15.4: Aromatic Ions
- Page ID
- 31565
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- use the Hückel 4n + 2 rule to explain the stability of the cyclopentadienyl anion, the cycloheptatrienyl cation and similar species.
- use the Hückel 4n + 2 rule to determine whether or not a given unsaturated cyclic hydrocarbon anion or cation is aromatic.
- draw the resonance contributors for the cyclopentadienyl anion, cation and radical, and similar species.
Aromatic Ions
As previously, noted a ring must be fully conjugated to have the potential to be aromatic. This means that every atom in the ring must have a p orbital which can overlap with adjacent p orbitals. Until now the atoms providing the p orbitals have been neutral sp2 hybridized carbons, however it is also possible for sp2 hybridized carbons to have a charge. There are several examples of cationic and anionic compounds with unexpected stabilities that suggest that they are aromatic. It is important to understand how charged carbons in these compounds will affect the determination of aromaticity.
There are two main situations which need to be considered: The conjugation of a carbocation and the conjugation of a carbanion. A carbocation carbon is surrounded by three electron groups giving it sp2 hybridization. The remaining unhybridized p orbital holds the carbocation's positive charge and is vacant of pi electrons. Although a carbocation is capable of extending conjugation it does not add to the compound's pi electron count.
A carbanion carbon is surrounded by four electron groups and would normally be sp3 hybridized. However, to obtain the stabilizing effects of conjugation, carbanion carbons can becomes sp2 hybridized putting the set of lone pair electrons into the unhybridized p orbital. Because the carbanion's p orbital contains two electrons in the form of a set of lone pair electrons, it increases a compound's pi electron count by 2.
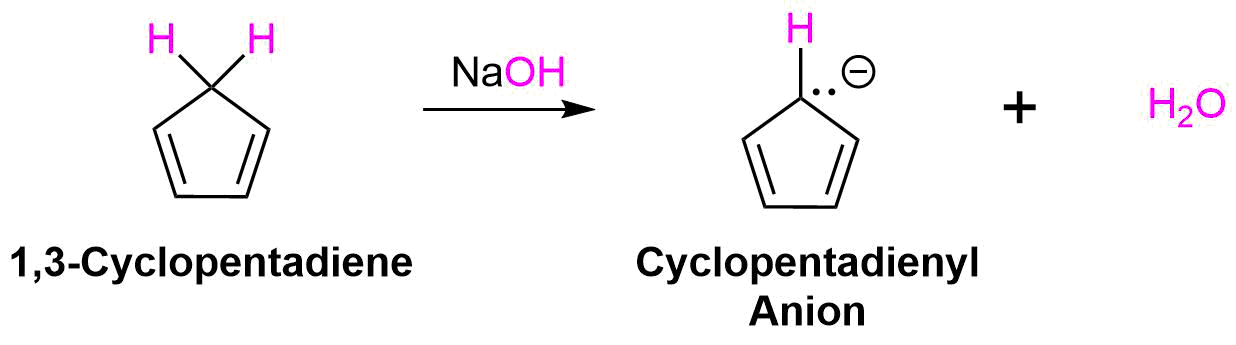
Cyclopentadiene Ion
One of the most well know examples of an aromatic ion is the 1,3-cyclopentadiene ion. 1,3-Cyclopentadiene is nonaromatic due to the presence of an intervening sp3 hybridized -CH2- carbon atom which prevents pi electrons from delocalizing about the entire ring. Also, it only has 4 pi electrons which does not follow Hückel's 4n + 2 rule. However, if a proton is removed form the CH2 group to form the cyclopentadienyl anion, the carbon atom becomes sp2 hybridized and the two electrons of the resulting lone pair occupy the newly produced p orbital. This increases the number of pi electrons in the cyclopentadienyl anion to 6 which follows the 4n +2 rule. Moreover, this new p orbital overlaps with the p orbitals already present allowing for cyclic delocalization of pi electrons about the entire ring.
