26.2: Quinones
- Page ID
- 22372
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Quinones are not aromatic compounds but are conjugated cyclic diketones. Yet it is convenient to discuss their chemistry at this point because quinones and the related aromatic arenols are readily interconverted, and their chemistry is largely interdependent.
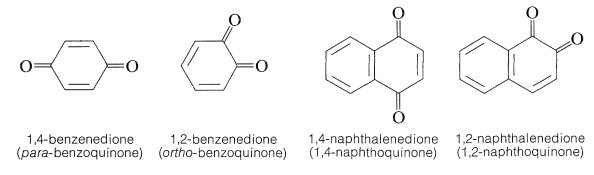
A variety of quinonelike structures have been prepared, the most common of which are the 1,2 - and 1,4-quinones as exemplified by 1,2- and 1,4-benzenediones. Usually the 1,2-quinones are more difficult to make and are more reactive than the 1,4-quinones. A few 1,6- and 1,8-quinones also are known.
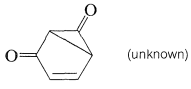
No 1,3-quinones are known, possibly because they would have nonplanar, highly strained structures and therefore would be unstable:
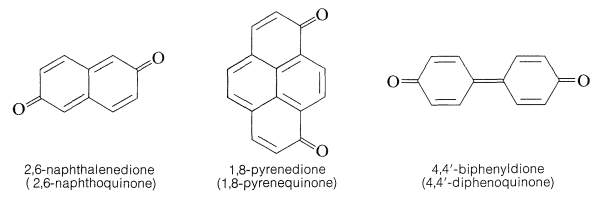
A number of quinones are known in which the quinone arrangement extends over more than one ring. Examples are:
Reduction of Quinones
A characteristic and important reaction of quinones is reduction to the corresponding arenediols. The reduction products of 1,4-quinones are called hydroquinones:
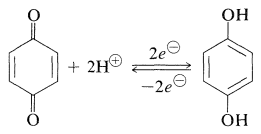 \(\tag{26-2}\)
\(\tag{26-2}\)
Reduction can be achieved electrochemically and with a variety of reducing agents (metals in aqueous acid, catalytic hydrogenation). Such reductions are unusual among organic reactions in being sufficiently rapid and reversible to give easily reproducible electrode potentials in an electrolytic cell. The position of the 1,4-benzenediol-1,4,-benzenedione equilibrium (Equation 26-2) is proportional to the square of the hydrogen-ion concentration. Therefore the electrode potential is sensitive to pH; a change of one unit of pH in water solution changes the potential of the electrode by \(0.059 \: \text{V}\). Before the invention of the glass-electrode pH meter, the half-cell potential developed by this equilibrium was used widely to determine pH values of aqueous solutions. The method is not very useful above pH 9 because the quinone reacts irreversibly with alkali.
Numerous studies have been made of the relationship between half-cell reduction potentials and the structures of quinones. As might be expected, the potentials are greatest when the resonance stabilization associated with formation of the aromatic ring is greatest.
When alcohols solutions of hydroquinone and quinone are mixed, a brown-red color develops and a green-black 1:1 complex crystallizes that is known as quinhydrone. This substance is a charge-transfer complex (Section 24-6C), with the diol acting as the electron donor and the dione as the electron acceptor. Quinhydrone is not very soluble and dissociates considerably to its components in solution.
The reduction of a quinone requires two electrons, and it is possible that these electrons could be transferred either together or one at a time. The product of a single-electron transfer leads to what appropriately is called a semiquinone, \(1\), with both a negative charge and an odd electron (a radical anion):
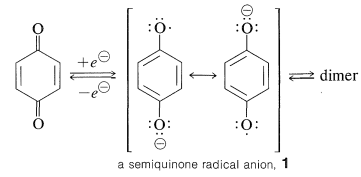
The formation of relatively stable semiquinone radicals by electrolytic reduction of quinones has been established by a variety of methods. Some semiquinone radicals undergo reversible dimerization reactions to form peroxides.
