Distillation
- Page ID
- 221845
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Distillation of compounds is a method of separation which exploits the differences in boiling point of a crude mixture. Several methods exist:
Short Path (simple) distillation
- Clamp the distillation apparatus to monkey bars in a well ventilated hood.
- Run water through the condenser such that it fills from the bottom first (when using a Liebing, Allihn or Graham condenser condenser. It is not quite as important when using a Dimroth condenser.)
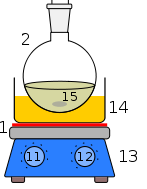
- Place flask (1) in a cool bath of oil, sand or on a heating mantle.
- Place receiving flask 6 away from heat source - sometimes it is advantageous to place it in ice.
- If your distillation setup allows for the inclusion of a thermometer, insert it. Make sure your thermometer goes to temperatures HIGHER than the ones you will be boiling to prevent damage to the thermometer.
- Slowly heat flask 1 and allow the condensation to collect into flask 6.
- Do not heat oil baths above 200 C. Consider Vacuum Distillation for high boiling compounds to prevent injury and facilitate separation.
Fractional Distillation
Fractional Distillation involves the use of a fractionating column.
Vacuum Distillation
When preparing to perform a vacuum distillation, use a pressure-temperature nomograph to determine what the adjusted boiling point of your compound will be.
- The vacuum will quickly remove air from porous boiling chips, so a magnetic stir bar or gas bubbler is required in flask 1 to keep the rate of boiling under control.
- Apply vacuum to the rig via 5 before heating flask 1. Heating the sample first may cause it to bump over into 6 when vacuum is applied.
- Slowly heat flask 1 and allow distillation to occur as you would with a simple distillation. Often times during vacuum distillation the boiling point range is greater that it is during a simple distillation.
- Release the vacuum before attempting to remove any flasks from the rig
Kugelrohr Distillation

 Separation of small amounts of compounds with close boiling points is difficult in a fractional distillation setup and can alternatively be performed via Kugelrohr Distillation. The setup is shown in the image above. Typically high boiling compounds are used and vacuum is applied. To perform a Kugelrohr Distillation:
Separation of small amounts of compounds with close boiling points is difficult in a fractional distillation setup and can alternatively be performed via Kugelrohr Distillation. The setup is shown in the image above. Typically high boiling compounds are used and vacuum is applied. To perform a Kugelrohr Distillation:
- Place compound mixture in small round bottom flask (see figure to right). The amount of compound should be small enough that, when the round bottom is tilted horizontally, it does not spill out.
- Attach Kugelrohr flasks - typically two additional flasks are used.
- Assemble Kugelrohr, placing the round bottom flask inside the heating jacket, keeping the two Kugelrohr flasks outside. Typically there is a "shutter" that can be used to reduce the amount of heat transfer from the heating jacket to the first Kugelrohr flask - this should be closed.
- Induce vacuum, if desired. If your Kugelrohr is so equipped, you can cool the external Kugelrohr flasks with an ice bath. Alternatively, the outer bulb can be cooled by slowly dripping dichloromethane onto the outer bulb with a seperating funnel (fumehood!).
- Heat the Kugelrohr and start rotating motor until distillation occurs. If needed, most Kugelrohr setups will allow you to "insert" the second Kugelrohr flask into the heating jacket - by doing this and reducing the temperature slightly, it is possible to produce a more pure distillation. This can be repeated until you have run out of Kugelrohr flasks.