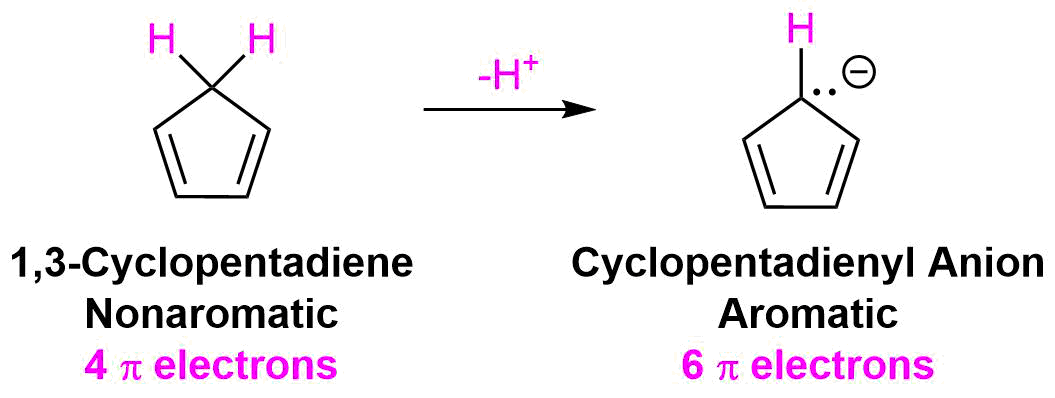
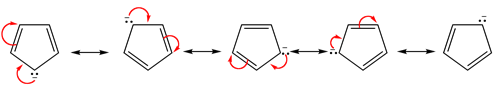
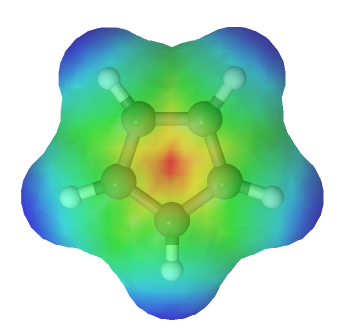
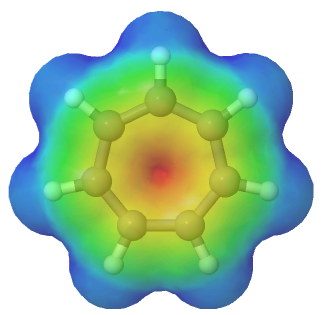
The lone pair electrons and the negative charge are conjugated about the entire ring making each carbon in the cyclopentadienyl anion equivalent with 1/5 the negative charge. The resonance hybrid can be shown by drawing a series of five resonance form. Also, the electrostatic potential map of the cyclopentadienyl anion show the negative charge, seen in red/yellow is distributed over the entire ring. The fact that the red/yellow color is held in the center of the ring indicates that the extra electrons of the ion are involved in the aromatic p-electron system.

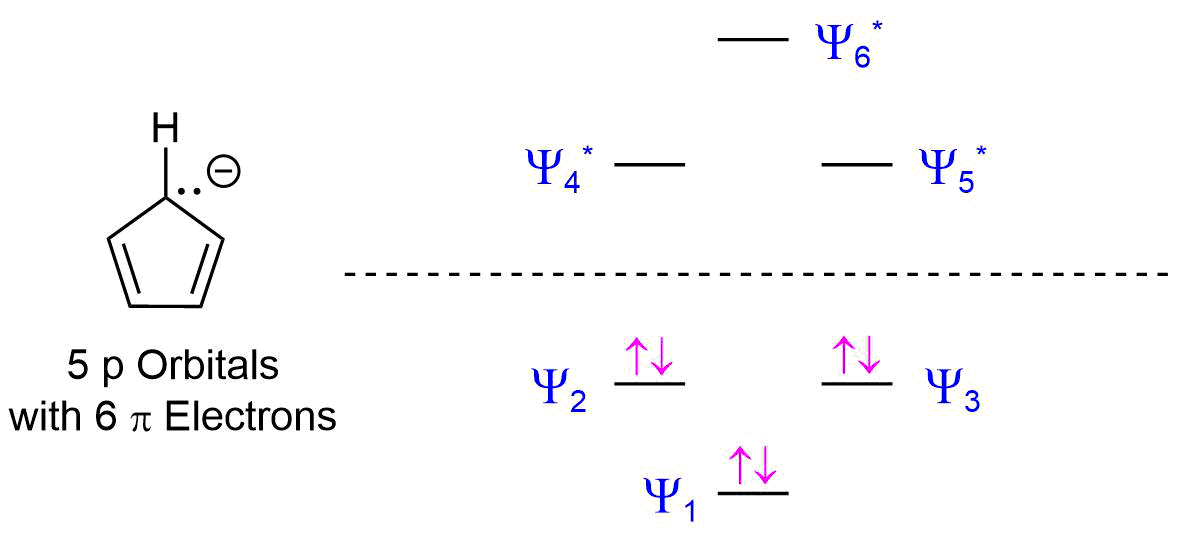
The reason why the 4n +2 rule still works for a 5 p orbital ring system can be seen by looking the molecular orbital diagram of the cyclopentadienyl anion. As discussed in Section 15.3, the molecular orbital diagram of a 5 p orbital system is made up of 3 bonding MO's and 2 antibonding MO's. The 6 pi electrons gained by forming an anion is enough to completely fill the bonding MO's in the diagram giving the cyclopentadienyl anion aromaticity.