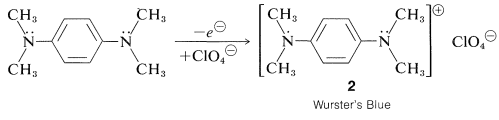
A particularly stable cation-radical of the semiquinone type is formed by mild oxidation of \(\ce{N}\),\(\ce{N}\),\(\ce{N'}\),\(\ce{N'}\)-tetramethyl-1,4-benzenediamine. The cation, which is isolable as a brilliant-blue perchlorate salt, \(2\), is called "Wurster's Blue":
Quinones of Biological Importance
Oxidation and reduction in biochemical systems involve many reactions that are similar to the arenediol-arenediol couple. We have mentioned several previously: \(\ce{NADP}^\oplus \rightleftharpoons \ce{NADPH}\) (Section 20-9), and \(\ce{FADH_2} \rightleftharpoons \ce{FAD}\) (Section 15-6C).
An important question in metabolic oxidation is just how reduction of oxygen \(\left( \frac{1}{2} \ce{O_2} + 2 \ce{H}^\oplus + 2 \ce{e} \rightarrow \ce{H_2O} \right)\) is linked to the oxidation of \(\ce{NADH}\) \(\left( \ce{NADH} \rightarrow \ce{NAD}^\oplus + \ce{H}^\oplus + 2 \ce{e} \right)\). The route for transfer of electrons from \(\ce{NADH}\) to oxygen (oxidation plus phosphorylation; Section 20-10) is indirect, complicated, and involves, in an early stage, oxidation of \(\ce{NADH}\) by flavin mononucleotide \(\left( \ce{FMN} \right)\) by the reaction \(\ce{FMN} + \ce{NADH} + \ce{H}^\oplus \rightarrow \ce{FMNH_2} + \ce{NAD}^\oplus\). But the reduced form of \(\ce{FMN}\), \(\ce{FMNH_2}\), does not react directly with oxygen. Instead, it reduces a quinone called coenzyme Q (CoQ) to the corresponding arenediol:
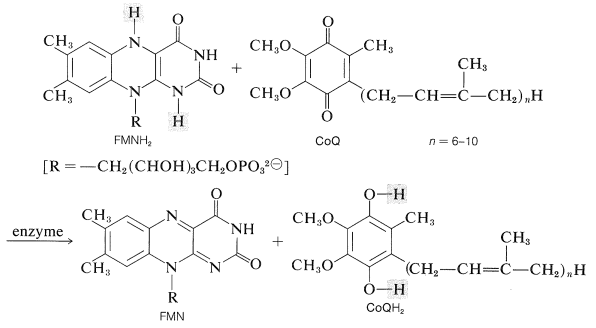
The effect of this step is to form a slightly polar reductant (CoQ\(\ce{H_2}\)) from a strongly polar reductant \(\left( \ce{FMNH_2} \right)\), and this permits the reduced material to penetrate into a less polar region of the oxidative apparatus. The reduced CoQ does not react directly with oxygen but is a participant in a chain of oxidation-reduction reactions involving electron transfer between a number of iron-containing proteins known as cytochromes. At the end of this chain of reaction, the reduced form of a copper-containing cytochrome actually reacts with oxygen. The sequence of electron-carriers may be summarized as
\[\ce{NADH} \rightarrow \ce{FMNH_2} \rightarrow \textbf{CoQ}\ce{H_2} \rightarrow \text{cytochromes} \rightarrow \ce{O_2}\]
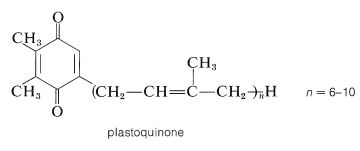
A related process occurs in photosynthesis (Section 20-9). You will recall that a critical part of photosynthesis involves the transfer of electrons from the photosystem that oxidizes water \(\left( \ce{H_2O} \rightarrow \frac{1}{2} \ce{O_2} + 2 \ce{H}^\oplus + 2 \ce{e} \right)\) to the photosystem that reduces \(\ce{NADP}^\oplus\) \(\left( \ce{NADP}^\oplus + 2 \ce{H}^\oplus + 2 \ce{e} \rightarrow \ce{NADPH} + \ce{H}^\oplus \right)\). As in oxidative phosphorylation, the electron-transfer route is complex. However, one of the electron carriers is a quinone called plastoquinone that closely resembles coenzyme Q found in animals. Plastoquinone, like coenzyme Q, is reduced to the hydroquinone form, which is part of an electron-transport chain involving iron- and copper-containing proteins:
