5.xx: Enantiomers and Diastereomers
- Page ID
- 92135
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to discuss how the results of work carried out by Biot and Pasteur contributed to the development of the concept of the tetrahedral carbon atom.
Because enantiomers have identical physical and chemical properties in achiral environments, separation of the stereoisomeric components of a racemic mixture or racemate is normally not possible by the conventional techniques of distillation and crystallization. In some cases, however, the crystal habits of solid enantiomers and racemates permit the chemist (acting as a chiral resolving agent) to discriminate enantiomeric components of a mixture. As background for the following example, it is recommended that the section on crystal properties be reviewed.
Tartaric acid, its potassium salt known in antiquity as "tartar", has served as the locus of several landmark events in the history of stereochemistry. 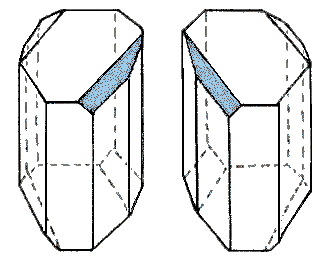 In 1832 the French chemist Jean Baptiste Biot observed that tartaric acid obtained from tartar was optically active, rotating the plane of polarized light clockwise (dextrorotatory). An optically inactive, higher melting, form of tartaric acid, called racemic acid was also known. A little more than a decade later, young Louis Pasteur conducted a careful study of the crystalline forms assumed by various salts of these acids. He noticed that under certain conditions, the sodium ammonium mixed salt of the racemic acid formed a mixture of enantiomorphic hemihedral crystals; a drawing of such a pair is shown on the right. Pasteur reasoned that the dissymmetry of the crystals might reflect the optical activity and dissymmetry of its component molecules. After picking the different crystals apart with a tweezer, he found that one group yielded the known dextrorotatory tartaric acid measured by Biot; the second led to a previously unknown levorotatory tartaric acid, having the same melting point as the dextrorotatory acid. Today we recognize that Pasteur had achieved the first resolution of a racemic mixture, and laid the foundation of what we now call stereochemistry.
In 1832 the French chemist Jean Baptiste Biot observed that tartaric acid obtained from tartar was optically active, rotating the plane of polarized light clockwise (dextrorotatory). An optically inactive, higher melting, form of tartaric acid, called racemic acid was also known. A little more than a decade later, young Louis Pasteur conducted a careful study of the crystalline forms assumed by various salts of these acids. He noticed that under certain conditions, the sodium ammonium mixed salt of the racemic acid formed a mixture of enantiomorphic hemihedral crystals; a drawing of such a pair is shown on the right. Pasteur reasoned that the dissymmetry of the crystals might reflect the optical activity and dissymmetry of its component molecules. After picking the different crystals apart with a tweezer, he found that one group yielded the known dextrorotatory tartaric acid measured by Biot; the second led to a previously unknown levorotatory tartaric acid, having the same melting point as the dextrorotatory acid. Today we recognize that Pasteur had achieved the first resolution of a racemic mixture, and laid the foundation of what we now call stereochemistry.
Optical activity was first observed by the French physicist Jean-Baptiste Biot. He concluded that the change in direction of plane-polarized light when it passed through certain substances was actually a rotation of light, and that it had a molecular basis. His work was supported by the experimentation of Louis Pasteur. Pasteur observed the existence of two crystals that were mirror images in tartaric acid, an acid found in wine. Through meticulous experimentation, he found that one set of molecules rotated polarized light clockwise while the other rotated light counterclockwise to the same extent. He also observed that a mixture of both, a racemic mixture (or racemic modification), did not rotate light because the optical activity of one molecule canceled the effects of the other molecule. Pasteur was the first to show the existence of chiral molecules.
When you have one enantiomer that rotates plane polarized light clockwise, why does a racemic mixture of that compound not rotate plane polarized light?
- Answer
-
Since a racemic mixture consists of equal amounts of both enantiomers, one enantiomer will rotate plane polarized light clockwise, while the other enantiomer will rotate plane polarized light counterclockwise an equal amount, causing them to cancel out, and the racemic mixture being not optically active.

