8.1: Nucleotides -the building blocks of nucleic acids
- Page ID
- 431335
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Understand the function of nucleic acids.
- Learn the structures and names of nucleotides -the building blocks of nucleic acids, and their constituents, including ribose, deoxyribose, nitrogen bases, and phosphate groups.
Nucleic acids are biopolymers that carry the codes for synthesizing proteins and pass them on from generation to generation, i.e., they are genetic materials. In other words, nucleic acids are the instruction manual for biochemical reactions in living things.
Nucleotides are the building blocks, i.e., the repeat units or monomers of nucleic acids.
Nucleotides are composed of three sub-units:
- a 5-carbon carbohydrate,
- a base that is an aromatic compound containing nitrogen, and
- an anion of phosphoric acid, i.e., phosphate (\(\ce{PO4^{3-}}\)).
5-Carbon carbohydrate
Two 5-carbon carbohydrates, i.e., ribose and deoxyribose, are found in nucleic acids, as shown in the figure below. Both are D-sugars in five-membered cyclic form with hydroxyl (\(\ce{-OH}\)) group at anomeric (\(\ce{C}\)#1') pointing towards \(\ce{O}\) in the ring, i.e., in a \(\beta\) configuration. The number labels have prime symbols, i.e., 1', 2', 3', etc., to distinguish them from regular numbers 1, 2, 3, etc., used for the nitrogen bases of the nucleic acids. The only difference between ribose and deoxyribose is that hydroxyl (\(\ce{-OH}\)) group at \(\ce{C}\)#2' is missing in the latter, which gives the deoxy- prefix to its name.

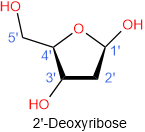
There are two types of nucleic acids: ribose in its nucleotides, called ribonucleic acid (RNA), and deoxyribose in its nucleotides, called deoxyribonucleic acid (DNA).
Nitrogen bases
Nitrogen bases in nucleic acids are derivatives of two aromatic compounds, purine and pyrimidine, shown below.


Cyclic compounds that contain atoms other than \(\ce{C's}\) in the cycle are called heterocyclic compounds. Purine and pyrimidine are heterocyclic aromatic compounds because they contain \(\ce{N's}\) in the cycles. Purine is bicyclic, containing a six-membered ring with two \(\ce{N's}\) fused with a five-membered ring with two \(\ce{N's}\). Pyrimidine is a six-membered cycle with two \(\ce{N's}\). Both purine and pyrimidine are planer molecules like benzene.
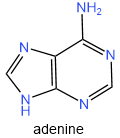
There are five nitrogen bases found in nucleic acids: two are purines, i.e., adenine (A) and guanine (G), and three are pyrimidines, i.e., cytosine (C), thymine (T), and uracil (U), as shown below. To remember what are pyrimidines, remember "cut pyramid" where 'c' is cytosine, 'u' for uracil, 't' for thymine, and 'pyramid' for pyrimidines
| Purines | Pyrimidines | |||
|---|---|---|---|---|
 |
 |
 |
 |
 |
DNA contains four nitrogen bases, i.e., adenine, guanine, cytosine, and thymine. RNA also has four nitrogen bases, i.e., adenine, guanine, cytosine, and uracil. Note that the first three, i.e., adenine, guanine, and cytosine, are common in DNA and RNA, but the fourth, i.e., ti.e.ine in DNA, is replaced with uracil in RNA.
Nucleosides
When a monomer sugar like ribose or deoxyribose reacts with an amine, the \(\ce{-OH}\) group at \(\ce{C}\)#1' is replaced with a \(\ce{N}\) of the amine. The product is called N-glycoside, and the \(\ce{C-N}\) bond in the N-glycoside is called an N-glycosidic bond, as shown in the following example.