One of the effects of the aromaticity of the cyclopentadienyl anion is that the acidity of 1,3-cyclopentadiene is unusually strong. As previously discussed, stabilizing the conjugate base increases the acidity of the corresponding acid. In this case, the conjugate base of 1,3-cyclopentadiene, the cyclopentadienyl anion, is stabilized through aromaticity. This makes 1,3-cyclopentadiene one of the most acidic hydrocarbons known with a pKa of 16. This is almost 1030 times more acidic than cyclopentane. Because of its acidity, cyclopentadiene can be deprotonated by moderately strong bases such as NaOH.
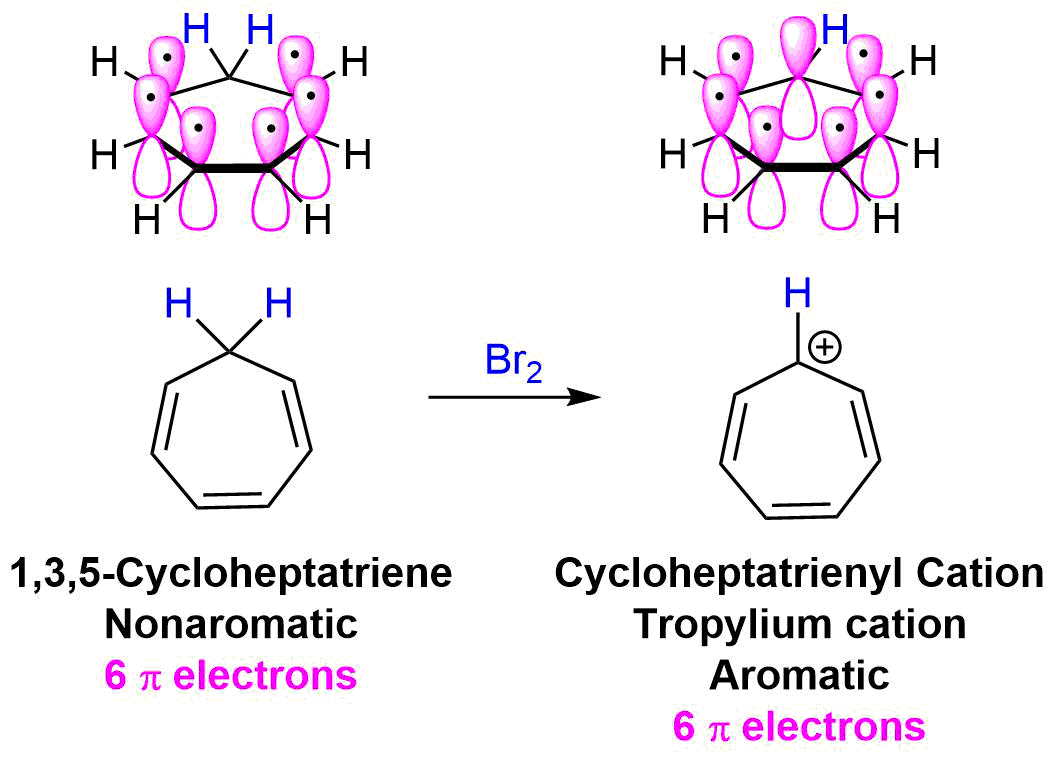
Tropylium ion
The molecule 1,3,5-cycloheptatriene has six pi electrons but is nonaromatic due the presence of an sp3 hybridized -CH2- group which prevents cyclic delocalization. When 1,3,5-cycloheptatriene is reacted with a reagent that can remove a hydride ion (H:-), the 1,3,5-cycloheptatrienyl cation, which is commonly known as the tropylium cation, is formed with unexpected ease. Despite the presence of an electron deficient carbocation, the tropylium cation, is unusually stable and can be isolated as a salt.
