3.3.0: Bond Types
- Page ID
- 210700
Learning Objectives
- Explain the formation of cations, anions, and ionic compounds
- Predict the charge of common metallic and nonmetallic elements, and write their electron configurations
- Describe the formation of covalent bonds
- Define electronegativity and assess the polarity of covalent bonds
Video \(\PageIndex{1}\): Why do atoms bond?
Video \(\PageIndex{2}\): And how do atoms bond?
As you have learned, ions are atoms or molecules bearing an electrical charge. A cation (a positive ion) forms when a neutral atom loses one or more electrons from its valence shell, and an anion (a negative ion) forms when a neutral atom gains one or more electrons in its valence shell.
Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. The properties of ionic compounds shed some light on the nature of ionic bonds. Ionic solids exhibit a crystalline structure and tend to be rigid and brittle; they also tend to have high melting and boiling points, which suggests that ionic bonds are very strong. Ionic solids are also poor conductors of electricity for the same reason—the strength of ionic bonds prevents ions from moving freely in the solid state. Most ionic solids, however, dissolve readily in water. Once dissolved or melted, ionic compounds are excellent conductors of electricity and heat because the ions can move about freely.
Neutral atoms and their associated ions have very different physical and chemical properties. Sodium atoms form sodium metal, a soft, silvery-white metal that burns vigorously in air and reacts explosively with water. Chlorine atoms form chlorine gas, Cl2, a yellow-green gas that is extremely corrosive to most metals and very poisonous to animals and plants. The vigorous reaction between the elements sodium and chlorine forms the white, crystalline compound sodium chloride, common table salt, which contains sodium cations and chloride anions (Figure Figure \(\PageIndex{1}\)). The compound composed of these ions exhibits properties entirely different from the properties of the elements sodium and chlorine. Chlorine is poisonous, but sodium chloride is essential to life; sodium atoms react vigorously with water, but sodium chloride simply dissolves in water.

Figure \(\PageIndex{1}\): (a) Sodium is a soft metal that must be stored in mineral oil to prevent reaction with air or water. (b) Chlorine is a pale yellow-green gas. (c) When combined, they form white crystals of sodium chloride (table salt). (credit a: modification of work by “Jurii”/Wikimedia Commons)
The Formation of Ionic Compounds
Binary ionic compounds are composed of just two elements: a metal (which forms the cations) and a nonmetal (which forms the anions). For example, NaCl is a binary ionic compound. We can think about the formation of such compounds in terms of the periodic properties of the elements. Many metallic elements have relatively low ionization potentials and lose electrons easily. These elements lie to the left in a period or near the bottom of a group on the periodic table. Nonmetal atoms have relatively high electron affinities and thus readily gain electrons lost by metal atoms, thereby filling their valence shells. Nonmetallic elements are found in the upper-right corner of the periodic table.
As all substances must be electrically neutral, the total number of positive charges on the cations of an ionic compound must equal the total number of negative charges on its anions. The formula of an ionic compound represents the simplest ratio of the numbers of ions necessary to give identical numbers of positive and negative charges. For example, the formula for aluminum oxide, Al2O3, indicates that this ionic compound contains two aluminum cations, Al3+, for every three oxide anions, O2− [thus, (2 × +3) + (3 × –2) = 0].
It is important to note, however, that the formula for an ionic compound does not represent the physical arrangement of its ions. It is incorrect to refer to a sodium chloride (NaCl) “molecule” because there is not a single ionic bond, per se, between any specific pair of sodium and chloride ions. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of opposite charge. This results in the ions arranging themselves into a tightly bound, three-dimensional lattice structure. Sodium chloride, for example, consists of a regular arrangement of equal numbers of Na+ cations and Cl– anions (Figure Figure \(\PageIndex{2}\)).

Figure \(\PageIndex{2}\): The atoms in sodium chloride (common table salt) are arranged to (a) maximize opposite charges interacting. The smaller spheres represent sodium ions, the larger ones represent chloride ions. In the expanded view (b), the geometry can be seen more clearly. Note that each ion is “bonded” to all of the surrounding ions—six in this case.
