1.2: Chemical Bonding
- Page ID
- 58710
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)
#mt-toc-container {
display: none !important;
}
Objectives
After completing this section, you should be able to
- explain how covalent bonds are formed as a result of the ability of atoms to share electrons.
- draw Lewis structures (also known as Lewis formulas or electron-dot formulas) of simple species, such as CH4, H2O, H3O+, CH3OH, and NH3, without the aid of a periodic table.
- draw structural formulas (i.e., Kekulé structures or line-bond structures) of simple species without the aid of a periodic table.
- describe the formation of covalent bonds in terms of the overlapping of atomic orbitals.
Key Terms
Make certain that you can define, and use in context, the key terms below.
- bond strength
- covalent bond
- ionic bond
- Lewis structure
- lone-pair electron
- non-bonding electron
- sigma (σ) bond
- pi (π) bond
- valence bond theory
Study Notes
To draw Lewis structures successfully, you need to know the number of valence electrons present in each of the atoms involved. Memorize the number of valence electrons possessed by each of the elements commonly encountered in organic chemistry: C, H, O, N, S, P and the halogens.
Ionic bonding
Ions are atoms or molecules which are electrically charged. Cations are positively charged and anions carry a negative charge. Ions form when atoms gain or lose electrons. Since electrons are negatively charged, an atom that loses one or more electrons will become positively charged; an atom that gains one or more electrons becomes negatively charged.
Ionic bonding is the attraction between positively- and negatively-charged ions. These oppositely charged ions attract each other to form ionic networks (or lattices). Electrostatics explains why this happens: opposite charges attract and like charges repel. When many ions attract each other, they form large, ordered, crystal lattices in which each ion is surrounded by ions of the opposite charge. Generally, when metals react with non-metals, electrons are transferred from the metals to the non-metals. The metals form positively-charged ions and the non-metals form negatively-charged ions.
Ionic bonds form when metals and non-metals chemically react. By definition, a metal is relatively stable if it loses electrons to form a complete valence shell and becomes positively charged. Likewise, a non-metal becomes more stable by gaining electrons to complete its valence shell and become negatively charged. When metals and non-metals react, the metals lose electrons by transferring them to the non-metals, which gain them. Consequently, ions are formed, which instantly attract each other—ionic bonding.
Example 1.5.1: Sodium Chloride
For example, in the reaction of Na (sodium) and Cl (chlorine), each Cl atom takes one electron from a Na atom. Therefore each Na becomes a Na+ cation and each Cl atom becomes a Cl- anion. Due to their opposite charges, the ions of sodium and chloride attract each other to form an ionic lattice. The formula (ratio of positive to negative ions) in the lattice is NaCl.
For full video of making NaCl from sodium metal and chlorine gase, see https://www.youtube.com/watch?v=WVonuBjCrNo. These ions are arranged in solid NaCl in a regular three-dimensional arrangement (or lattice):
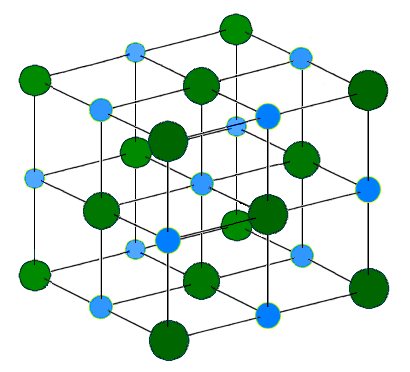
Figure: NaCl lattice. (left) 3-D structure and (right) simple 2D slice through lattes. Images used with permission from Wikipedia and Mike Blaber.
The chlorine has a high affinity for electrons, and the sodium has a low ionization potential. Thus the chlorine gains an electron from the sodium atom. This can be represented using electron-dot symbols (here we will consider one chlorine atom, rather than Cl2):
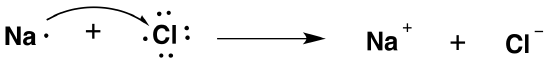
The arrow indicates the transfer of the electron from sodium to chlorine to form the Na+ metal ion and the Cl- chloride ion. Each ion now has an octet of electrons in its valence shell:
- Na+: 2s22p6
- Cl-: 3s23p6
The importance of noble gas structures![]()
A lot of importance is attached to the electronic structures of noble gases like neon or argon which have eight electrons in their outer energy levels (or two in the case of helium). These noble gas structures are thought of as being in some way a "desirable" thing for an atom to have. You may well have been left with the strong impression that when other atoms react, they try to achieve noble gas structures. As well as achieving noble gas structures by transferring electrons from one atom to another as in ionic bonding, it is also possible for atoms to reach these stable structures by sharing electrons to give covalent bonds.
