All About Water
- Page ID
- 3500
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A chemist's view of the world is not as narrow as one might think! Yes, we start with the atom, and then go on to the rules governing the kinds of structural units that can be made from them. We are taught early on to predict the properties of bulk matter from these geometric arrangements.

And then we come to H2O, and are shocked to find that many of these predictions are way off, and that water (and by implication, life itself) should not even exist on our planet! But we soon learn that this tiny combination of three nuclei and eight electrons possesses special properties that make it unique among the more than 15 million chemical species we presently know. When we stop to ponder the consequences of this, chemistry moves from being an arcane science to a voyage of wonder and pleasure as we learn to relate the microscopic world of the atom to the greater world in which we all live.

The molecule of water
A molecule is an aggregation of atomic nuclei and electrons that is sufficiently stable to possess observable properties — and there are few molecules that are more stable and difficult to decompose than H2O. In water, each hydrogen nucleus is bound to the central oxygen atom by a pair of electrons that are shared between them; chemists call this shared electron pair a covalent chemical bond. In H2O, only two of the six outer-shell electrons of oxygen are used for this purpose, leaving four electrons which are organized into two non-bonding pairs. The four electron pairs surrounding the oxygen tend to arrange themselves as far from each other as possible in order to minimize repulsions between these clouds of negative charge. This would ordinarily result in a tetrahedral geometry in which the angle between electron pairs (and therefore the H-O-H bond angle) is 109.5°. However, because the two non-bonding pairs remain closer to the oxygen atom, these exert a stronger repulsion against the two covalent bonding pairs, effectively pushing the two hydrogen atoms closer together. The result is a distorted tetrahedral arrangement in which the H—O—H angle is 104.5°.
Although the water molecule carries no net electric charge, its eight electrons are not distributed uniformly; there is slightly more negative charge (purple) at the oxygen end of the molecule, and a compensating positive charge (green) at the hydrogen end. The resulting polarity is largely responsible for water's unique properties.
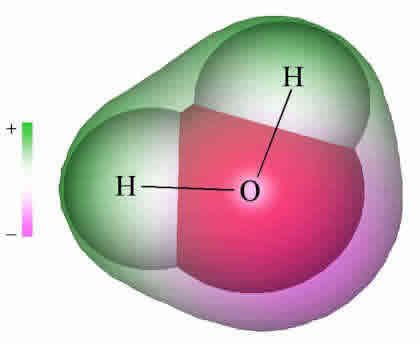
Because molecules are smaller than light waves, they cannot be observed directly, and must be "visualized" by alternative means. This computer-generated image comes from calculations that model the electron distribution in the H2O molecule. The outer envelope shows the effective "surface" of the molecule as defined by the extent of the cloud of negative electric charge created by the eight electrons.
Hydrogen bonding
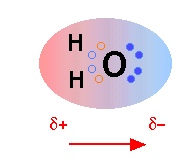
The H2O molecule is electrically neutral, but the positive and negative charges are not distributed uniformly. This is illustrated by the gradation in color in the schematic diagram here. The electronic (negative) charge is concentrated at the oxygen end of the molecule, owing partly to the nonbonding electrons (solid blue circles), and to oxygen's high nuclear charge which exerts stronger attractions on the electrons. This charge displacement constitutes an electric dipole, represented by the arrow at the bottom; you can think of this dipole as the electrical "image" of a water molecule.
As we all learned in school, opposite charges attract, so the partially-positive hydrogen atom on one water molecule is electrostatically attracted to the partially-negative oxygen on a neighboring molecule. This process is called (somewhat misleadingly) hydrogen bonding. Notice that the hydrogen bond (shown by the dashed green line) is somewhat longer than the covalent O—H bond. This means that it is considerably weaker; it is so weak, in fact,that a given hydrogen bond cannot survive for more than a tiny fraction of a second.
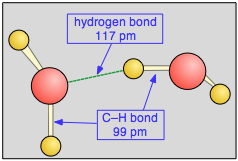
Figure: Hydrogen bond between two water molecules
The anomalous properties of water
Water has long been known to exhibit many physical properties that distinguish it from other small molecules of comparable mass. Chemists refer to these as the "anomalous" properties of water, but they are by no means mysterious; all are entirely predictable consequences of the way the size and nuclear charge of the oxygen atom conspire to distort the electronic charge clouds of the atoms of other elements when these are chemically bonded to the oxygen.