Removal of a hydride from the -CH2- group in 1,3,5-cycloheptatriene creates an sp2 hybridized carbocation with a vacant p orbital. The new p orbital allows for cyclic conjugation to occur among the seven p orbitals in the tropylium cation. The vacant p orbital does not change the pi electron count so the tropylium cation has 6 pi electrons which obeys the 4n + 2 rule for aromaticity.
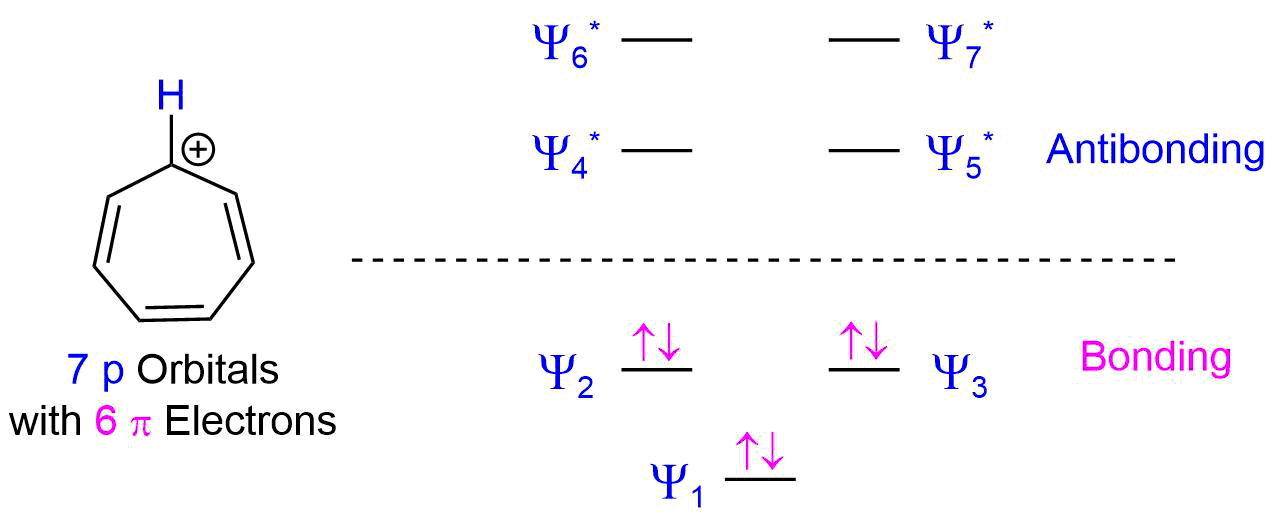
The molecular orbital diagram for the 7 p orbitals in the tropylium cation has three bonding MO's and 4 antibonding MO's. The tropylium cation's 6 pi electrons completely fill the bonding molecular orbitals which is consistent with the tropylium cation being aromatic and therefore unusually stable.
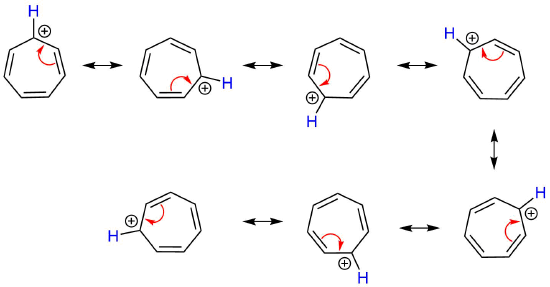
As predicted with aromaticity, the positive charge is completely conjugated about the entire ring giving each carbon +1/7 charge. The true resonance hybrid can be depicted by drawing a series of seven resonance form. Also, the electrostatic potential map of the tropylium cation shows the positive charge is evenly distributed over the entire ring. The equivalency of the carbons in the seven membered ring is experimentally supported by an 1H NMR spectrum of the tropylium cation which contains one peak showing that all seven protons are equivalent.