Chemical Bonds
Chemical bonds are the attractive forces that hold atoms together in the form of compounds. They are formed when electrons are shared between two atoms. There are 3 types of bonds...covalent bonds, polar covalent bonds and ionic bonds. The simplest example of bonding can be demonstrated by the H2 molecule. We can see from the periodic table that each hydrogen atom has a single electron. If 2 hydrogen atoms come together to form a bond, then each hydrogen atom effectively has a share in both electrons and thus each resembles a noble gas and is more stable. The 2 electrons that are shared can be represented either by 2 dots or a single dash between the atoms.

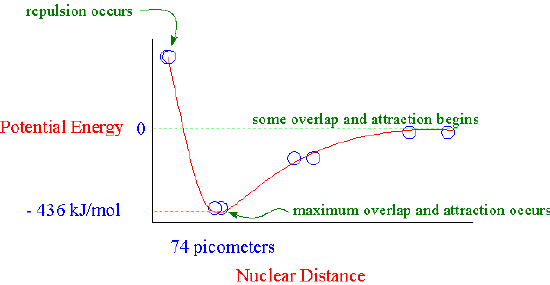
Valence bond theory describes a chemical bond as the overlap of atomic orbitals. In the case of the hydrogen molecule, the 1s orbital of one hydrogen atom overlaps with the 1s orbital of the second hydrogen atom to form a molecular orbital called a sigma bond. Attraction increases as the distance between the atoms gets closer but nuclear-nuclear repulsion becomes important if the atoms approach too close.
There are 3 methods of showing the formulas of molecules. Molecular formulas show only the types and numbers of atoms in the molecule. Structural formulas show the atoms in their correct placement in the molecule and allow for distinguishing isomers. Electron-dot formulas are similar to structural formulas but also include all of the non-bonding outer electrons. Knowledge of electron placement allows us to understand not only the shape of molecules but their chemical character. If we understand the chemical character of a molecule, we can predict how it will react with other molecules without having to blindly memorize reactions.
Covalent bonds and Lewis structures
Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of electrons in the molecule. Lewis structures can also be useful in predicting molecular geometry in conjunction with hybrid orbitals. A compound may have multiple resonance forms that are also all correct Lewis structures. This section will discuss the rules for correctly writing Lewis structures.
Before we begin, there are a few things to know. An electron is represented as a dot. A bond, which is made up of 2 shared electrons, is represented by two dots between the bonded atoms or a line. Double bonds and triple bonds are represented as two and three lines (pairs of electrons), respectively. Lone pairs on the outer rims of an atom are represented as two dots. The electrons represented in a Lewis structure are the outer-shell electrons, which are called valence electrons. This is because they are the ones involved in chemical reactions.
While alkali metals (such as sodium and potassium), alkaline earth metals (such as magnesium and calcium), and halogens (such as fluorine and chlorine) often form ions in order to achieve a full octet, the principle elements of organic chemistry - carbon, hydrogen, nitrogen, and oxygen - instead tend to fill their second shell orbitals by sharing electrons with other atoms, forming what we call covalent bonds. Consider the simplest case of hydrogen gas. An isolated hydrogen atom has only one electron, located in the 1s orbital. If two hydrogen atoms come close enough so that their respective 1s orbitals overlap, the two electrons can be shared between the two nuclei, and a covalently bonded H2 molecule is formed. In the ‘Lewis method’, each pair of electrons that is shared between two atoms is drawn as a single line, designating a single covalent bond.
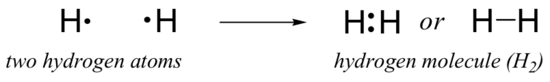
Hydrogen represents is a special case, of course – a hydrogen atom cannot fulfill the octet rule; it needs only two electrons to have a full shell (you could think of this as the ‘doublet rule’ for hydrogen).
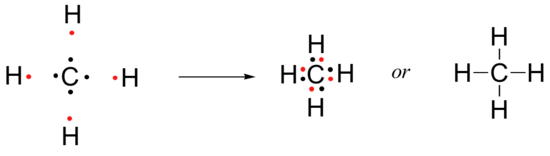
One of the simplest organic molecules is methane with the molecular formula CH4. Methane is the ‘natural gas’ burned in home furnaces and hot water heaters, as well as in electrical power generating plants. To illustrate the covalent bonding in methane using the Lewis method, we first must recognize that, although a carbon atom has a total of six electrons, the two electrons in the inner 1s orbital do not participate in bonding interactions. It is the other four- those in the 2s and 2p orbitals - that form covalent bonds with other atoms. Only the partially occupied, highest energy shell of orbitals - in this case the 2s and 2p orbitals - can overlap with orbitals on other atoms to form covalent bonds. Electrons in these orbitals are termed ‘valence electrons’.

