Electrophilic Addition of Hydrogen Halides
- Page ID
- 885
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Due to the nature of the \(\pi\) bond, \(\pi\) bonds can act like a nucleophile and undergo addition of electrophiles. The \(\pi\) electrons have a high electron density in the \(\pi\) electron cloud which can be easily polarized (give up the electrons) and can act like a nucleophile (nucleus-loving due to too many electrons; wants to give them away to electrophile). This behavior is very similar to the behavior that the lone pair electrons have on a Lewis base.
Introduction
There are many types of electrophilic addition, but this section will focus on the addition of hydrogen halides.
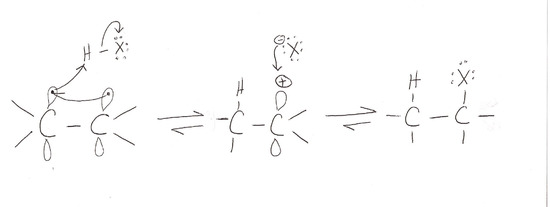
Reaction
The addition of hydrogen halides is one of the easiest electrophilic addition reactions because it uses the simplest electrophile: the proton. Hydrogen halides provide both a electrophile (proton) and a nucleophile (halide). First, the electrophile will attack the double bond and take up a set of \(\pi\) electrons, attaching it to the molecule (1). This is basically the reverse of the last step in the E1 reaction (deprotonation step). The resulting molecule will have a single carbon- carbon bond with a positive charge on one of them (carbocation). The next step is when the nucleophile (halide) bonds to the carbocation, producing a new molecule with both the original hydrogen and halide attached to the organic reactant (2). The second step will only occur if a good nucleophile is used.
Mechanism of Electrophilic Addition of Hydrogen Halide to Ethene
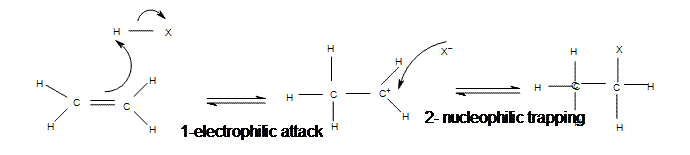
Mechanism of Electrophilic Addition of Hydrogen Halide to Propene
All of the halides (HBr, HCl, HI, HF) can participate in this reaction and add on in the same manner. Although different halides do have different rates of reaction, due to the H-X bond getting weaker as X gets larger (poor overlap of orbitals).
| Name | Rate | Size of "X" | Overlap of Orbitals |
|---|---|---|---|
| HF | slowest | smallest | good |
| HCl | slow | small | okay |
| HBr | fast | large | bad |
| HI | fastest | largest | horrifically bad |
- Gaseous hydrogen halides are used and bubbled through the reactant or the hydrogen halide is added in a solvent such as acetic acid to produce decent amounts of the product. It also helps to do a aqueous work up afterward to get a good yield of product. This reaction is usually carried out at lower temperatures.
Regiochemistry
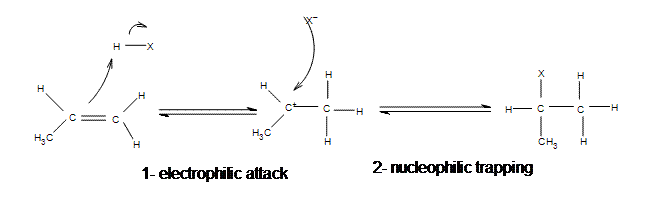
The regiochemistry of this reaction can be explained through Markovnikov's Rule. The basics of this rule states that the proton will add to the less substituted carbon and the halogen goes to the more substituted carbon. This may seem odd that the larger atom is going to the more sterically hindered spot, but this rule is more about the stability of the intermediate. The addition will occur in a way that produces the most stable carbocation after the initial protonation (the intermediate needs to have a stable carbocation). Tertiary and secondary carbocations are the most stable due to hyperconjugation, so the more substituted the carbon is the more stable the carbocation will be.