The strong electrostatic attraction between Na+ and Cl– ions holds them tightly together in solid NaCl. It requires 769 kJ of energy to dissociate one mole of solid NaCl into separate gaseous Na+ and Cl– ions:
Video \(\PageIndex{3}\): A review of ionic bonds from last term.
Electronic Structures of Cations
When forming a cation, an atom of a main group element tends to lose all of its valence electrons, thus assuming the electronic structure of the noble gas that precedes it in the periodic table. For groups 1 (the alkali metals) and 2 (the alkaline earth metals), the group numbers are equal to the numbers of valence shell electrons and, consequently, to the charges of the cations formed from atoms of these elements when all valence shell electrons are removed. For example, calcium is a group 2 element whose neutral atoms have 20 electrons and a ground state electron configuration of 1s22s22p63s23p64s2. When a Ca atom loses both of its valence electrons, the result is a cation with 18 electrons, a 2+ charge, and an electron configuration of 1s22s22p63s23p6. The Ca2+ ion is therefore isoelectronic with the noble gas Ar.
For groups 12–17, the group numbers exceed the number of valence electrons by 10 (accounting for the possibility of full d subshells in atoms of elements in the fourth and greater periods). Thus, the charge of a cation formed by the loss of all valence electrons is equal to the group number minus 10. For example, aluminum (in group 13) forms 3+ ions (Al3+).
Exceptions to the expected behavior involve elements toward the bottom of the groups. In addition to the expected ions Tl3+, Sn4+, Pb4+, and Bi5+, a partial loss of these atoms’ valence shell electrons can also lead to the formation of Tl+, Sn2+, Pb2+, and Bi3+ ions. The formation of these 1+, 2+, and 3+ cations is ascribed to the inert pair effect, which reflects the relatively low energy of the valence s-electron pair for atoms of the heavy elements of groups 13, 14, and 15. Mercury (group 12) also exhibits an unexpected behavior: it forms a diatomic ion, \(\ce{Hg_2^2+}\) (an ion formed from two mercury atoms, with an Hg-Hg bond), in addition to the expected monatomic ion Hg2+ (formed from only one mercury atom).
Transition and inner transition metal elements behave differently than main group elements. Most transition metal cations have 2+ or 3+ charges that result from the loss of their outermost s electron(s) first, sometimes followed by the loss of one or two d electrons from the next-to-outermost shell. For example, iron (1s22s22p63s23p63d64s2) forms the ion Fe2+ (1s22s22p63s23p63d6) by the loss of the 4s electrons and the ion Fe3+ (1s22s22p63s23p63d5) by the loss of the 4s electrons and one of the 3d electrons. Although the d orbitals of the transition elements are—according to the Aufbau principle—the last to fill when building up electron configurations, the outermost s electrons are the first to be lost when these atoms ionize. When the inner transition metals form ions, they usually have a 3+ charge, resulting from the loss of their outermost s electrons and a d or f electron.
Example \(\PageIndex{1}\): Determining the Electronic Structures of Cations
There are at least 14 elements categorized as “essential trace elements” for the human body. They are called “essential” because they are required for healthy bodily functions, “trace” because they are required only in small amounts, and “elements” in spite of the fact that they are really ions. Two of these essential trace elements, chromium and zinc, are required as Cr3+ and Zn2+. Write the electron configurations of these cations.
Solution
First, write the electron configuration for the neutral atoms:
- Zn: [Ar]3d104s2
- Cr: [Ar]3d54s1
Next, remove electrons from the highest energy orbital. For the transition metals, electrons are removed from the s orbital first and then from the d orbital. For the p-block elements, electrons are removed from the p orbitals and then from the s orbital. Zinc is a member of group 12, so it should have a charge of 2+, and thus loses only the two electrons in its s orbital. Chromium is a transition element and should lose its s electrons and then its d electrons when forming a cation. Thus, we find the following electron configurations of the ions:
- Zn2+: [Ar]3d10
- Cr3+: [Ar]3d3
Exercise \(\PageIndex{1}\)
Potassium and magnesium are required in our diet. Write the electron configurations of the ions expected from these elements.