A carbon atom, then, has four valence electrons with which to form covalent bonds. In order to fulfill the octet rule and increase the occupancy of its second shell to eight electrons, it must participate in four electron-sharing interactions - in other words, it must form four covalent bonds. In a methane molecule, the central carbon atom shares its four valence electrons with four hydrogen atoms, thus forming four bonds and fulfilling the octet rule (for the carbon) and the ‘doublet rule’ (for each of the hydrogens).
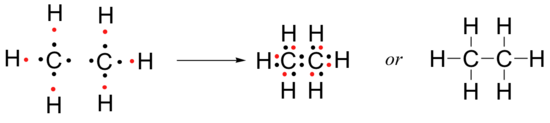
The next relatively simple organic molecule to consider is ethane, which has the molecular formula C2H6. If we draw each atom with its valence electron(s) separately, we can see that the octet/doublet rule can be fulfilled for all of them by forming one carbon-carbon bond and six carbon-hydrogen bonds.
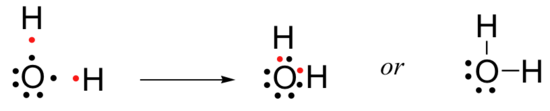
The same approach can be used for molecules in which there is no carbon atom. In a water molecule, two of the six valence electrons on the oxygen atom are used to form bonds to hydrogen atoms, while the remaining four are non-bonding ‘lone pairs’.
What about multiple bonds? The molecular formula for ethene (also known as ethylene, a compound found in fruits, such as apples, that signals them to ripen) is C2H4. Arranging the atoms and surrounding them with their valence electrons, you can see that the octet/doublet rule can be fulfilled for all atoms only if the two carbons share two pairs of electrons between them - in other words, only if a double bond is formed.

Because a hydrogen atom has only a 1s orbital to work with, it cannot form more than one single bond, otherwise it would exceed its doublet rule.
Following this pattern, the triple bond in ethyne molecular formula C2H2, (also known as acetylene, the fuel used in welding torches), is formed when the two carbon atoms share three pairs of electrons between them.
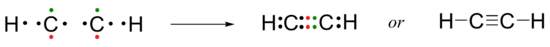
What about ions? The hydroxide ion, OH-, is drawn simply by showing the oxygen atom with its six valence electrons, then adding one more electron to account for the negative charge. Now the oxygen has three non-bonding lone pairs, and can only form one bond to a hydrogen. (Bear in mind that this is merely a description of the thought process going into drawing a Lewis structure, and is not meant to describe any actual chemical process).

To draw a Lewis structure of the hydronium ion, H3O+, you again start with the oxygen atom with its six valence electrons, then take one away to account for the positive charge (there is now one more proton than there are electrons). The oxygen now can form bonds to three hydrogen atoms.
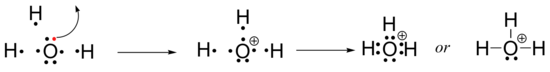
Exercise 1.5.2
Draw a Lewis structure for the ammonium ion, NH4+.
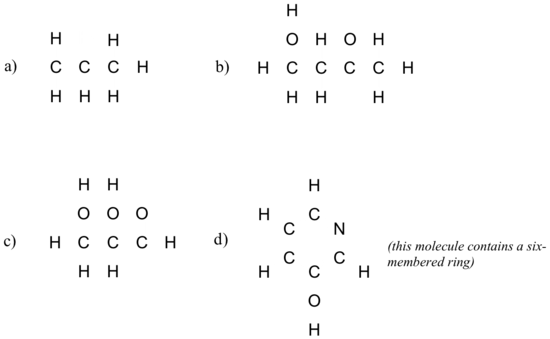
Exercise 1.5.3
Draw Lewis structures for neutral molecules with the connectivity shown below. Show all lone pairs. The octet rule should be fulfilled for all atoms.
Formal charges
Consider the Lewis structure of methanol, CH3OH (methanol is the so-called ‘wood alcohol’ that unscrupulous bootleggers sometimes sold during the Prohibition days, often causing the people who drank it to go blind). Just like in a water molecule, the oxygen atom in a methanol molecule has two non-bonding lone pairs of electrons. And just like a water molecule can be protonated to form the H3O+ cation, a methanol molecule can be protonated to form the CH3OH2+ cation.