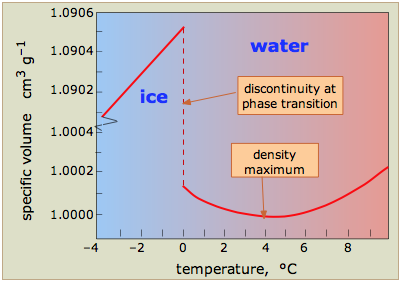
Water is one of the few known substances whose solid form is less dense than the liquid. The plot at the right shows how the volume of water varies with the temperature; the large increase (about 9%) on freezing shows why ice floats on water and why pipes burst when they freeze. The expansion between –4° and 0° is due to the formation of larger hydrogen-bonded aggregates. Above 4°, thermal expansion sets in as vibrations of the O—H bonds becomes more vigorous, tending to shove the molecules farther apart.
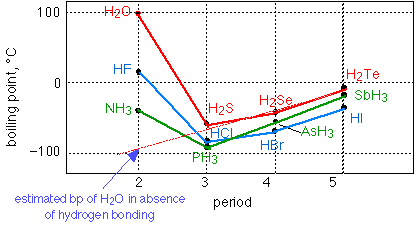
The other widely-cited anomalous property of water is its high boiling point. As this graph shows, a molecule as light as H2O "should" boil at around –90°C; that is, it would exist in the world as a gas rather than a liquid if H-bonding were not present. Notice that H-bonding is also observed with fluorine and nitrogen.
Surface tension and wetting
Have you ever watched an insect walk across the surface of a pond? The water strider takes advantage of the fact that the water surface acts like an elastic film that resists deformation when a small weight is placed on it. (If you are careful, you can also "float" a small paper clip or steel staple on the surface of water in a cup.) This is all due to the surface tension of the water. A molecule within the bulk of a liquid experiences attractions to neighboring molecules in all directions, but since these average out to zero, there is no net force on the molecule. For a molecule that finds itself at the surface, the situation is quite different; it experiences forces only sideways and downward, and this is what creates the stretched-membrane effect.

The distinction between molecules located at the surface and those deep inside is especially prominent in H2O, owing to the strong hydrogen-bonding forces. The difference between the forces experienced by a molecule at the surface and one in the bulk liquid gives rise to the liquid's surface tension.
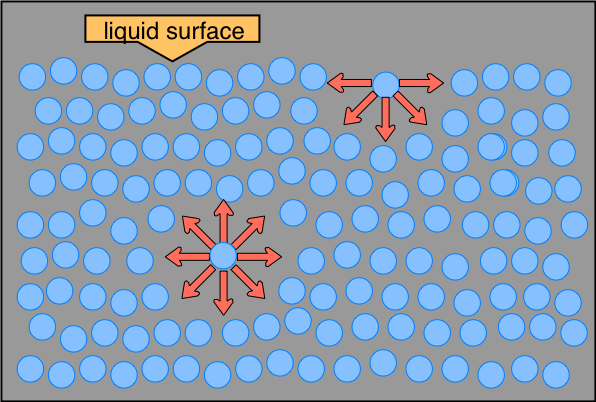
This drawing highlights two H2O molecules, one at the surface, and the other in the bulk of the liquid. The surface molecule is attracted to its neighbors below and to either side, but there are no attractions pointing in the 180° solid angle angle above the surface. As a consequence, a molecule at the surface will tend to be drawn into the bulk of the liquid. But since there must always be some surface, the overall effect is to minimize the surface area of a liquid.

The geometric shape that has the smallest ratio of surface area to volume is the sphere, so very small quantities of liquids tend to form spherical drops. As the drops get bigger, their weight deforms them into the typical tear shape.
Wetting
Take a plastic mixing bowl from your kitchen, and splash some water around in it. You will probably observe that the water does not cover the inside surface uniformly, but remains dispersed into drops. The same effect is seen on a dirty windshield; turning on the wipers simply breaks hundreds of drops into thousands. By contrast, water poured over a clean glass surface will wet it, leaving a uniform film.