- Answer
-
K+: [Ar], Mg2+: [Ne]
Electronic Structures of Anions
Most monatomic anions form when a neutral nonmetal atom gains enough electrons to completely fill its outer s and p orbitals, thereby reaching the electron configuration of the next noble gas. Thus, it is simple to determine the charge on such a negative ion: The charge is equal to the number of electrons that must be gained to fill the s and p orbitals of the parent atom. Oxygen, for example, has the electron configuration 1s22s22p4, whereas the oxygen anion has the electron configuration of the noble gas neon (Ne), 1s22s22p6. The two additional electrons required to fill the valence orbitals give the oxide ion the charge of 2– (O2–).
Example \(\PageIndex{2}\): Determining the Electronic Structure of Anions
Selenium and iodine are two essential trace elements that form anions. Write the electron configurations of the anions.
Solution
Se2–: [Ar]3d104s24p6
I–: [Kr]4d105s25p6
Exercise \(\PageIndex{2}\)
Write the electron configurations of a phosphorus atom and its negative ion. Give the charge on the anion.
- Answer
-
P: [Ne]3s23p3
P3–: [Ne]3s23p6
In ionic compounds, electrons are transferred between atoms of different elements to form ions. But this is not the only way that compounds can be formed. Atoms can also make chemical bonds by sharing electrons between each other. Such bonds are called covalent bonds. Covalent bonds are formed between two atoms when both have similar tendencies to attract electrons to themselves (i.e., when both atoms have identical or fairly similar ionization energies and electron affinities). For example, two hydrogen atoms bond covalently to form an H2 molecule; each hydrogen atom in the H2 molecule has two electrons stabilizing it, giving each atom the same number of valence electrons as the noble gas He.
Compounds that contain covalent bonds exhibit different physical properties than ionic compounds. Because the attraction between molecules, which are electrically neutral, is weaker than that between electrically charged ions, covalent compounds generally have much lower melting and boiling points than ionic compounds. In fact, many covalent compounds are liquids or gases at room temperature, and, in their solid states, they are typically much softer than ionic solids. Furthermore, whereas ionic compounds are good conductors of electricity when dissolved in water, most covalent compounds, being electrically neutral, are poor conductors of electricity in any state.
Formation of Covalent Bonds
Video \(\PageIndex{4}\): What are covalent bonds?
Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. For example, the hydrogen molecule, H2, contains a covalent bond between its two hydrogen atoms. Figure \(\PageIndex{3}\) illustrates why this bond is formed. Starting on the far right, we have two separate hydrogen atoms with a particular potential energy, indicated by the red line. Along the x-axis is the distance between the two atoms. As the two atoms approach each other (moving left along the x-axis), their valence orbitals (1s) begin to overlap. The single electrons on each hydrogen atom then interact with both atomic nuclei, occupying the space around both atoms. The strong attraction of each shared electron to both nuclei stabilizes the system, and the potential energy decreases as the bond distance decreases. If the atoms continue to approach each other, the positive charges in the two nuclei begin to repel each other, and the potential energy increases. The bond length is determined by the distance at which the lowest potential energy is achieved.