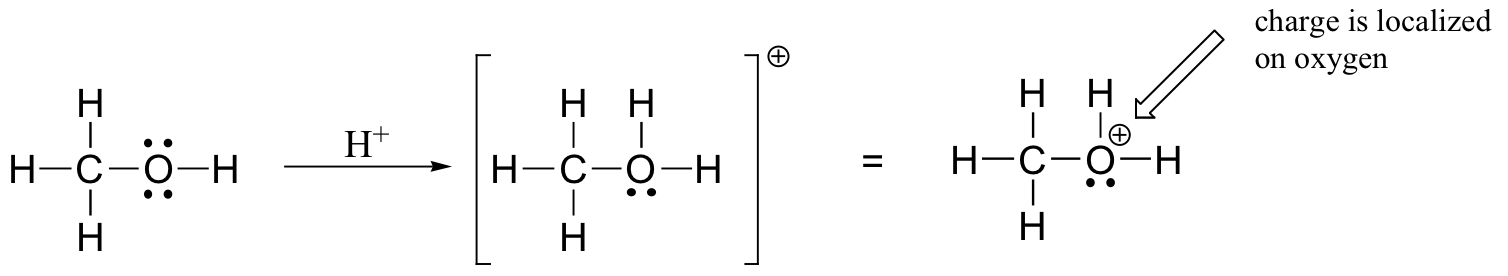
This polyatomic cation, as you can see, has an overall charge of +1. But we can be more specific than that - we can also state that the positive charge is located specifically on the oxygen atom, rather than on the carbon or any of the hydrogens. When a charge can be located on a particular atom in a polyatomic ion, this atom is said to have a ‘formal charge’. Figuring out the formal charge on different atoms of a polyatomic ion is a straightforward process - it’s simply a matter of adding up valence electrons. Remember that an oxygen atom needs six valence electrons (in addition to the two electrons in the non-valence 1s orbital) to completely balance the charge of the eight protons in its nucleus. Let’s figure out how many electrons the oxygen atom in our CH3OH2+ ion ‘owns’.
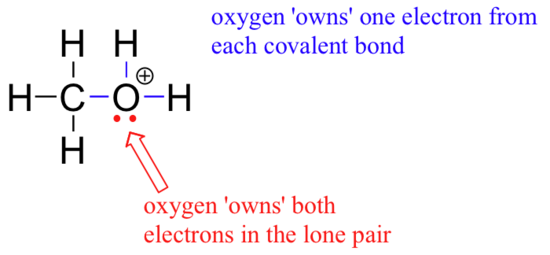
First, we see that there is one lone pair of electrons that the oxygen is not sharing with any other atom - thus it 'owns' both of these electrons. In addition, the oxygen atom is sharing one pair of electrons each with three other atoms - since these electrons are shared, we decide that oxygen 'owns' one electron from each pair, meaning that it owns three bonding electrons. In total then, the oxygen owns five valence electrons: two non-bonding and three bonding. This is one short of the six valence electrons needed to achieve neutrality - thus the oxygen atom has a formal charge of +1.
What is the formal charge, if any, on the other atoms? The carbon needs to ‘own’ four valence electrons (once again, in addition to the two electrons in its 1s orbital) in order to balance the six protons in its nucleus. Because the carbon atom has four single bonds in this structure, and no lone pairs, it does indeed own four electrons (remember that it owns one of each pair of electrons that it shares in a covalent bond). So the carbon has a formal charge of zero.
Each hydrogen needs to own only one electron to balance the charge of its single proton. Indeed, in the CH3OH2+ structure, each hydrogen atom has one single bond, meaning that each one owns one electron. Thus all of the hydrogen atoms have formal charges of zero. Notice that, as you would expect, the sum of the formal charges of all the atoms in the ion equals the total charge on the ion - this will always be true for every example you encounter, and is a good way to check to make sure you are figuring individual formal charges correctly.
An abbreviated formula for determining formal charges can be expressed as follows:
formal charge = (# valence electrons) - (# non-bonding electrons) - ( ½ # bonding electrons)
When drawing structures, it is very important to show all non-zero formal charges, being clear about where the charges are located. When all non-zero formal charges are shown in the structure, the overall charge on an ion does not need to be indicated - that information is obvious from the sum of the formal charges.
At this point, thinking back to what you learned in general chemistry, you are probably asking "What about dipoles? Doesn’t an oxygen atom in an O-H bond ‘own’ more of the electron density than the hydrogen, because of its greater electronegativity?" You are absolutely correct, and we will be reviewing the concept of dipoles later on. For the purpose of calculating formal charges, however, dipoles don’t matter - we always consider the two electrons in a bond to be shared equally, even if that is not an accurate reflection of chemical reality. Formal charges are just that - a formality, a method of electron book-keeping that is tied into the Lewis system for drawing the structures of organic compounds and ions. Later, however, we will see how the concept can help us to visualize how organic molecules react.