Purines connect through \(\ce{N}\)#9 and pyrimidine through \(\ce{N}\)#1 with anomeric carbon (\(\ce{C}\)#1') of ribose or deoxyribose to form the N-glycoside.
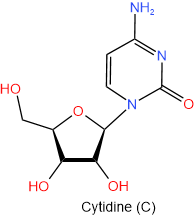
The N-glycosides are named by using the name of purine but ending with -osine e.g., adenine becomes adenosine and guanine becomes gunosine, or by using the name of pyrimidine ending with -idine, e.g., cytosine becomes cytidine and uracil become uridine. One letter abbreviation of the nitrogen bases remains the same for the corresponding nucleoside, e.g., adenine (A) and adenosine (A). The four nucleosides found in RNA are shown below with their names and one-letter abbreviations.
 |
 |
 |
 |
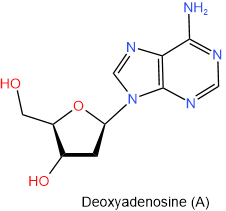
In DNA there is deoxyribose in place of ribose. So the names of nucleosides found in DNA begin with the deoxy- prefix. The four nucleosides found in DNA are shown below with their names and one-letter abbreviations.
 |
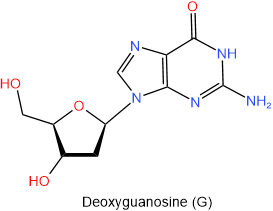 |
 |
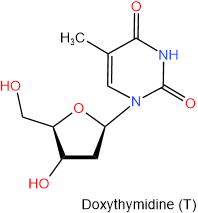 |
Phosphate
Phosphoric acid (\(\ce{H3PO4}\)) is an oxyacid with three acidic protons, i.e., it is triprotic acid. Just as two carboxylic acids condense with each other and form a carboxylic acid anhydride with the elimination of water molecule, two phosphoric acids condense and form diphosphoric acid, also called pyrophosphoric acid, with the elimination of water.
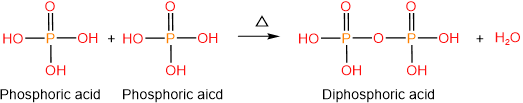
Similarly, three phosphoric acids can condense to form triphosphoric acid. Phosphoric acid, diphosphoric acid, and triphosphoric acid are shown below.



Under physiological conditions at pH ~7.4, the phosphoric acids lose proton and exist as phosphate anions, as shown below.

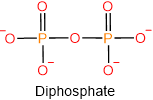

Phosphoric acids also condense with alcohols (\(\ce{R-OH}\)) and form mono-, di-, or triesters, as illustrated below.


Nucleotide
Phosphate esters of nucleosides are called nucleotides. The nucleotides found in nucleic acids are formed when \(\ce{-OH}\) group at \(\ce{C}\)#5' of ribose or deoxyribose makes ester with a monophosphate, diphosphate, or triphosphate, as shown below.



The names of the nucleotides are derived by using the name of the corresponding nucleoside with monophosphate, diphosphate, or triphosphate added at the end. Abbreviation of the nucleotide starts with the one-letter abbreviation of the nitrogen base followed by MP, DP, or TP depending on whether the phosphate group is monophosphate, diphosphate, or triphosphate, as shown in the example structures above.
The nucleotides like ADP and ATP are high-energy molecules used as energy currency in biochemical systems. For example, when ATP converts to ADP by releasing a phosphate, energy is released to do work or synthesize other compounds. Synthesis of nucleic acids begins with triphosphate esters that convert into monophosphate esters when incorporated in the nucleic acid polymer. The energy released in converting triphosphate to monophosphate drives the reaction forward.
The four nucleotides found in RNA are shown below with their names and abbreviations.
 ,
,  ,
,  , and
, and 
The nucleotides found in DNA are named similarly to those of RNA: start with the name of the corresponding nucleoside and end with monophosphate, diphosphate, or triphosphate. Remember that the names of DNA nucleosides begin with deoxy. The abbreviations of the nucleotides found in DNA start with a small alphabet d representing the deoxy- prefix of the nucleotides. The four nucleotides found in DNA are shown below with their names and abbreviations.
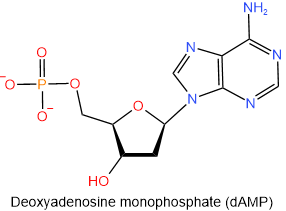 ,
, 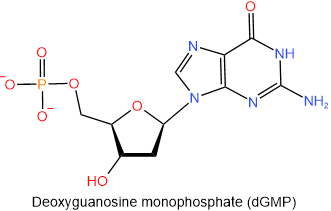 ,
,  ,
, 