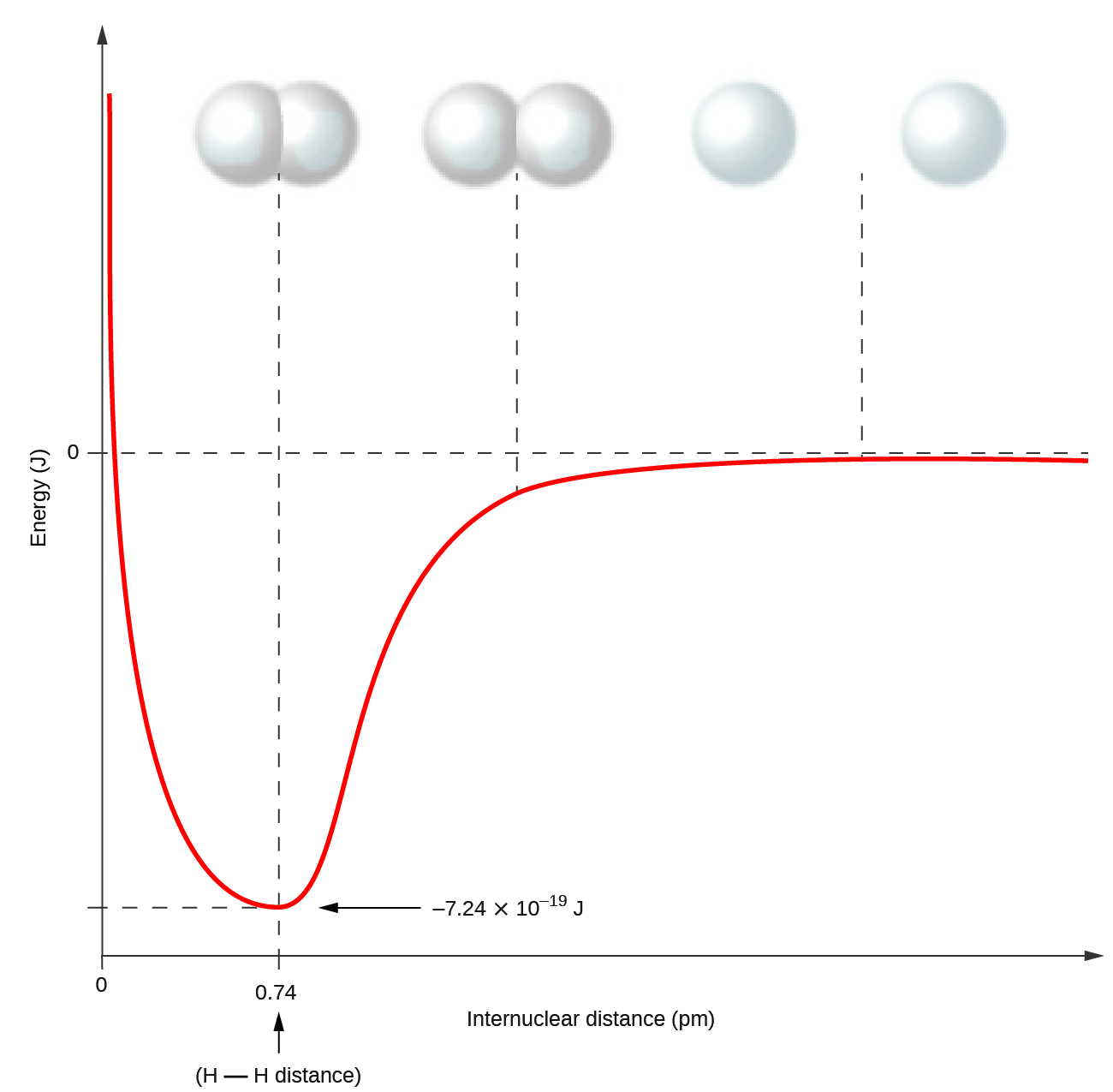
Figure \(\PageIndex{3}\): The potential energy of two separate hydrogen atoms (right) decreases as they approach each other, and the single electrons on each atom are shared to form a covalent bond. The bond length is the internuclear distance at which the lowest potential energy is achieved.