Exercise 1.5.4
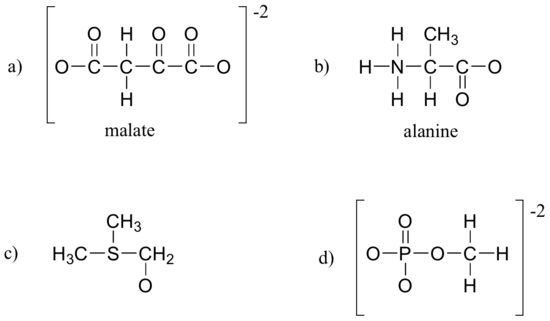
Label all non-zero formal charges on the molecules/ions below. All atoms have a full octet of electrons (lone pairs are not shown).
Hybridization of Atomic Orbitals and the Shape of Molecules
If the four hydrogen atoms in a methane molecule, CH4, were bound to the three 2p orbitals and the 2s orbital of the carbon atom, the H-C-H bond angles would be 90º for 3 of the hydrogen atoms and the 4th hydrogen atom would be at 135º from the others. Experimental evidence has shown that the bond angles in methane are not arranged that way but are 109.5º giving the overall shape of a tetrahedron. The tetrahedral structure makes much more sense in that hydrogen atoms would naturally repel each other due to their negative electron clouds and form this shape. If you think electron-electron repulsion isn't significant, try walking through a wall! There is plenty of space for your nuclei to pass through the nuclei of the wall material but ouch, it just doesn't work that way.
Experimental evidence has also shown that the H-N-H bond angles in ammonia (NH3) are 107º and the H-O-H bond angles in water are 105º. It is clear from these bond angles that the non-bonding pairs of electrons occupy a reasonable amount of space and are pushing the hydrogen atoms closer together compared to the angles found in methane.
The valence shell electron-pair repulsion model (VSEPR) was devised to account for these molecular shapes. In this model, atoms and pairs of electrons will be arranged to minimize the repulsion of these atoms and pairs of electrons. Since the non-bonded electron pairs are held somewhat closer to the nucleus than the attached hydrogen atoms, they tend to crowd the hydrogen atoms. Thus ammonia exists as a distorted tetrahedron (trigonal pyramidal) rather than a trigonal plane and water also exists as a distorted tetrahedron (bent) rather than a linear molecule with the hydrogen atoms at a 180º bond angle.
This concept proposes that since the attached groups are not at the angles of the p orbitals and their atomic orbitals would not have maximum overlap (to form strong bonds) the s and p orbitals will be hybridized to match the bond angles of the attached groups.
The number of these new hybrid orbitals must be equal to the numbers of atoms and non-bonded electron pairs surrounding the central atom!
This valence shell repulsion model can be illustrated at home with a very fun experiment!
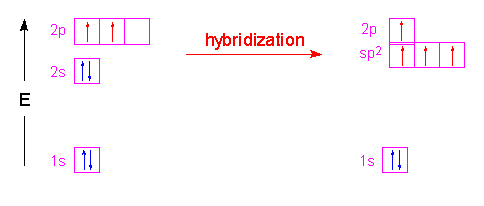
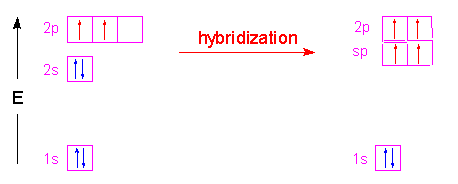
Example 1.5.5: Methane
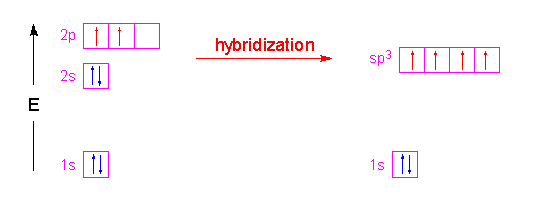
In the case of methane, the three 2p orbitals of the carbon atom are combined with its 2s orbital to form four new orbitals called sp3 hybrid orbitals. The name is simply a tally of all the orbitals that were blended together to form these new hybrid orbitals. Four hybrid orbitals were required since there are four atoms attached to the central carbon atom. These new orbitals will have an energy slightly above the 2s orbital and below the 2p orbitals as shown in the following illustration. Notice that no change occurred with the 1s orbital.
These hybrid orbitals have 75% p-character and 25% s-character which gives them a shape that is shorter and fatter than a p-orbital. The new shape looks a little like...
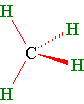
A stick and wedge drawing of methane shows the tetrahedral angles. The wedge is coming out of the paper and the dashed line is going behind the paper. The solid lines are in the plane of the paper.
A space-filling model of methane would look like...

