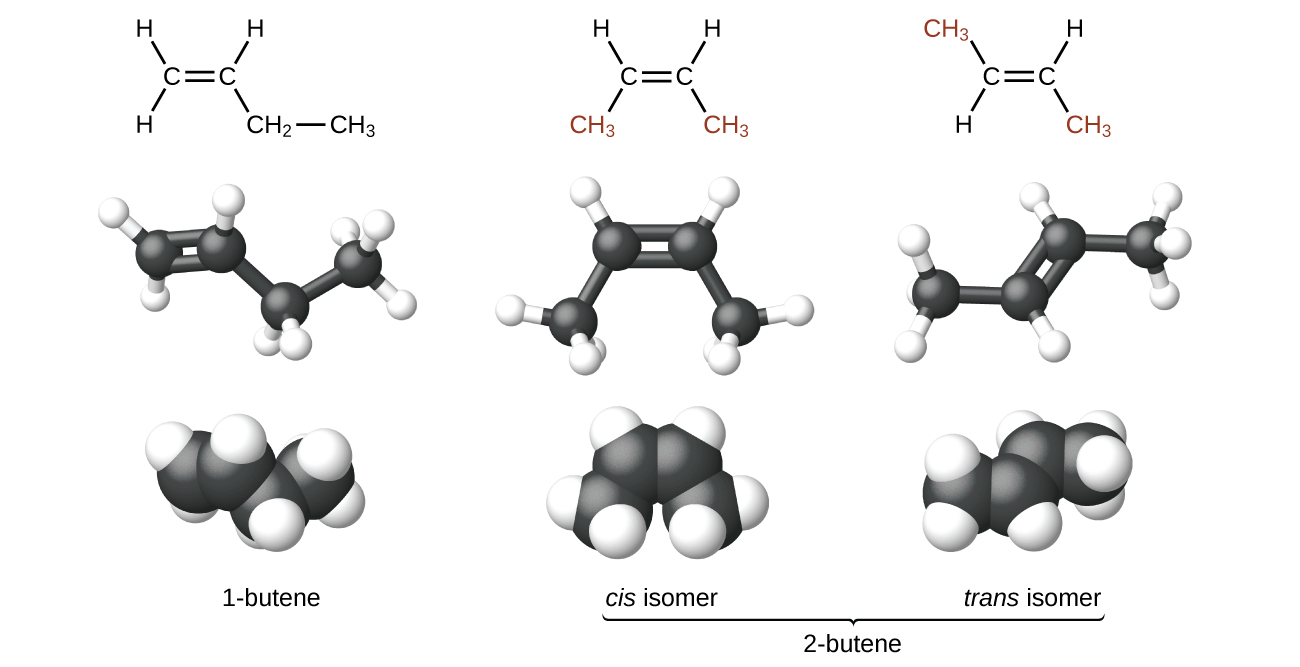
Figure \(\PageIndex{4}\): Structural and Geometric isomers of butene. OpenStax, CC BY 4.0 <https://creativecommons.org/licenses/by/4.0>, via Wikimedia Commons
Optical Isomers
Stereoisomers that are not geometric isomers are known as optical isomers. Optical isomers, which were given their name because of their interactions with plane-polarized light, differ in the placement of substituted groups around one or more atoms of the molecule. These types of isomers are labeled as enantiomers or diastereomers.
Enantiomers are non-superimposable (no symmetry) mirror images. Your hands are a common example of a pair of enantiomers. Your left hand and right hand are mirror images of one another, but no matter how you turn, twist, or rotate them, they are not superimposable.
Objects or molecules that have non-superimposable mirror images are called chiral. Chiral molecules contain a chiral (or chirality) center. A chiral center is a tetrahedral carbon (carbon with four single bonds) bonded to four different atoms or group of atoms. Note that we have to look beyond the first atom attached to the central carbon atom. The four shaded areas shown below (Figure \(\PageIndex{6}\)) indicate the four unique groups attached to the central carbon atom, which is a chiral center.

If you look back at the molecule shown above in Figure \(\PageIndex{5}\), three distinct groups are present. If the R group is anything other than the three groups shown, the molecule is chiral. However, if the R group is the same as one of the groups shown (H, NH2, or COOH), the molecule will be considered achiral. Achiral objects or molecules have symmetry because they have superimposable (identical) mirror images.

Another type of optical isomers are diastereomers. Diastereomers are non-mirror image isomers that have a different arrangement around one or more chiral centers while some of the atoms have the same arrangement. As shown in the figure below, note that the orientation of groups on the first and third carbons are different but those on the second one remains the same. Therefore, these do not represent the same molecule. The solid wedge indicates a group coming out of the page/screen towards you and the dashed line indicates that a group is going away from you "behind" the page/screen.

Figure \(\PageIndex{8}\): Diastereomers differ at one or more atom. These molecules are not mirror images and they are not superimposable. Rhannosh, CC BY-SA 3.0 <https://creativecommons.org/licenses/by-sa/3.0>, via Wikimedia Commons
Epimers are a sub-group of diastereomers that differ at only one chiral center. All epimers are diastereomers but not all diastereomers are epimers. As shown in the figure below, the orientation of the groups on the first and second carbons are the same and only the third carbon changes.

Example \(\PageIndex{1}\)
Indicate whether the following molecule has a chiral center. If so, identify the carbon.
Solution
- This molecule has one chiral center, carbon 3 of the parent chain. When identifying chiral centers, eliminate the carbon atoms that cannot be chiral centers. This includes CH2 groups, CH3 groups, and any carbon that is part of a double or triple bond. Then check to see if each of the remaining carbon atoms have four different groups attached. Carbon 2 has two CH3 groups attached, so it is not chiral.
- This molecule does not have a chiral center. Carbon 1 is not tetrahedral since it has a double bond to the oxygen. Carbons 2, 3, 4, and 6 have multiple hydrogen atoms. Carbon 5 has two CH3 groups.
Example \(\PageIndex{1}\)
Indicate whether the following molecule has a chiral center. If so, identify the carbon.
Key Takeaway
- Stereoisomers have the same connectivity, but a different 3D structure.
- Enantiomers are non-superimposable mirror images (referred to as chiral molecules).
- Chiral centers, which are present in chiral molecules, are carbon atoms that have four different groups of atoms attached to it.
- Diastereomers are non-mirror image stereoisomers.
- Epimers are stereoisomers that differ around only one chiral center.
Contributors and Attributions
Allison Soult, Ph.D. (Department of Chemistry, University of Kentucky)