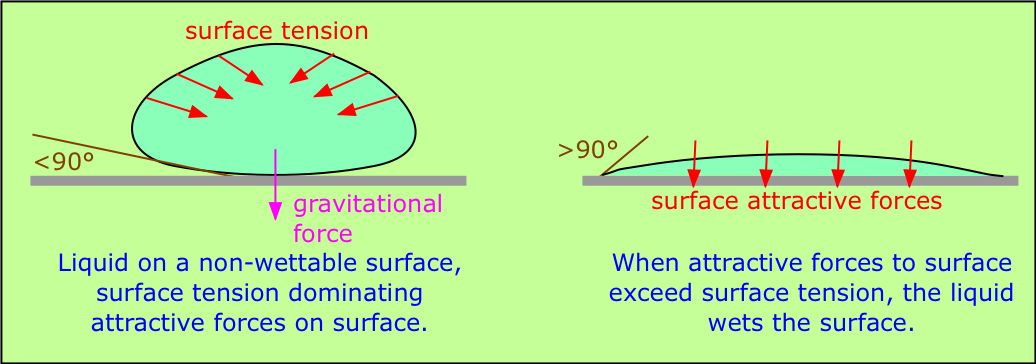
When a liquid is in contact with a solid surface, its behavior depends on the relative magnitudes of the surface tension forces and the attractive forces between the molecules of the liquid and of those comprising the surface. If an H2O molecule is more strongly attracted to its own kind, then surface tension will dominate, increasing the curvature of the interface. This is what happens at the interface between water and a hydrophobic surface such as a plastic mixing bowl or a windshield coated with oily material. A clean glass surface, by contrast, has -OH groups sticking out of it which readily attach to water molecules through hydrogen bonding; this causes the water to spread out evenly over the surface, or to wet it. A liquid will wet a surface if the angle at which it makes contact with the surface is more than 90°. The value of this contact angle can be predicted from the properties of the liquid and solid separately.
If we want water to wet a surface that is not ordinarily wettable, we add a detergent to the water to reduce its surface tension. A detergent is a special kind of molecule in which one end is attracted to H2O molecules but the other end is not, so these ends stick out above the surface and repel each other, cancelling out the surface tension forces due to the water molecules alone.
Water the liquid
The nature of liquid water and how the H2O molecules within it are organized and interact are questions that have attracted the interest of chemists for many years. There is probably no liquid that has received more intensive study, and there is now a huge literature on this subject.
The following facts are well established:
- H2O molecules attract each other through the special type of dipole-dipole interaction known as hydrogen bonding
- a hydrogen-bonded cluster in which four H2Os are located at the corners of an imaginary tetrahedron is an especially favorable (low-potential energy) configuration, but...
- the molecules undergo rapid thermal motions on a time scale of picoseconds (10–12 second), so the lifetime of any specific clustered configuration will be fleetingly brief.

A variety of techniques including infrared absorption, neutron scattering, and nuclear magnetic resonance have been used to probe the microscopic structure of water. The information garnered from these experiments and from theoretical calculations has led to the development of around twenty "models" that attempt to explain the structure and behavior of water. More recently, computer simulations of various kinds have been employed to explore how well these models are able to predict the observed physical properties of water.
This work has led to a gradual refinement of our views about the structure of liquid water, but it has not produced any definitive answer. There are several reasons for this, but the principal one is that the very concept of "structure" (and of water "clusters") depends on both the time frame and volume under consideration. Thus questions of the following kinds are still open:
- How do you distinguish the members of a "cluster" from adjacent molecules that are not in that cluster?
- Since individual hydrogen bonds are continually breaking and re-forming on a picosecond time scale, do water clusters have any meaningful existence over longer periods of time? In other words, clusters are transient, whereas "structure" implies a molecular arrangement that is more enduring. Can we then legitimately use the term "clusters" in describing the structure of water?
- The possible locations of neighboring molecules around a given H2O are limited by energetic and geometric considerations, thus giving rise to a certain amount of "structure" within any small volume element. It is not clear, however, to what extent these structures interact as the size of the volume element is enlarged. And as mentioned above, to what extent are these structures maintained for periods longer than a few picoseconds?
The view first developed in the 1950's that water is a collection of "flickering clusters" of varying sizes (right) has gradually been abandoned as being unable to account for many of the observed properties of the liquid.
