27.1: Waxes, Fats, and Oils
- Page ID
- 36482
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- identify waxes as being mixtures of long‑chain esters, and write the general structure for such compounds.
- identify fats and oils as being triacylglycerols, and write a general structure for such compounds.
- relate the physical properties of animal fats and vegetable oils to their structures.
- predict the behaviour of a given fat or oil when it is subjected to some of the more common reactions discussed in previous units; examples would include hydrolysis, reduction and ozonolysis.
Make certain that you can define, and use in context, the key terms below.
- fat
- fatty acids
- triacylglycerols
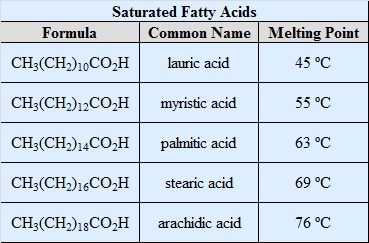
You are not expected to memorize the trivial names or formulas of the fatty acids listed in Table \(\PageIndex{1}\). The systematic names for these compounds are shown in the tables below.
Saturated Fatty Acids
| Trivial Name | Systematic Name |
|---|---|
| lauric acid | dodecanoic acid |
| myristic acid | tetradecanoic acid |
| palmitic acid | hexadecanoic acid |
| stearic acid | octadecanoic acid |
| arachidic acid | eicosanoic acid |
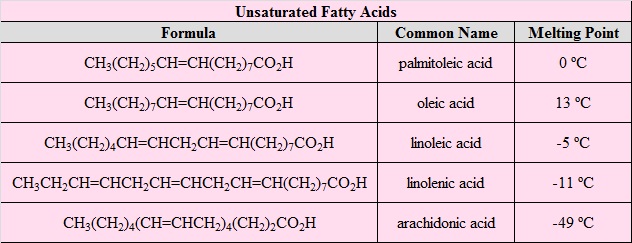
Unsaturated Fatty Acids
| Trivial Name | Systematic Name |
|---|---|
| palmitoleic acid | (Z)‑9‑hexadecenoic acid |
| oleic acid | (Z)‑9‑octadecenoic acid |
| ricinoleic acid | (Z)‑12‑hydroxy‑9‑octadecenoic acid |
| linoleic acid | (Z,Z)‑9,12‑octadecadienoic acid |
| linolenic acid | (Z,Z,Z)‑9,12,15‑octadecatrienoic acid |
| arachidonic acid | (Z,Z,Z,Z)‑5,8,11,14‑eicosatetraenoic acid |
The systematic names in these tables may be somewhat unfamiliar, but they are derived in exactly the same way as the names of the simpler carboxylic acids that we discussed in Chapter 20. You may wish to review the alkane names (Section 3.2) and the E,Z system (Section 7.5) to satisfy yourself that you understand how these names originate.
In many older textbooks, the term “fatty acid” is used to describe all carboxylic acids, not only those that are obtained from the hydrolysis of triacylglycerols.
Fats play an important role in human nutrition, and most people are aware of the desirability of limiting their dietary intake of saturated fats, as these compounds have been associated with heart disease. Unsaturated fats are generally considered to be much more desirable from the point of view of good health. Notice that all the fatty acids derived from naturally occurring fats have a Z (i.e., cis) configuration.
Linoleic acid is an “essential” nutrient; that is, it cannot be made by the body in sufficient quantity to meet our physiological needs, and must be obtained from food. A deficiency in linoleic acid results in skin problems and liver abnormalities. Historically, linolenic and arachidonic acids were also thought to be essential nutrients, but recent research suggests that they can be synthesized in the body if sufficient linoleic acid is present.
Fatty Acids
Fatty acids are structural components of fats, oils, and all other categories of lipids, except steroids. Fatty acids are long-chain carboxylic acids that have the general formula CH3(CH2)nCOOH, where n usually ranges from 2 to 28 and is always an even number. More than 70 have been identified in nature. They are generally unbranched, and can be classified by the presence and number of carbon-to-carbon double bonds.
A saturated fat is a fat that consists of triglycerides whose carbon chains consist entirely of carbon-carbon single bonds. Therefore, the carbon chains are saturated with the maximum number of hydrogen atoms possible. An unsaturated fat is a fat that consists of triglycerides whose carbon chains contain one or more carbon-carbon double bonds. A fat with one double bond is called monounsaturated, while a fat with multiple double bonds is called polyunsaturated (see figure below).
Each of the double bonds in unsaturated fatty acids can have either cis or trans stereochemistry and the type of fatty acid is further defined as a cis fatty acid or trans fatty acid. These are what are commonly referred to in food as cis or trans fats.

