Regioselectivity
If more than one reaction could occur between a set of reactants under the same conditions giving products that are constitutional isomers and if one product forms in greater amounts than the others, the overall reaction is said to be regioselective.
Say three reactions could occur between the hypothetical reactants A and B under the same conditions giving the constitutionally isomeric products C, D, and E.

There are two possibilities:
1. The three products form in equal amounts, i.e., of the total product 33% is C, another 33% D, the remaining 33% E. (These percentages are called relative yields of the products.)
%252C_D_(33%2525)%252C_and_E_(33%2525).svg?revision=1)
If this is what is observed, the overall reaction between A and B is not regioselective.
2. One product forms in greater amounts than the others. Say, for example, the relative yields of C, D, and E are 25%, 50%, and 25%, respectively.
%252C_D_(50%2525)%252C_and_E_(25%2525).svg?revision=1)
If this is what is observed, the overall reaction between A and B is regioselective.
eg:
_with_HBr_to_give_1-bromo-1-methylcylohexane_(2)_and_1-bromo-2-methylcylcohexane_(3).svg?revision=1)
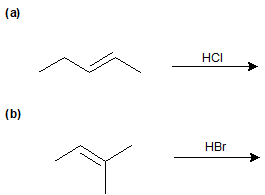
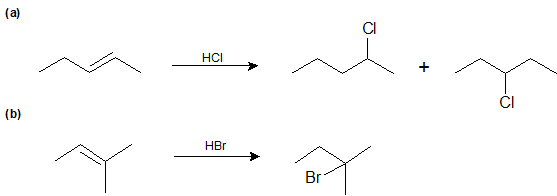
Experimentally, 2 is the major product; 3 is the minor product. Thus, the overall reaction between 1 and HBr is regioselective toward 2.
If more than one reaction could occur between a set of reactants under the same conditions giving products that are constitutional isomers and if only one product is observed, the overall reaction is said to be 100% regioselective or regiospecific.
eg:
_with_HBr_to_give_2-bromopropane_(5)_and_1-bromopropane_(6).svg?revision=1)
The only observed product is 5. (Relative yields of 5 and 6 are 100% and 0%, respectively.) Thus the overall reaction between 4 and HBr is regiospecific toward 5.
Regiospecificity is merely the limiting case of regioselectivity. All regiospecific reactions are regioselective, but not all regioselective reactions are regiospecific.
Addition to unsymmetrical alkenes
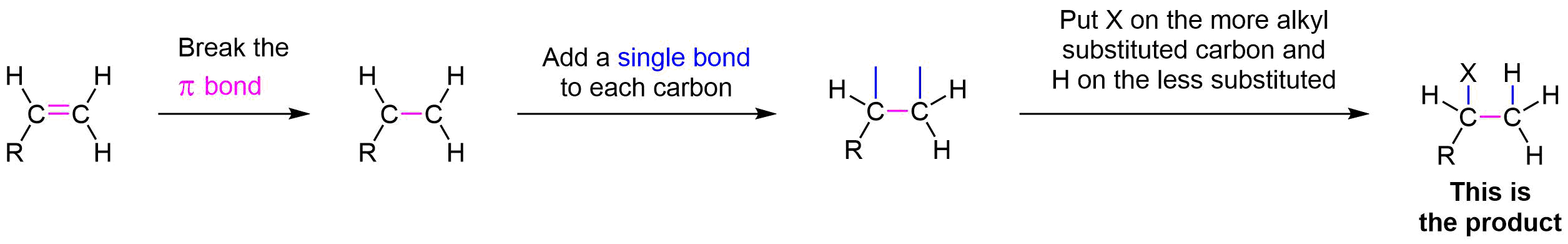
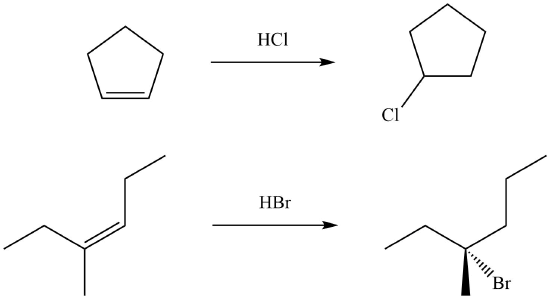
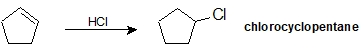
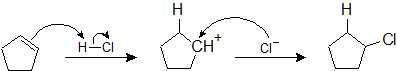
During the electrophilic addition of HX to an alkene, the halide (X) could attach to either carbon in the double bond producing two different isomers as products. But, when an unsymmertrically alkyl substituted alkene undergoes an electrophillic addition with HX a single isomer is typically produced. For example, if propene were reacted with HBr, two products could possibly form: 2-bromolpropane and 1-bromopropane. However, 2-bromopropane is produced as the reaction's only product. Reactions are called regiospecific when only one of multiple possible isomers is exclusively formed.

Example \(\PageIndex{1}\)
If HCl adds to an unsymmetrical alkene like propene what will the major product be?
Solution

The regiospecificity of electrophilic additions to alkenes is commonly known as Markovnikov's rule, after the Russian chemist Vladimir Markovnikov who proposed it in 1869. The electrophilic addition of HX to an alkene is said to follow Markovnikov's rule.
Markovnikov's rule: During the electrophilic addition of HX to an alkene, the H adds to the carbon of the double bond with the fewest number of alkyl substitutent. The halide (X) adds to the double bond carbon with the most alkyl substituents. Although Markovnikov's rule has been specifically stated for the electrophilic addition of HX, later in this chapter many more reaction will be shown to also follow Markovnikov's rule.


It is important to point out that Markovnikov's rule only truly applies when there is a difference between the number of alky groups attached to each carbon in the double bond. When both carbon of the double bond have the same degree of alkyl substitution, Markovnikovs's rule becomes void and a mixture of both possible isomers is produced.

To consider an explanation for why Markovnikov's rule holds true, the mechanism of the reaction needs to be considered. As seen in the previous section, the mechanism starts with the addition of H to a carbon in the double bond. This in turn caused the other double bond carbon to become a carbocation intermediate. In the second step of the mechanism the halide ion attacks the carbocation to form a C-X. Because Markovnikov's rule says that the halogen adds to the carbon in the double bond with the most alky substituents, it can be said the carbocation also prefers to form on the carbon with the most alkyl substituents. The reason for this holds the explaination for Markovnikov's rule. To simplify this idea, Markovnikov's rule can be restated in a different form.
Markovnikov's rule: During the electrophilic addition of HX to an alkene, the carbocation intermediate forms on the double bond carbon with the greatest number of alkyl substitutents.