It is essential to remember that energy must be added to break chemical bonds (an endothermic process), whereas forming chemical bonds releases energy (an exothermic process). In the case of H2, the covalent bond is very strong; a large amount of energy, 436 kJ, must be added to break the bonds in one mole of hydrogen molecules and cause the atoms to separate:
\[\ce{H2}(g)⟶\ce{2H}(g)\hspace{20px}ΔH=\mathrm{436\:kJ}\]
Conversely, the same amount of energy is released when one mole of H2 molecules forms from two moles of H atoms:
\[\ce{2H}(g)⟶\ce{H2}(g)\hspace{20px}ΔH=\mathrm{−436\:kJ}\]
Pure vs. Polar Covalent Bonds
If the atoms that form a covalent bond are identical, as in H2, Cl2, and other diatomic molecules, then the electrons in the bond must be shared equally. We refer to this as a pure covalent bond. Electrons shared in pure covalent bonds have an equal probability of being near each nucleus. In the case of Cl2, each atom starts off with seven valence electrons, and each Cl shares one electron with the other, forming one covalent bond:
\[\ce{Cl + Cl⟶Cl2}\]
The total number of electrons around each individual atom consists of six nonbonding electrons and two shared (i.e., bonding) electrons for eight total electrons, matching the number of valence electrons in the noble gas argon. Since the bonding atoms are identical, Cl2 also features a pure covalent bond.
When the atoms linked by a covalent bond are different, the bonding electrons are shared, but no longer equally. Instead, the bonding electrons are more attracted to one atom than the other, giving rise to a shift of electron density toward that atom. This unequal distribution of electrons is known as a polar covalent bond, characterized by a partial positive charge on one atom and a partial negative charge on the other. The atom that attracts the electrons more strongly acquires the partial negative charge and vice versa. For example, the electrons in the H–Cl bond of a hydrogen chloride molecule spend more time near the chlorine atom than near the hydrogen atom. Thus, in an HCl molecule, the chlorine atom carries a partial negative charge and the hydrogen atom has a partial positive charge. Figure \(\PageIndex{4}\) shows the distribution of electrons in the H–Cl bond. Note that the shaded area around Cl is much larger than it is around H. Compare this to Figure \(\PageIndex{3}\), which shows the even distribution of electrons in the H2 nonpolar bond.

Figure \(\PageIndex{4}\): (a) The distribution of electron density in the HCl molecule is uneven. The electron density is greater around the chlorine nucleus. The small, black dots indicate the location of the hydrogen and chlorine nuclei in the molecule. (b) Symbols δ+ and δ– indicate the polarity of the H–Cl bond.
We sometimes designate the positive and negative atoms in a polar covalent bond using a lowercase Greek letter “delta,” δ, with a plus sign or minus sign to indicate whether the atom has a partial positive charge (δ+) or a partial negative charge (δ–). This symbolism is shown for the H–Cl molecule in Figure \(\PageIndex{4b}\).
Summary
Video \(\PageIndex{5}\): A review of atomic bonding.
Atoms gain or lose electrons to form ions with particularly stable electron configurations. The charges of cations formed by the representative metals may be determined readily because, with few exceptions, the electronic structures of these ions have either a noble gas configuration or a completely filled electron shell. The charges of anions formed by the nonmetals may also be readily determined because these ions form when nonmetal atoms gain enough electrons to fill their valence shells.
Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. In pure covalent bonds, the electrons are shared equally. In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other.
Glossary
- inert pair effect
- tendency of heavy atoms to form ions in which their valence s electrons are not lost
- ionic bond
- strong electrostatic force of attraction between cations and anions in an ionic compound
- bond length
- distance between the nuclei of two bonded atoms at which the lowest potential energy is achieved
- covalent bond
- bond formed when electrons are shared between atoms
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).
- Adelaide Clark, Oregon Institute of Technology
- Fuse School, Open Educational Resource free of charge, under a Creative Commons License: Attribution-NonCommercial CC BY-NC (View License Deed: https://creativecommons.org/licenses/by-nc/4.0/)
- Crash Course Chemistry: Crash Course is a division of Complexly and videos are free to stream for educational purposes.
- Sci Show is a division of Complexly and videos are free to stream for educational purposes.
- TED-Ed’s commitment to creating lessons worth sharing is an extension of TED’s mission of spreading great ideas. Within TED-Ed’s growing library of TED-Ed animations, you will find carefully curated educational videos, many of which represent collaborations between talented educators and animators nominated through the TED-Ed website.