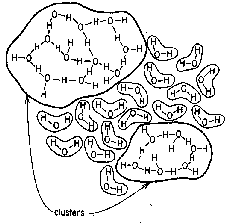
Current views of water structure
The present thinking, influenced greatly by molecular modeling simulations beginning in the 1980s, is that on a very short time scale (less than a picosecond), water is more like a "gel" consisting of a single, huge hydrogen-bonded cluster. On a 10-12-10-9 sec time scale, rotations and other thermal motions cause individual hydrogen bonds to break and re-form in new configurations, inducing ever-changing local discontinuities whose extent and influence depends on the temperature and pressure.
|
Recent work from Richard SayKally's lab shows that the hydrogen bonds in liquid water break and re-form so rapidly (often in distorted configurations) that the liquid can be regarded as a continuous network of hydrogen-bonded molecules. |
This computer-generated nanoscale view of liquid water is from the lab of Gene Stanley of Boston University. The oxygen atoms are red, the hydrogen atoms white |
Local structures and water clusters
It is quite likely that over very small volumes, localized (H2O)n polymeric clusters may have a fleeting existence, and many theoretical calculations have been made showing that some combinations are more stable than others. While this might prolong their lifetimes, it does not appear that they remain intact long enough to detect as directly observable entities in ordinary bulk water at normal pressures.
Theoretical models suggest that the average cluster may encompass as many as 90 H2O molecules at 0°C, so that very cold water can be thought of as a collection of ever-changing ice-like structures. At 70° C, the average cluster size is probably no greater than about 25.
It must be emphasized that no stable clustered unit or arrangement has ever been isolated or identified in pure bulk liquid water. A 2006 report suggests that a simple tetrahedral arrangement is the only long-range structure that persists at time scales of a picosecond or beyond. And a 2007 study suggests that infrared radiation can stabilize clathrate-like clusters for up to several hours.
Water clusters are of considerable interest as models for the study of water and water surfaces, and many articles on them are published every year. Some notable work reported in 2004 extended our view of water to the femtosecond time scale. The principal finding was that 80 percent of the water molecules are bound in chain-like fashion to only two other molecules at room temperature, thus supporting the prevailing view of a dynamically-changing, disordered water structure.
Liquid and solid water
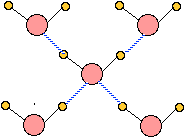 Ice, like all solids, has a well-defined structure; each water molecule is surrounded by four neighboring H2Os. two of these are hydrogen-bonded to the oxygen atom on the central H2O molecule, and each of the two hydrogen atoms is similarly bonded to another neighboring H2O.
Ice, like all solids, has a well-defined structure; each water molecule is surrounded by four neighboring H2Os. two of these are hydrogen-bonded to the oxygen atom on the central H2O molecule, and each of the two hydrogen atoms is similarly bonded to another neighboring H2O.
The hydrogen bonds are represented by the dashed lines in this 2-dimensional schematic diagram. In reality, the four bonds from each O atom point toward the four corners of a tetrahedron centered on the O atom. This basic assembly repeats itself in three dimensions to build the ice crystal.
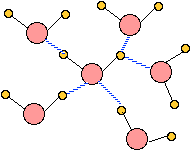 When ice melts to form liquid water, the uniform three-dimensional tetrahedral organization of the solid breaks down as thermal motions disrupt, distort, and occasionally break hydrogen bonds. The methods used to determine the positions of molecules in a solid do not work with liquids, so there is no unambiguous way of determining the detailed structure of water. The illustration here is probably typical of the arrangement of neighbors around any particular H2O molecule, but very little is known about the extent to which an arrangement like this gets propagated to more distant molecules.
When ice melts to form liquid water, the uniform three-dimensional tetrahedral organization of the solid breaks down as thermal motions disrupt, distort, and occasionally break hydrogen bonds. The methods used to determine the positions of molecules in a solid do not work with liquids, so there is no unambiguous way of determining the detailed structure of water. The illustration here is probably typical of the arrangement of neighbors around any particular H2O molecule, but very little is known about the extent to which an arrangement like this gets propagated to more distant molecules.
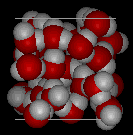
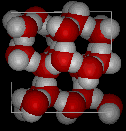 Here are three-dimensional views of a typical local structure of water (left) and ice (right.) Notice the greater openness of the ice structure which is necessary to ensure the strongest degree of hydrogen bonding in a uniform, extended crystal lattice. The more crowded and jumbled arrangement in liquid water can be sustained only by the greater amount thermal energy available above the freezing point.
Here are three-dimensional views of a typical local structure of water (left) and ice (right.) Notice the greater openness of the ice structure which is necessary to ensure the strongest degree of hydrogen bonding in a uniform, extended crystal lattice. The more crowded and jumbled arrangement in liquid water can be sustained only by the greater amount thermal energy available above the freezing point.
 |
The stable arrangement of hydrogen-bonded water molecules in ice gives rise to the beautiful hexagonal symmetry that reveals itself in every snowflake. |
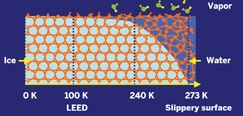 Why is ice slippery?