Antiaromatic Ions
In a similar fashion, cyclically conjugated ions with 4n pi electrons can be predicted to be antiaromatic and therefore highly unstable. An excellent example is the cyclopentadienyl cation. Above, the cyclopentadienyl anion was shown to be aromatic, however, the formation of a carbocation produces a different result. Although the vacant p orbital provided by the carbocation allows for cyclic conjugation to occur the compound only has 4 pi electrons.
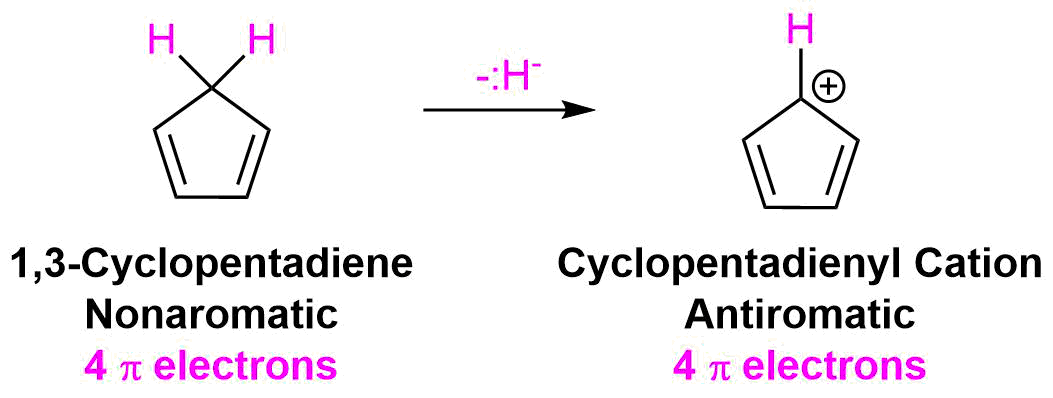
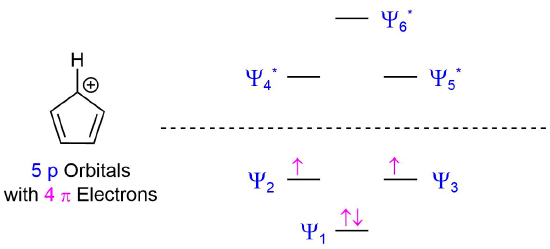
After placing these 4 pi electrons into the molecular orbital for a cyclic 5 p orbital species the bonding molecular orbitals remain unfilled. Two of the pi electrons are unpaired in degenerate molecular orbitals creating the highly unstable triplet state. As predicted by Hückel's Rule, the cyclopentadienyl cation has 4n pi electrons and should be anitaromatic and very unstable.
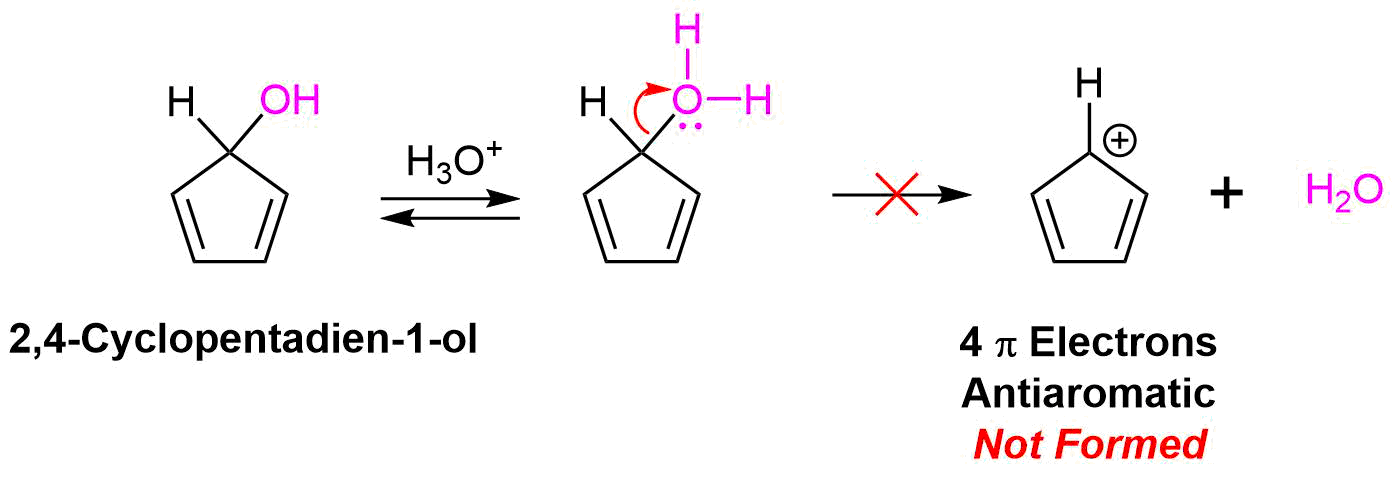
Although there is some discussion as to whether the cyclopentadienyl cation is truly antiaromatic, there is experimental evidence which shows it is usually unstable. In particular, the compound 2,4-cyclopentadien-1-ol is usually resistant to SN1 reactions with acid halides. The carbocation intermediate created during the mechanism of this reaction would be expected to easily form due to the resonance stabilization provided by the two double bonds. Because the carbocation intermediate is antiaromatic it does not form so the reaction does not occur.