Example 1.5.6: Ammonia
In the case of ammonia, the three 2p orbitals of the nitrogen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. We need a hybrid orbital for each atom and the pair of non-bonding electrons. Ammonia has three hydrogen atoms and one non-bonded pair of electrons when we draw the electron-dot formula. In order to determine the hybridization of an atom, you must first draw the electron-dot formula.

A stick and wedge drawing of ammonia showing the non-bonding electrons in a probability area for the hybrid orbital...
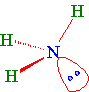
A space-filling model of ammonia would look like...(Note the non-bonded electron pair is not shown in this model.)

Example 1.5.7: Water
In the case of water, the three 2p orbitals of the oxygen atom are combined with the 2s orbital to form four sp3 hybrid orbitals. The two non-bonded electron pairs will occupy hybrid orbitals. Again we need a hybrid orbital for each atom and each pair of non-bonding electrons. Water has two hydrogen atoms and two non-bonded pairs of electrons when we draw the electron-dot formula.

A stick and wedge drawing of water showing the non-bonding electron pairs in probability areas for the hybrid orbital...

A space-filling model of water would look like...(Note the non-bonded electron pairs are not shown in this model.)
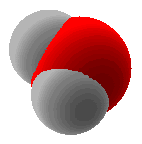
Example 1.5.8: Boron Trifluoride
Now let's look at something a bit different. In the boron trifluoride molecule, only three groups are arranged around the central boron atom. In this case, the 2s orbital is combined with only two of the 2p orbitals (since we only need three hybrid orbitals for the three groups...thinking of groups as atoms and non-bonding pairs) forming three hybrid orbitals called sp2 hybrid orbitals. The other p-orbital remains non-hybridized and is at right angles to the trigonal planar arrangement of the hybrid orbitals. The trigonal planar arrangement has bond angles of 120º.

In the following stick model, the empty p orbital is shown as the probability area...one end shaded blue and the other is white...there are no electrons in this orbital!

A space-filling model of boron trifluoride would look like...

Example 1.5.9: Beryllium Dichloride
Finally let's look at beryllium dichloride. Since only two groups are attached to beryllium, we only will have two hybrid orbitals. In this case, the 2s orbital is combined with only one of the 2p orbitals to yield two sp hybrid orbitals. The two hybrid orbitals will be arranged as far apart as possible from each other with the result being a linear arrangement. The two non-hybridized p-orbitals stay in their respective positions (at right angles to each other) and perpendicular to the linear molecule.
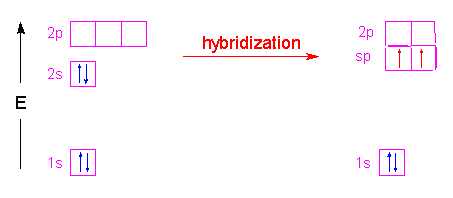
In the following stick model, the empty p orbitals are shown as the probability areas...one green and one blue.
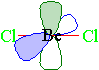
A space-filling model of beryllium dichloride would look like...

Summary of Hybridization
In the following summary, groups are considered to be atoms and/or pairs of electrons and hybrid orbitals are the red lines and wedges. When the octet of an element is exceeded, then hybridization will involve d-orbitals. Non-hybridized p-orbitals are shown as probability areas in blue and green for sp hybridization and blue for sp2 hybridization. A single electron as found in a radical would occupy an non-hybridized p-orbital.
| Number of Groups Attached to a Central Atom | Description and 3-Dimensional Shape |
|
Two Groups sp |
 |
|
Three Groups sp2 |
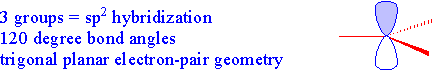 |
|
Four Groups sp3 |
 |
Hybridization Involving Multiple Bonds
Only a maximum of two electrons can occupy any orbital whether it is an atomic orbital or a molecular orbital due to electron-electron repulsion. When we draw a double or a triple-bond between two atoms, we imply that either four or six electrons are directly between these two atoms. Since this is impossible, we must have these extra electrons off to the side in what we refer to as pi bonds. Therefore, all multiple bonds are composed of two different kinds of molecular bonds called pi bonds and sigma bonds.
- The sigma (σ) bond is defined as the linear overlap of atomic orbitals in which two electrons are directly between the two bonded nuclei.
- Pi (π) bonds are defined as the parallel overlap of p-orbitals. A double bond has one sigma bond and one pi bond. A triple bond thus consists of a sigma bond and two pi bonds with the pi bonds in different planes.