Why is ice slippery?
At temperatures as low as 200K, the surface of ice is highly disordered and water-like. As the temperature approaches the freezing point, this region of disorder extends farther down from the surface and acts as a lubricant.
The illustration is taken from from an article in the April 7, 2008 issue of C&EN honoring the physical chemist Gabor Somorjai who pioneered modern methods of studying surfaces.
"Pure" water
To a chemist, the term "pure" has meaning only in the context of a particular application or process. The distilled or de-ionized water we use in the laboratory contains dissolved atmospheric gases and occasionally some silica, but their small amounts and relative inertness make these impurities insignificant for most purposes. When water of the highest obtainable purity is required for certain types of exacting measurements, it is commonly filtered, de-ionized, and triple-vacuum distilled. But even this "chemically pure" water is a mixture of isotopic species: there are two stable isotopes of both hydrogen (H1 and H2, the latter often denoted by D) and oxygen (O16 and O18) which give rise to combinations such as H2O18, HDO16, etc., all of which are readily identifiable in the infrared spectra of water vapor. And to top this off, the two hydrogen atoms in water contain protons whose magnetic moments can be parallel or antiparallel, giving rise to ortho- and para-water, respectively. The two forms are normally present in a o/p ratio of 3:1.
The amount of the rare isotopes of oxygen and hydrogen in water varies enough from place to place that it is now possible to determine the age and source of a particular water sample with some precision. These differences are reflected in the H and O isotopic profiles of organisms. Thus the isotopic analysis of human hair can be a useful tool for crime investigations and anthropology research.
It has recently been found (Langmuir 2003, 19, 6851-6856) that freshly distilled water takes a surprisingly long time to equilibrate with the atmosphere, that it undergoes large fluctuations in pH and redox potential, and that these effects are greater when the water is exposed to a magnetic field. The reasons for this behavior are not clear, but one possibility is that dissolved O2 molecules, which are paramagnetic, might be involved.
Drinking water
Our ordinary drinking water, by contrast, is never chemically pure, especially if it has been in contact with sediments. Groundwaters (from springs or wells) always contain ions of calcium and magnesium, and often iron and manganese as well; the positive charges of these ions are balanced by the negative ions carbonate/bicarbonate, and occasionally some chloride and sulfate. Groundwaters in some regions contain unacceptably high concentrations of naturally-occuring toxic elements such as selenium and arsenic.
One might think that rain or snow would be exempt from contamination, but when water vapor condenses out of the atmosphere it always does so on a particle of dust which releases substances into the water, and even the purest air contains carbon dioxide which dissolves to form carbonic acid. Except in highly polluted atmospheres, the impurities picked up by snow and rain are too minute to be of concern.
Various governments have established upper limits on the amounts of contaminants allowable in drinking water; the best known of these are the U.S. EPA Drinking Water Standards.
What kind of water is most healthy to drink?
I am not aware of any evidence indicating that any one type of water (including highly "pure" water) is more beneficial to health than any other, as long as the water is pathogen-free and meets accepted standards such as those mentioned above. For those who are sensitive to residual chlorine or still have concerns, a good activated-carbon filter is usually satisfactory. More extreme measures such as reverse-osmosis or distillation are only justified in demonstrably extreme situations.
 "Pure" rainwater always contains some dissolved carbon dioxide which makes it slightly acidic. When this water comes into contact with sediments, it tends to dissolve them, and in the process becomes alkaline. The pH of drinking water can vary from around 5 to 9, and it has no effect on one's health. The idea that alkaline water is better to drink than acidic water is widely promoted by alternative-health hucksters who market worthless "water ionizer" machines for this purpose. Acidic water is sometimes described by engineers as "aggressive"; this refers to its tendency to corrode metal distribution pipes, but in this sense it is no more active than the hydrochloric acid already present in your gastric fluid!
"Pure" rainwater always contains some dissolved carbon dioxide which makes it slightly acidic. When this water comes into contact with sediments, it tends to dissolve them, and in the process becomes alkaline. The pH of drinking water can vary from around 5 to 9, and it has no effect on one's health. The idea that alkaline water is better to drink than acidic water is widely promoted by alternative-health hucksters who market worthless "water ionizer" machines for this purpose. Acidic water is sometimes described by engineers as "aggressive"; this refers to its tendency to corrode metal distribution pipes, but in this sense it is no more active than the hydrochloric acid already present in your gastric fluid!