Examples of regioselective behavior in Electrophilic Addition: Markovnikov's Rule
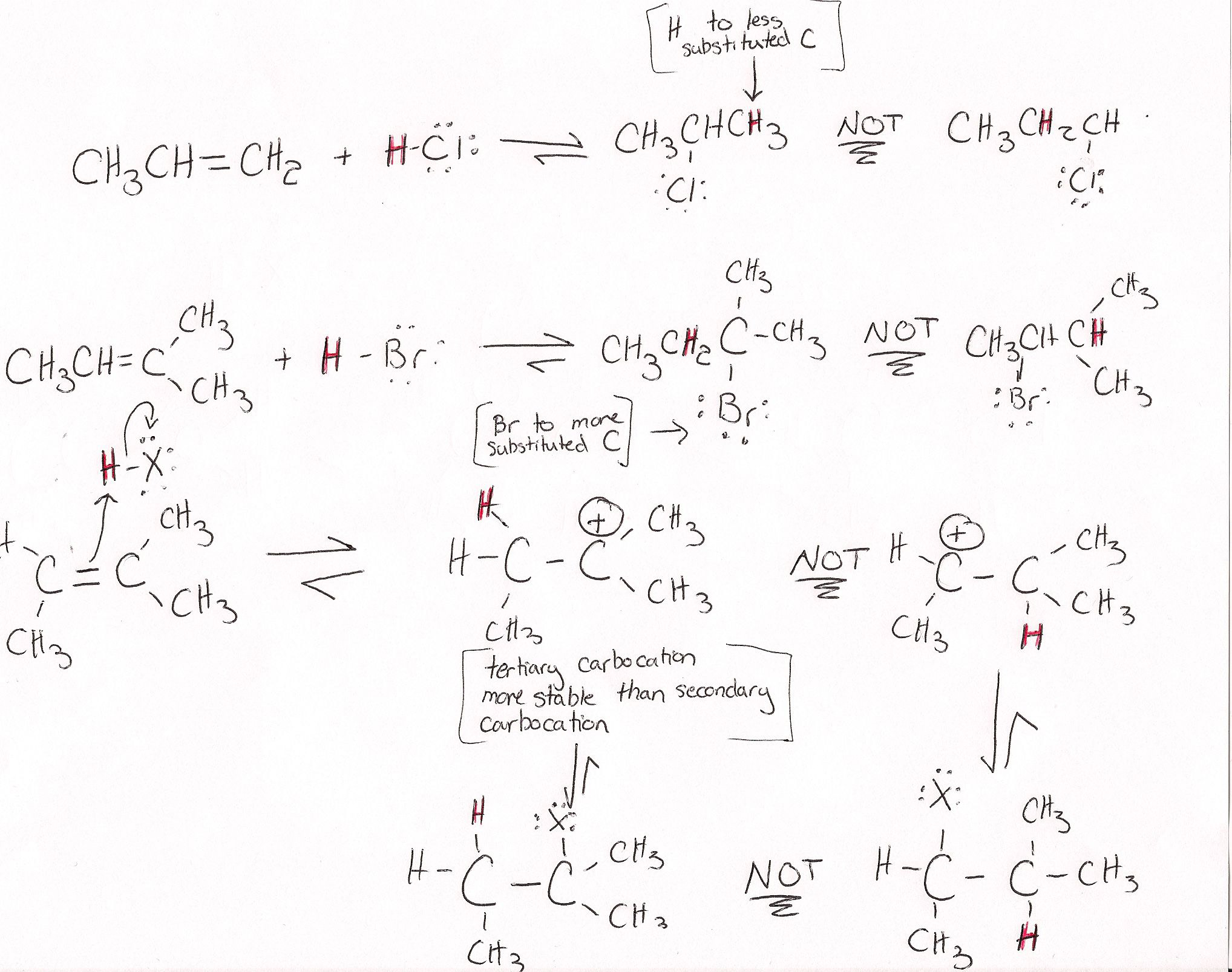
Since the intermediate of this reaction is a carbocation the carbocation wants to be the most stable it can be. In some case this will mean that the carbocation will rearrange. This is very likely when there are no good nucleophiles present to trap the carbocation in a certain position. The rearrangement of the carbocation depends on many things but generally rearrangements will occur when there are no good nucleophiles present such as in a strong acid.
Stereochemistry
The stereochemistry for this reaction is random. The important thing to understand is the regiochemistry and the Markovnikov's Rule (this will come up a lot!).
References
- Vollhardt, K. Peter C., and Neil E. Schore. Organic Chemistry: Structure and Function. New York: W.H. Freeman and Company, 2007.
- Francis A. Coreg, Richard J. Sundberg. Advanced Organic Chemistry: Part B: Reactions and Synthesis. Springer, 2007.
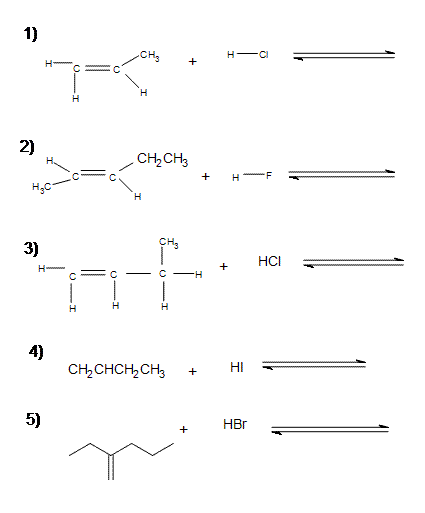
Problems
Write out the following reactions and think about the rate of the reaction:
Contributors
- Ivy Gardner