 26.8
26.8
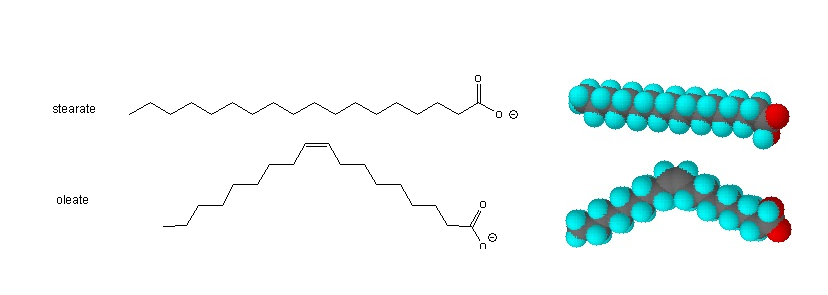
The higher melting points of the saturated fatty acids reflect the uniform rod-like shape of their molecules. Viewed as a whole, however, the saturated fatty acid molecule is relatively straight (Figure 17.1.2b). Such molecules pack closely together into a crystal lattice, maximizing the strength of dispersion forces and causing fatty acids and the fats derived from them to have relatively high melting points.
The cis-double bond(s) in the unsaturated fatty acids introduce a kink in their shape, which makes it more difficult to pack their molecules together in a stable repeating array or crystalline lattice. As a result, the intermolecular attractions of unsaturated fatty acids (and unsaturated fats) are weaker, causing these substances to have lower melting points. Most are liquids at room temperature.
Waxes
Waxes are esters of long-chain fatty acids with long-chain monohydric alcohols (one hydroxyl group). The carboxylic acid and the alcohol typically each have an even number of carbons. Cetyl palmitate is a typical wax, it is the ester of cetyl alcohol ( CH3(CH2)15OH) and palmitic acid (CH3(CH2)14COOH). Most waxes have a similar structure.
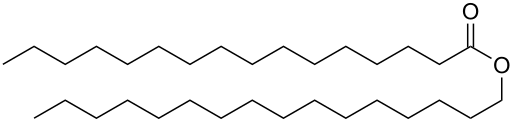
Cetyl palmitate, a typical wax ester.
Waxes are widely distributed in nature. The leaves and fruits of many plants have waxy coatings, which may protect them from dehydration and small predators. The feathers of birds and the fur of some animals have similar coatings which serve as a water repellent. Carnauba wax is valued for its toughness and water resistance. The formulas for three well known waxes are given below, with the carboxylic acid moiety colored red and the alcohol colored blue.
| Spermaceti | Beeswax | Carnauba wax | |
|---|---|---|---|
| Alcohol | CH3(CH2)14CH2-OH | CH3(CH2)28CH2-OH | CH3(CH2)28CH2-OH |
| Fatty Acid | CH3(CH2)14COOH | CH3(CH2)14COOH | CH3(CH2)24COOH |
| Wax | CH3(CH2)14CO2-(CH2)15CH3 | CH3(CH2)24CO2-(CH2)29CH3 | CH3(CH2)30CO2-(CH2)33CH3 |
Triglycerides
Fats and oils often exist as triglycerides (or triacylcylgerols because they are esters composed of three fatty acid units joined to glycerol, a trihydroxy alcohol (IUPAC name is 1,2,3-propanetriol)):
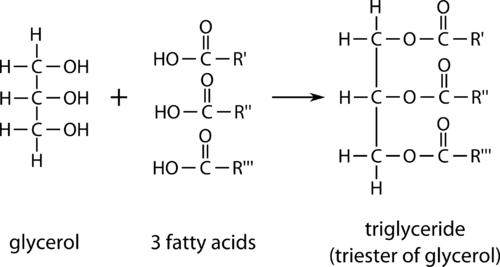
If all three OH groups on the glycerol molecule are esterified with the same fatty acid, the resulting ester is called a simple triglyceride. Although simple triglycerides have been synthesized in the laboratory, they rarely occur in nature. Instead, a typical triglyceride obtained from naturally occurring fats and oils contains two or three different fatty acid components and is thus termed a mixed triglyceride.