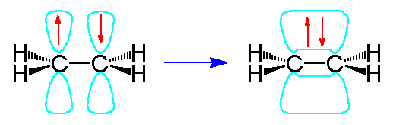
Example 1.5.10: Ethene
In the ethene molecule, C2H4, both carbon atoms will be sp2 hybridized and have one unpaired electron in a non-hybridized p-orbital.
These p-orbitals will undergo parallel overlap and form one pi bond with bean-shaped probability areas above and below the plane of the six atoms. This pair of bean-shaped probability areas constitutes one pi bond and the pair of electrons in this bond can be found in either bean-shaped area.
The 3-dimensional model of ethene is planar with H-C-H and H-C-C bond angles of 120º...the pi bond is not shown in this picture.
Example 1.5.11: Acetylene
Now let's look at acetylene, C2H2. Both carbon atoms will be sp hybridized and have one electron in each of two non-hybridized p-orbitals.
These p orbitals will undergo parallel overlap to form two pi bonds at right angles to each other.
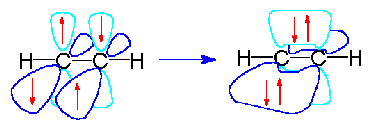
The 3-dimensional model of acetylene is therefore linear...the pi bonds are not shown in this picture.

The Relationship between Bond Order and Bond Energy
Triple bonds between like atoms are shorter than double bonds, and because more energy is required to completely break all three bonds than to completely break two, a triple bond is also stronger than a double bond. Similarly, double bonds between like atoms are stronger and shorter than single bonds. Bonds of the same order between different atoms show a wide range of bond energies, however. Table 1.5.1 lists the average values for some commonly encountered bonds. Although the values shown vary widely, we can observe four trends:
| Single Bonds | Multiple Bonds | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| H–H | 432 | C–C | 346 | N–N | ≈167 | O–O | ≈142 | F–F | 155 | C=C | 602 |
| H–C | 411 | C–Si | 318 | N–O | 201 | O–F | 190 | F–Cl | 249 | C≡C | 835 |
| H–Si | 318 | C–N | 305 | N–F | 283 | O–Cl | 218 | F–Br | 249 | C=N | 615 |
| H–N | 386 | C–O | 358 | N–Cl | 313 | O–Br | 201 | F–I | 278 | C≡N | 887 |
| H–P | ≈322 | C–S | 272 | N–Br | 243 | O–I | 201 | Cl–Cl | 240 | C=O | 749 |
| H–O | 459 | C–F | 485 | P–P | 201 | S–S | 226 | Cl–Br | 216 | C≡O | 1072 |
| H–S | 363 | C–Cl | 327 | S–F | 284 | Cl–I | 208 | N=N | 418 | ||
| H–F | 565 | C–Br | 285 | S–Cl | 255 | Br–Br | 190 | N≡N | 942 | ||
| H–Cl | 428 | C–I | 213 | S–Br | 218 | Br–I | 175 | N=O | 607 | ||
| H–Br | 362 | Si–Si | 222 | I–I | 149 | O=O | 494 | ||||
| H–I | 295 | Si–O | 452 | S=O | 532 | ||||||
|
Source: Data from J. E. Huheey, E. A. Keiter, and R. L. Keiter, Inorganic Chemistry, 4th ed. (1993). |
|||||||||||
- Bonds between hydrogen and atoms in the same column of the periodic table decrease in strength as we go down the column. Thus an H–F bond is stronger than an H–I bond, H–C is stronger than H–Si, H–N is stronger than H–P, H–O is stronger than H–S, and so forth. The reason for this is that the region of space in which electrons are shared between two atoms becomes proportionally smaller as one of the atoms becomes larger (Figure 1.5.1a)
- Bonds between like atoms usually become weaker as we go down a column (important exceptions are noted later). For example, the C–C single bond is stronger than the Si–Si single bond, which is stronger than the Ge–Ge bond, and so forth. As two bonded atoms become larger, the region between them occupied by bonding electrons becomes proportionally smaller. (Figure 1.5.1b) Noteworthy exceptions are single bonds between the period 2 atoms of groups 15, 16, and 17 (i.e., N, O, F), which are unusually weak compared with single bonds between their larger congeners. It is likely that the N–N, O–O, and F–F single bonds are weaker than might be expected due to strong repulsive interactions between lone pairs of electrons on adjacent atoms. The trend in bond energies for the halogens is therefore \[Cl–Cl > Br–Br > F–F > I–I\] Similar effects are also seen for the O–O versus S–S and for N–N versus P–P single bonds.
Bonds between hydrogen and atoms in a given column in the periodic table are weaker down the column; bonds between like atoms usually become weaker down a column.
