27.0: Introduction to Lipids
- Page ID
- 36481
After completing this section, you should be able to identify fats and steroids as being examples of lipids.
Make certain that you can define, and use in context, the key term below.
- lipid
Lipids are naturally occurring organic compounds that can be extracted from cells and tissues using nonpolar solvents. Although many lipids have complex structures, their chemistry can often be understood quite readily by applying the basic concepts you have studied in previous chapters. We begin the chapter with a study of fats and oils, and explain the different origins of these structurally similar substances. A discussion of soaps follows, as soap is obtained by the reaction of a fat with sodium hydroxide. Phospholipids are substances whose structures are somewhat similar to those of fats, except that the former contain a phosphate group. Prostaglandins are lipids that play important roles in biological systems and are of great interest to those involved in the medical and pharmaceutical professions. Terpenoids form a series of compounds whose structural similarity can be seen through the application of the isoprene rule. The chapter concludes with a discussion of steroids, with particular attention being paid to the stereochemistry of these substances.
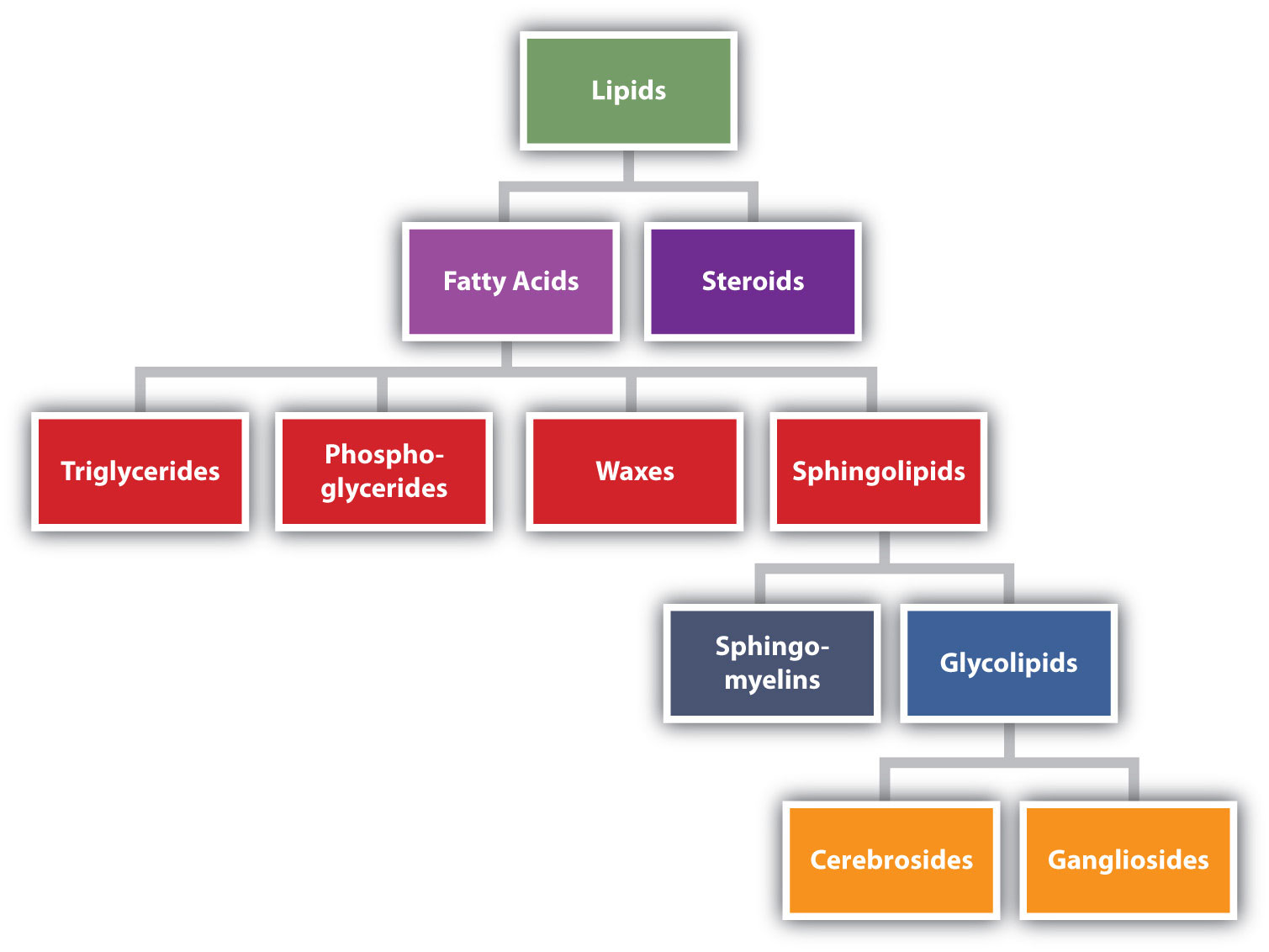
Lipids are not defined by the presence of specific functional groups, as carbohydrates are, but by a physical property—solubility. Compounds isolated from body tissues are classified as lipids if they are more soluble in organic solvents, such as dichloromethane, than in water. By this criterion, the lipid category includes not only fats and oils, which are esters of the trihydroxy alcohol glycerol and fatty acids, but also compounds that incorporate functional groups derived from phosphoric acid, carbohydrates, or amino alcohols, as well as steroid compounds such as cholesterol. Figure \(\PageIndex{1}\) presents one scheme for classifying the various kinds of lipids.