Ion-free water
One occasionally hears that mineral-free water, and especially distilled water, are unhealthy because they "leach out" required minerals from the body. There is no truth to this; the fact is that mineral ions do not pass through cell walls by ordinary osmotic diffusion, but rather are actively transported by metabolic processes. An extensive 2008 study failed to confirm earlier reports that low calcium/magnesium in drinking water correlates with cardiovascular disease. Any well-balanced diet should supply all the mineral substances we need.
It is well known that people who are engaged in heavy physical activity or are in a very hot environment should avoid drinking large quantities of even ordinary water. In order to prevent serious electrolyte imbalance problems, it is necessary to make up for the salts lost through perspiration. This can be accomplished by ingestion of salted foods or beverages (including "sports beverages"), or salt tablets.
Water in our bodies
About two-thirds of the weight of an adult human consists of water. About two-thirds of this water is located within cells, while the remaining third consists of extracellular water, mostly in the blood plasma and in the interstitial fluid that bathes the cells. This water, amounting to about five percent of body weight (about 5 L in the adult), serves as a supporting fluid for the blood cells and acts as a means of transporting chemicals between cells and the external environment. It is basically a 0.15M solution of salt (NaCl) containing smaller amounts of other electrolytes, the most important of which are bicarbonate (HCO3–) and protein anions.
The water content of our bodies is tightly controlled in respect to both total volume and its content of dissolved substances, particulary ions. Drinking constitutes only one source of our water; many foods, especially those containing cells (fruits, vegetables, meats) are an important secondary source. In addition, a considerable amount of water (350-400 mL/day) is produced metabolically — that is, from the oxidation of glucose derived from foods.
The quantity of water exchanged within various parts of our bodies is surprisingly large. The kidneys process about 180 L/day, returning most of the water to the blood stream. Lymph flow amounts to 1-2.5 L/day, and turnover of fluids in the bowel to 8-9 L/day. These figures are dwarfed by the 80,000 L/day of water that diffuses in both directions through capillary walls.
How much water should I drink?
The idea that everyone should drink "eight glasses" of water a day is one of those urban legends that never seems to go away; it is nicely debunked at this medical myths site.
The body's daily water loss
- Loss through breath: 800 mL
- Minimal sweat loss: 100 mL
- Fecal loss: 200 mL
- Minimal urine loss: 500 mL
Total: 1600 mL
Ultimately, total water intake plus metabolic production must balance water loss. For a healthy unstressed adult, the figures shown here are typical minimum values. Notice that the major loss is through simple breathing. The minimal urinary loss is determined by the need to remove salts and other solutes taken in with foods or produced by metabolic processes. Individuals (such as many elderly) having reduced kidney function produce more dilute urine, and must therefore take in more water. And of course stress factors such as strenuous exercise, exposure to very high temperatures, or diarrhea can greatly increase the need for water intake.
Consumption of overly large quantities of water can lead to electrolyte imbalance resulting in water intoxication. Children, with their low body masses, are especially susceptible.
Bound water
As we explained above, bulk liquid water consists of a seething mass of various-sized chain-like groups and that flicker in and out of existence on a time scale of picoseconds. But in the vicinity of a solid surface or of another molecule or ion that possesses an unbalanced electric charge, water molecules can become oriented and sometimes even bound into relatively stable structures.
Water in ionic hydration shells
Water molecules interact strongly with ions, which are electrically-charged atoms or molecules. Dissolution of ordinary salt (NaCl) in water yields a solution containing the ions Na+ and Cl –. Owing to its high polarity, the H2O molecules closest to the dissolved ion are strongly attached to it, forming what is known as the inner or primary hydration shell. Positively-charged ions such as Na+ attract the negative (oxygen) ends of the H2O molecules, as shown in the diagram below. The ordered structure within the primary shell creates, through hydrogen-bonding, a region in which the surrounding waters are also somewhat ordered; this is the outer hydration shell, or cybotactic region.