Among other naturally occurring substances having quinone-type structures, one of the most important is the blood antihemorrhagic factor, vitamin K\(_1\), which occurs in green plants and is a substituted 1,4-naphthalenedione:
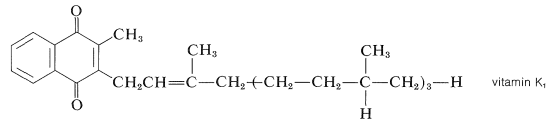
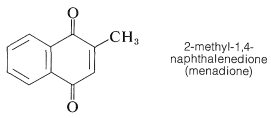
The structure of vitamin K\(_1\) has been established by degradation and by synthesis. Surprisingly, the long alkyl side chain of vitamin K\(_1\) is not necessary for its action in aiding blood clotting because 2-methyl-1,4-naphthoquinone is almost equally active on a molar basis.
Besides playing a vital role in the oxidation-reduction processes of living organisms, quinones occur widely as natural pigments found mainly in plants, fungi, lichens, marine organisms, and insects (see alizarin, Section 28-4A, as representative of a natural anthraquinone-type dye).
Photographic Developers
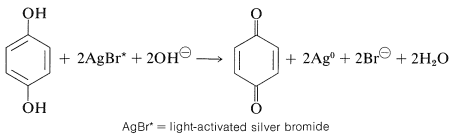
Photography makes important practical use of the arenediol-arenediol oxidation-reduction system. Exposure of the minute grains of silver bromide in a photographic emulsion to blue light (or any visible light in the presence of suitably sensitizing dyes) produces a stable activated form of silver bromide, the activation involving generation of some sort of crystal defect. Subsequently, when the emulsion is brought into contact with a developer, which may be an alkaline aqueous solution of 1,4-benzenediol (hydroquinone) and sodium sulfite, the particles of activated silver bromide are reduced to silver metal much more rapidly than the ordinary silver bromide. Removal of the unreduced silver bromide with sodium thiosulfate ("fixing") leaves a suspension of finely divided silver in the emulsion in the form of the familiar photographic negative.
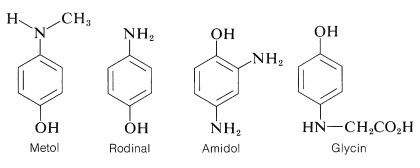
A variety of compounds are used as photographic developing agents. They are not all arenediols. In fact, most are aromatic aminoalcohols or diamines, but irrespective of their structural differences, they all possess the ability to undergo redox reactions of the type described for 1,4-benzenediol. Structural formulas and commercial names for several important developers are
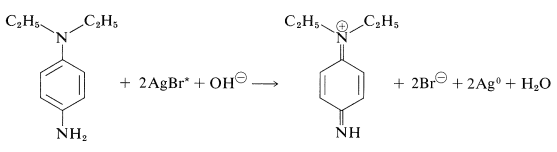
1,4-Benzenediamine also is an effective developing agent, but it may cause dermatitis in sensitive individuals. \(\ce{N}\),\(\ce{N}\)-Diethyl-1,4-benzenediamine is used as a developer in color photography. These substances react with silver bromide to produce benzenediamine derivatives:
Addition Reactions of Quinones
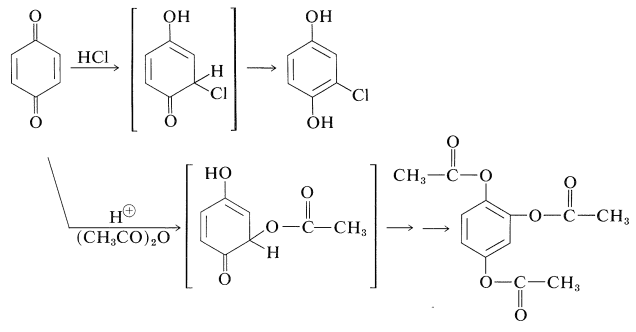
Being \(\alpha\),\(\beta\)-unsaturated ketones, quinones have the potential of forming 1,4-addition products in the same way as their open-chain analogs (Section 17-5B). 1,4-Benzenedione undergoes such additions rather readily. The products are unstable and undergo enolization to give substituted 1,4-benzenediols. Two examples are the addition of hydrogen chloride and the acid-catalyzed addition of ethanoic anhydride. In the latter reaction, the hydroxyl groups of the adduct are acylated by the anhydride. Hydrolysis of the product yields 1,2,4-benzenetriol:
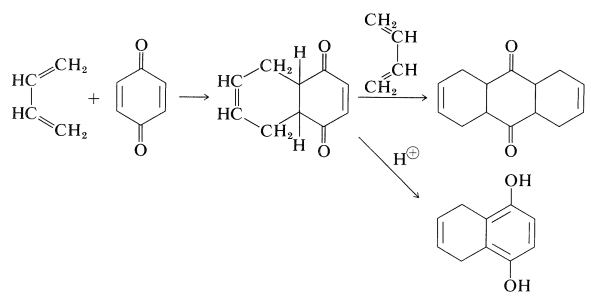
Quinones usually undergo Diels-Alder additions readily, provided that they have at least one double bond that is not part of an aromatic ring. With 1,4-benzenedione and 1,3-butadiene, either the mono- or diadduct can be obtained. The monoadduct enolizes under the influence of acid or base to a 1,4-benzenediol derivative:
Quinones of Cyclobutadiene
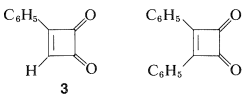
Benzoquinones owe their unusual properties as \(\alpha\),\(\beta\)-unsaturated ketones to the ease by which they are transformed to stable aromatic systems. How would these properties change if the quinone were derived from nonaromatic structures, such as cyclobutadiene, cyclooctatetraene, or pentalene? There is no final answer to this question because few such substances have been prepared, the best known so far being the mono- and diphenylcyclobutenediones:
For example, \(3\) can be prepared from sulfuric acid hydrolysis of the cycloaddition product of ethynylbenzene and trifluorochloroethene (Section 13-3D):
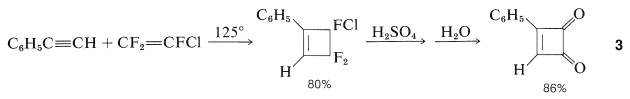
The dione, \(3\), is a yellow crystalline solid that, despite its strained four-membered ring, is much less reactive than 1,2-benzenedione (ortho-benzoquinone). It cannot be reduced to a cyclobutenediol, does not undergo Diels-Alder reactions, and with bromine gives a substitution product rather than addition. The bromo compound so formed hydrolyzes rapidly to a hydroxy compound, \(4\), which is an extraordinarily strong acid having an ionization constant about \(10^9\) times that of benzenol:
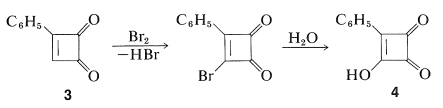
A related compound, 3,4-dihydroxy-1,2-cyclobutenedione, \(5\), also has been prepared and is a very strong dibasic acid. It is sometimes called "squaric acid":
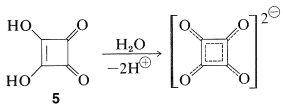
From the data so far available, it appears that the quinones corresponding to cyclobutadiene have more aromatic character than do the cyclobutadienes themselves.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


