The Extraction of Copper
- Page ID
- 3687
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To understand the basic principles of copper extraction from ores
- To understand the basis of it s purification by electrolysis
- To identify some common uses of copper
Extracting copper from its ores
The method used to extract copper from its ores depends on the nature of the ore. Sulfide ores such as chalcopyrite (\(CuFeS_2\)) are converted to copper by a different method from silicate, carbonate or sulfate ores. Chalcopyrite (also known as copper pyrites) and similar sulfide ores are the commonest ores of copper. The ores typically contain low percentages of copper and have to be concentrated before refining (e.g., via froth flotation).

The Process
The concentrated ore is heated strongly with silicon dioxide (silica) and air or oxygen in a furnace or series of furnaces.
- The copper(II) ions in the chalcopyrite are reduced to copper(I) sulfide (which is reduced further to copper metal in the final stage).
- The iron in the chalcopyrite ends up converted into an iron(II) silicate slag which is removed.
- Most of the sulfur in the chalcopyrite turns into sulfur dioxide gas. This is used to make sulfuric acid via the Contact Process.
An overall equation for this series of steps is:
\[2CuFeS_2 + 2SiO_2 +4O_2 \rightarrow Cu_2S + 2FeSiO_3 + 3SO_2 \label{1} \]
The copper(I) sulfide produced is converted to copper with a final blast of air.
\[ Cu_2S + O_2 \rightarrow 2Cu + SO_2 \label{2} \]
The end product of this is called blister copper - a porous brittle form of copper, about 98 - 99.5% pure.
Exploring the redox processes in this reaction
It is worthwhile spending some time sorting out what the reducing agent is in these reactions, because at first sight there does not appear to be one! Or, if you look superficially, it seems as if it might be oxygen! But that's silly! We'll start by looking at the second reaction because it is much easier to see what is happening.
\[ Cu_2S + O_2 \rightarrow 2Cu + SO_2 \label{3} \]
Let's look at the oxidation states of everything.
- In the copper(I) sulfide, the copper is +1 and the sulfur -2.
- The oxidation states of the elements oxygen (in the gas) and copper (in the metal) are 0.
- In sulfur dioxide, the oxygen has an oxidation state of -2 and the sulfur +4.
That means that both the copper and the oxygen have been reduced (decrease in oxidation state). The sulfur has been oxidized (increase in oxidation state). The reducing agent is therefore the sulfide ion in the copper(I) sulfide.
The other reaction is more difficult to deal with, because you can't work out all of the oxidation states by following the simple rules - there are too many variables in some of the substances. You have to use some chemical knowledge as well.
\[ 2CuFeS_2 + 2SiO_2 + 4O_2 \rightarrow Cu_2S + 2FeSiO_3 + 3SO_2 \label{4} \]
In the CuFeS2, you would have to know that the copper and iron are both in oxidation state +2, for example. You would also have to know that the oxidation state of the silicon remains unchanged at +4. So use that information to work out what has been oxidized and what reduced in this case!
You should find that copper has been reduced from +2 to +1; oxygen (in the gas) has been reduced from 0 to -2 (oxygen in the SiO2 is unchanged); and three of the four sulfurs on the left-hand side have been oxidized from -2 to +4 (the other is unchanged). Once again, the sulfide ions are acting as the reducing agent.
Extracting of Copper from Other Ores
Copper can be extracted from non-sulfide ores by a different process involving three separate stages:
- Reaction of the ore (over quite a long time and on a huge scale) with a dilute acid such as dilute sulfuric acid to produce a very dilute copper(II) sulfate solution.
- Concentration of the copper(II) sulfate solution by solvent extraction. The very dilute solution is brought into contact with a relatively small amount of an organic solvent containing something which will bind with copper(II) ions so that they are removed from the dilute solution. The solvent must not mix with the water. The copper(II) ions are removed again from the organic solvent by reaction with fresh sulfuric acid, producing a much more concentrated copper(II) sulfate solution than before.
- Electrolysis of the new solution. Copper(II) ions are deposited as copper on the cathode (for the electrode equation, see under the purification of copper below). The anodes for this process were traditionally lead-based alloys, but newer methods use titanium or stainless steel. The cathode is either a strip of very pure copper which the new copper plates on to, or stainless steel which it has to be removed from later.
Purification of copper
When copper is made from sulfide ores by the first method above, it is impure. The blister copper is first treated to remove any remaining sulfur (trapped as bubbles of sulfur dioxide in the copper - hence "blister copper") and then cast into anodes for refining using electrolysis.
Electrolytic Refining
The purification uses an electrolyte of copper(II) sulfate solution, impure copper anodes, and strips of high purity copper for the cathodes. The diagram shows a very simplified view of a cell.
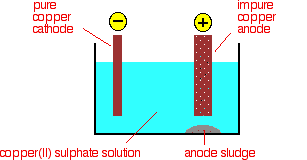
At the cathode, copper(II) ions are deposited as copper.
\[ Cu^{2+}(aq) + 2e^- \rightarrow Cu(s) \label{5a} \]
At the anode, copper goes into solution as copper(II) ions.
\[ Cu (s) \rightarrow Cu^{2+} (aq) + 2e^- \label{6a} \]
For every copper ion that is deposited at the cathode, in principle another one goes into solution at the anode. The concentration of the solution should stay the same. All that happens is that there is a transfer of copper from the anode to the cathode. The cathode gets bigger as more and more pure copper is deposited; the anode gradually disappears. In practice, it isn't quite as simple as that because of the impurities involved.
What happens to the impurities?
Any metal in the impure anode which is below copper in the electrochemical series (reactivity series) does not go into solution as ions. It stays as a metal and falls to the bottom of the cell as an "anode sludge" together with any unreactive material left over from the ore. The anode sludge will contain valuable metals such as silver and gold.
Metals above copper in the electrochemical series (like zinc) will form ions at the anode and go into solution. However, they won't get discharged at the cathode provided their concentration does not get too high. The concentration of ions like zinc will increase with time, and the concentration of the copper(II) ions in the solution will fall. For every zinc ion going into solution there will obviously be one fewer copper ion formed. (See the next note if you aren't sure about this.) The copper(II) sulfate solution has to be continuously purified to make up for this.
Uses of copper
Amongst other things copper is used for:
- electrical wiring. It is a very good conductor of electricity and is easily drawn out into wires.
- domestic plumbing. It does not react with water, and is easily bent into shape.
- boilers and heat exchangers. It is a good conductor of heat and does not react with water.
- making brass. Brass is a copper-zinc alloy. Alloying produces a metal harder than either copper or zinc individually. Bronze is another copper alloy, but with tin.
- coinage. In many countries, as well as the more obvious copper-colored coins, "silver" coins are also copper alloys - this time with nickel; these are known as cupronickel alloys. UK pound coins and the gold-colored bits of euro coins are copper-zinc-nickel alloys.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