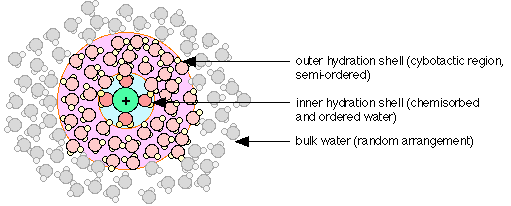
Some recent experiments have revealed a degree of covalent bonding between the d-orbitals of transition metal ions and the oxygen atoms of water molecules in the inner hydration shell.
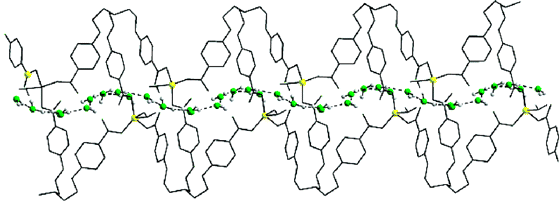
In 2003, some chemists in India found (Inorg. Chem. 44(4) pp 816 - 818) that a suitable molecular backbone (above) can cause water molecules to form a "thread" that can snake its way though the more open space of the larger molecules. What all of these examples show is that water can have highly organized local structures when it interacts with molecules capable of imposing these structures on the water.
Biowater: Bound water in biological systems
It has long been known that the intracellular water very close to any membrane or organelle (sometimes called vicinal water) is organized very differently from bulk water, and that this structured water plays a significant role in governing the shape (and thus biological activity) of large folded biopolymers. It is important to bear in mind, however, that the structure of the water in these regions is imposed solely by the geometry of the surrounding hydrogen bonding sites.
Water can hydrogen-bond not only to itself, but also to any other molecules that have -OH or -NH2 units hanging off of them. This includes simple molecules such as alcohols, surfaces such as glass, and macromolecules such as proteins. The biological activity of proteins (of which enzymes are an important subset) is critically dependent not only on their composition but also on the way these huge molecules are folded; this folding involves hydrogen-bonded interactions with water, and also between different parts of the molecule itself. Anything that disrupts these intramolecular hydrogen bonds will denature the protein and destroy its biological activity. This is essentially what happens when you boil an egg; the bonds that hold the eggwhite protein in its compact folded arrangement break apart so that the molecules unfold into a tangled, insoluble mass which, like Humpty Dumpty, cannot be restored to their original forms. Note that hydrogen-bonding need not always involve water; thus the two parts of the DNA double helix are held together by H—N—H hydrogen bonds.
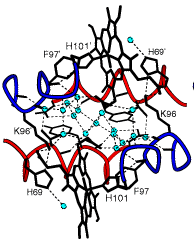
This image, taken from the work of William Royer Jr. of the U. Mass. Medical School, shows the water structure (small green circles) that exists in the space between the two halves of a kind of dimeric hemoglobin. The thin dotted lines represent hydrogen bonds. Owing to the geometry of the hydrogen-bonding sites on the heme protein backbones, the H2O molecules within this region are highly ordered; the local water structure is stabilized by these hydrogen bonds, and the resulting water cluster in turn stabilizes this particular geometric form of the hemoglobin dimer.
Can you run your car on water?
Not really. For water to act as a fuel, there must be some combination of oxygen and hydrogen that is energetically more stable than H2O, and no such molecule is known.
This fact has failed to put to rest the venerable urban legend that some obscure inventor discovered a process to do this, but the invention was secretly bought up by the oil companies in order to preserve their monopoly.
However, adding water to the fuel-air mixture in an internal combustion engine, a process known as water injection, has been employed for many years as a method of improving the performance of both piston- and turbine engines. Water injection kits are widely available, many offered by hucksters whose marketing falsely implies that their products allow you to "run your car on water". Don't believe it! And get some solid advice before you try this on a modern computer-controlled high compression engine.
In 2007, a widely-cited YouTube video appeared that showed a sample of salt water "burning". This occurs only in the presence of a strong radio-frequency field, which supposedly dissociates the water into H2 and O2. These two gases then recombine, producing the flame. Although there has been much uninformed hype about this being some kind of a breakthrough as a source of "energy from water", there is no reason to believe that the First Law of Thermodynamics has been repealed. If the energy supplied by the radio-frequency source is taken into account, you can be sure that there has been no net energy gain.
The actual mechanism of the process remains unclear. The fact that salt or some other ionic solute is required suggests that ions at the water's surface might be accelerated in the local field produced by the plasma discharge, helping to break up the molecules in the water vapor.
Contributors and Attributions
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook
- Thumbnail: pixabay.com/photos/water-dro...h-wet-1759703/