A triglyceride is called a fat if it is a solid at 25°C; it is called an oil if it is a liquid at that temperature. These differences in melting points reflect differences in the degree of unsaturation (as discussed above) and number of carbon atoms in the constituent fatty acids. Triglycerides obtained from animal sources are usually solids, while those of plant origin are generally oils. Therefore, we commonly speak of animal fats and vegetable oils. No single formula can be written to represent the naturally occurring fats and oils because they are highly complex mixtures of triglycerides in which many different fatty acids are represented. The table below shows the fatty acid compositions of some common fats and oils. The composition of any given fat or oil can vary depending on the plant or animal species it comes from as well as on dietetic and climatic factors.
| glycerides | |||||
|---|---|---|---|---|---|
| Fat or Oil | Saturated | Unsaturated | |||
| Palmitic | Stearic | Oleic | Linoleic | Other | |
| Animal Origin | |||||
| Butter | 29 | 9 | 27 | 4 | 31 |
| Lard | 30 | 18 | 41 | 6 | 5 |
| Beef | 32 | 25 | 38 | 3 | 2 |
| Vegetable Origin | |||||
| Corn oil | 10 | 4 | 34 | 48 | 4 |
| Soybean | 7 | 3 | 25 | 56 | 9 |
| Peanut | 7 | 5 | 60 | 21 | 7 |
| Olive | 6 | 4 | 83 | 7 | - |
Fats and oils can participate in a variety of chemical reactions—for example, because triglycerides are esters, they can be hydrolyzed in the presence of an acid, a base, or specific enzymes known as lipases. The hydrolysis of fats and oils in the presence of a base is used to make soap and is called saponification. Today most soaps are prepared through the hydrolysis of triglycerides (often from tallow, coconut oil, or both) using water under high pressure and temperature [(∼50 atm or 5,000 kPa) and 200°C]. Sodium carbonate or sodium hydroxide is then used to convert the fatty acids to their sodium salts (soap molecules):
Chemical reaction of the saponification (base hydrolysis) of a triglyceride.
As might be expected from the properties of the fatty acids, fats have a predominance of saturated fatty acids, and oils are composed largely of unsaturated acids. Thus, the melting points of triglycerides reflect their composition, as shown by the following examples. Natural mixed triglycerides have somewhat lower melting points, the melting point of lard being near 30 º C, whereas olive oil melts near -6 º C. Since fats are valued over oils by some Northern European and North American populations, vegetable oils are extensively converted to solid triglycerides (e.g. Crisco) by partial hydrogenation of their unsaturated components. Some of the remaining double bonds are isomerized (to trans) in this operation. These saturated and trans-fatty acid glycerides in the diet have been linked to long-term health issues such as atherosclerosis.

The double bonds in fats and oils can undergo hydrogenation and also oxidation. The hydrogenation of vegetable oils to produce semisolid fats is an important process in the food industry. Chemically, it is essentially identical to the catalytic hydrogenation reaction described for alkenes.
In commercial processes, the number of double bonds that are hydrogenated is carefully controlled to produce fats with the desired consistency (soft and pliable). Inexpensive and abundant vegetable oils (canola, corn, soybean) are thus transformed into margarine and cooking fats.
Many people have switched from butter to margarine or vegetable shortening because of concerns that saturated animal fats can raise blood cholesterol levels and result in clogged arteries. However, during the hydrogenation of vegetable oils, an isomerization reaction occurs that produces the trans fatty acids mentioned in the opening essay. However, studies have shown that trans fatty acids also raise cholesterol levels and increase the incidence of heart disease. Trans fatty acids do not have the bend in their structures, which occurs in cis fatty acids and thus pack closely together in the same way that the saturated fatty acids do. Consumers are now being advised to use polyunsaturated oils and soft or liquid margarine and reduce their total fat consumption to less than 30% of their total calorie intake each day.
Why do unsaturated fatty acids have lower melting points than saturated fatty acids?
- Answer
-
Unsaturated fatty acids cannot pack as tightly together as saturated fatty acids due to the presence of the cis double bond that puts a “kink” or bend in the hydrocarbon chain.
Classify each fatty acid as saturated or unsaturated and indicate the number of carbon atoms in each molecule.
- palmitoleic acid
- myristic acid
- linoleic acid
- Answer
-
- unsaturated; 16 carbon atoms
- saturated; 14 carbon atoms
- unsaturated; 18 carbon atoms
Write the condensed structural formula for each fatty acid.
- lauric acid
- palmitoleic acid
- linoleic acid
- Answer
-
- CH3(CH2)10COOH
- CH3(CH2)5CH=CH(CH2)7COOH
- CH3(CH2)3(CH2CH=CH)2(CH2)7COOH
Arrange these fatty acids (all contain 18 carbon atoms) in order of increasing melting point. Justify your arrangement.
a)
b)
c)
- Answer
-
c < a < b; an increase in the number of double bonds will lower the melting point because it is more difficult to closely pack the fatty acids together.
Which of these triglycerides would you expect to find in higher amounts in oils? In fats? Justify your choice.
- Answer
-
The triglyceride labeled as (a) is expected to be present in higher amounts in fats because it is composed of a greater number of saturated fatty acids. The triglyceride labeled as (b) is expected to be present in higher amounts in oils because it is composed of a greater number of unsaturated fatty acids.