- Because elements in periods 3 and 4 rarely form multiple bonds with themselves, their multiple bond energies are not accurately known. Nonetheless, they are presumed to be significantly weaker than multiple bonds between lighter atoms of the same families. Compounds containing an Si=Si double bond, for example, have only recently been prepared, whereas compounds containing C=C double bonds are one of the best-studied and most important classes of organic compounds.
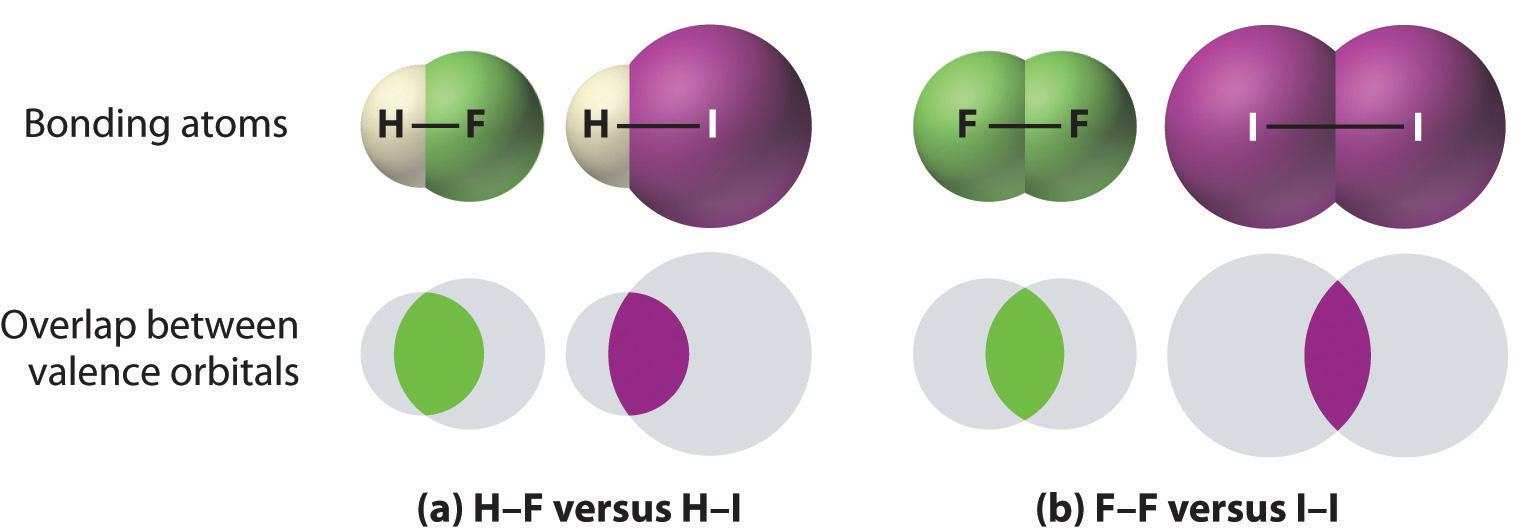
Figure 1.5.1 The Strength of Covalent Bonds Depends on the Overlap between the Valence Orbitals of the Bonded Atoms. The relative sizes of the region of space in which electrons are shared between (a) a hydrogen atom and lighter (smaller) vs. heavier (larger) atoms in the same periodic group; and (b) two lighter versus two heavier atoms in the same group. Although the absolute amount of shared space increases in both cases on going from a light to a heavy atom, the amount of space relative to the size of the bonded atom decreases; that is, the percentage of total orbital volume decreases with increasing size. Hence the strength of the bond decreases.
- Multiple bonds between carbon, oxygen, or nitrogen and a period 3 element such as phosphorus or sulfur tend to be unusually strong. In fact, multiple bonds of this type dominate the chemistry of the period 3 elements of groups 15 and 16. Multiple bonds to phosphorus or sulfur occur as a result of d-orbital interactions, as we discussed for the SO42− ion in Section 8.6. In contrast, silicon in group 14 has little tendency to form discrete silicon–oxygen double bonds. Consequently, SiO2 has a three-dimensional network structure in which each silicon atom forms four Si–O single bonds, which makes the physical and chemical properties of SiO2 very different from those of CO2.
Note
Bond strengths increase as bond order increases, while bond distances decrease.
|
Bond |
(kJ/mol) |
|
C-F |
485 |
|
C-Cl |
328 |
|
C-Br |
276 |
|
C-I |
240 |
|
C-C |
348 |
|
C-N |
293 |
|
C-O |
358 |
|
C-F |
485 |
|
C-C |
348 |
|
C=C |
614 |
|
C=C |
839 |
Exercises
wiki.page("Homework_Exercises/Exercises:_Organic_Chemistry/Exercises:_McMurry/01:_Structure_and_Bonding", "1.5 Exercises")
Contributors
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)