1) Are all bonds equivalent cyclopentadienyl anion? How many lines (signals) would you expect to see in a H1 and C13 NMR spectrum?
2) The following reaction occurs readily. Propose a reason why this occurs?
3) Is the cyclopropenium ion aromatic? Use the 4n + 2 rule and the MO diagram for a 3 p orbital ring system to explain your answer.
- Answer
-
1) Due to the cyclopentadienyl anion being fully conjugated a protons and carbons are equivalent. Both the 1H and 13C NMR spectrum would only have one signal.
2) The addition of two electrons gives the ring system 10 pi electrons which follows the 4n+2 rule making the dianion aromatic. When looking at the MO diagram for an 8 p orbital ring system, 10 pi electrons completely fills the three bonding MO's and the two nonbonding MO's allowing for aromaticity.
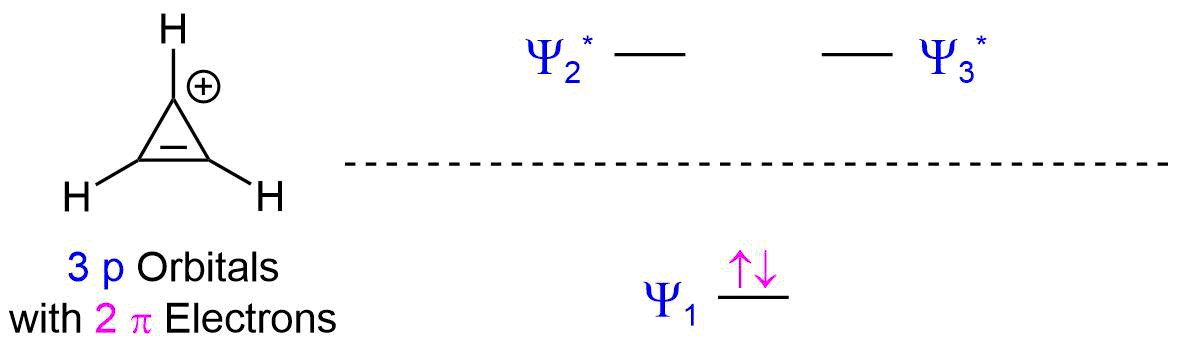
3) The cyclopropenium ion has 2 pi electrons which follows the 4n + 2 rule when n = 0. When considering the molecular orbital diagram for a 3 p orbital ring system the is one bonding MO and two antibonding MO's. The two pi electrons completely fill the bonding MO allowing for aromaticity.
Exercises
Draw the resonance structures for cycloheptatriene anion. Are all bonds equivalent? How many lines (signals) would you see in a H1 NMR? C13 NMR?
- Answer
-
All protons and carbons are the same, so therefore each spectrum will only have one signal each in the proton NMR and carbon NMR.
The following reaction occurs readily. Propose a reason why this occurs?
- Answer
-
The ring becomes aromatic with the addition of two electrons. Thereby obeying the 4n+2 rule.


