Fractional Distillation of Ideal Mixtures
- Page ID
- 3869
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page explains how the fractional distillation (both in the lab and industrially) of an ideal mixture of liquids relates to their phase diagram.
Using the phase diagram
On the last page, we looked at how the phase diagram for an ideal mixture of two liquids was built up. I want to start by looking again at material from the last part of that page. The next diagram is new - a modified version of diagrams from the previous page.
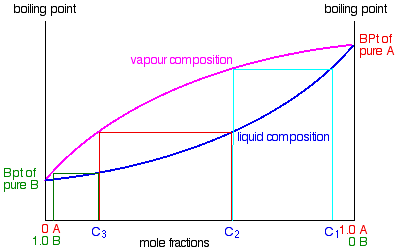
- If you boil a liquid mixture C1, you will get a vapor with composition C2, which you can condense to give a liquid of that same composition (the pale blue lines).
- If you reboil that liquid C2, it will give a vapor with composition C3. Again you can condense that to give a liquid of the same new composition (the red lines).
- Reboiling the liquid C3 will give a vapor still richer in the more volatile component B (the green lines). You can see that if you were to do this once or twice more, you would be able to collect a liquid which was virtually pure B.
The secret of getting the more volatile component from a mixture of liquids is obviously to do a succession of boiling-condensing-reboiling operations. It is not quite so obvious how you get a sample of pure A out of this.
Fractional Distillation in the lab
A typical lab fractional distillation would look like this:
The fractionating column is packed with glass beads (or something similar) to give the maximum possible surface area for vapor to condense on. Some fractionating columns have spikes of glass sticking out from the sides which serve the same purpose. If you sketch this, make sure that you do not completely seal the apparatus. There has to be a vent in the system otherwise the pressure build-up when you heat it will blow the apparatus apart. In some cases, where you are collecting a liquid with a very low boiling point, you may need to surround the collecting flask with a beaker of cold water or ice. The mixture is heated at such a rate that the thermometer is at the temperature of the boiling point of the more volatile component. Notice that the thermometer bulb is placed exactly at the outlet from the fractionating column.
Relating what happens in the fractionating column to the phase diagram
Suppose you boil a mixture with composition C1. The vapor over the top of the boiling liquid will be richer in the more volatile component, and will have the composition C2.
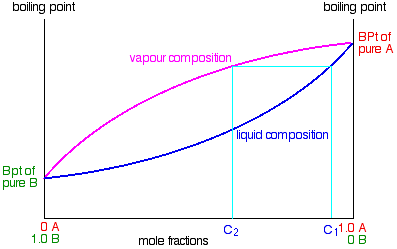
That vapor now starts to travel up the fractionating column. Eventually it will reach a height in the column where the temperature is low enough that it will condense to give a liquid. The composition of that liquid will, of course, still be C2. So what happens to that liquid now? It will start to trickle down the column where it will meet new hot vapor rising. That will cause the already condensed vapor to reboil.

Some of the liquid of composition C2 will boil to give a vapor of composition C3. Let's concentrate first on that new vapor and think about the unvaporized part of the liquid afterwards.
The vapor
This new vapor will again move further up the fractionating column until it gets to a temperature where it can condense. Then the whole process repeats itself. Each time the vapor condenses to a liquid, this liquid will start to trickle back down the column where it will be reboiled by up-coming hot vapor. Each time this happens the new vapor will be richer in the more volatile component. The aim is to balance the temperature of the column so that by the time vapor reaches the top after huge numbers of condensing and reboiling operations, it consists only of the more volatile component - in this case, B.
Whether or not this is possible depends on the difference between the boiling points of the two liquids. The closer they are together, the longer the column has to be.
The liquid
So what about the liquid left behind at each reboiling? Obviously, if the vapor is richer in the more volatile component, the liquid left behind must be getting richer in the other one. As the condensed liquid trickles down the column constantly being reboiled by up-coming vapor, each reboiling makes it richer and richer in the less volatile component - in this case, A. By the time the liquid drips back into the flask, it will be very rich in A indeed. So, over time, as B passes out of the top of the column into the condenser, the liquid in the flask will become richer in A. If you are very, very careful over temperature control, eventually you will have separated the mixture into B in the collecting flask and A in the original flask. Finally, what is the point of the packing in the column?
To make the boiling-condensing-reboiling process as effective as possible, it has to happen over and over again. By having a lot of surface area inside the column, you aim to have the maximum possible contact between the liquid trickling down and the hot vapor rising. If you didn't have the packing, the liquid would all be on the sides of the condenser, while most of the vapor would be going up the middle and never come into contact with it.
Fractional distillation industrially
There is no difference whatsoever in the theory involved. All that is different is what the fractionating column looks like. The diagram shows a simplified cross-section through a small part of a typical column.

The column contains a number of trays that the liquid collects on as the vapor condenses. The up-coming hot vapor is forced through the liquid on the trays by passing through a number of bubble caps. This produces the maximum possible contact between the vapor and liquid. This all makes the boiling-condensing-reboiling process as efficient as possible. The overflow pipes are simply a controlled way of letting liquid trickle down the column.
If you have a mixture of lots of liquids to separate (such as in petroleum fractionation), it is possible to tap off the liquids from some of the trays rather than just collecting what comes out of the top of the column. That leads to simpler mixtures such as gasoline, kerosene and so on.
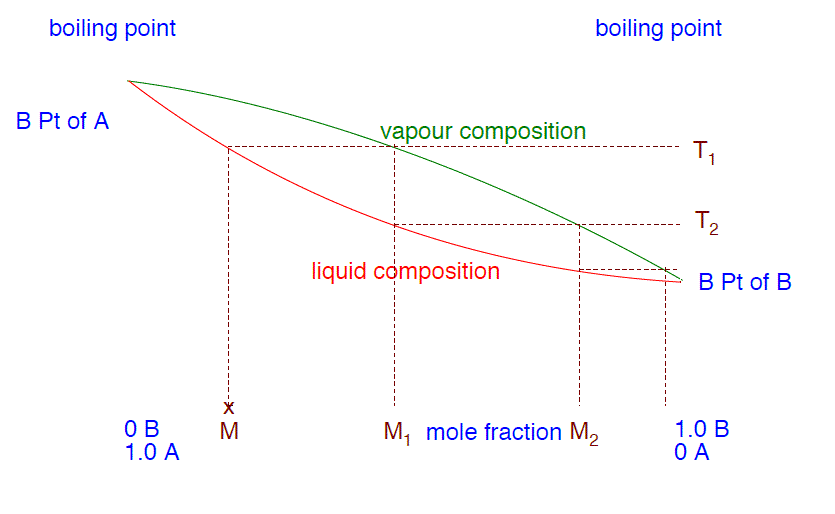
- Use the phase diagram below to explain how you can obtain a pure sample of B from a mixture M by successively boiling and condensing the liquid mixture.
- Why is it important to carefully control how strongly the original mixture is heated during the separation?
- Explain briefly how the separation occurs, making use of the phase diagram above if you think it helps.
Solution
- If you boil the mixture M, it will boil at a temperature T1. The vapor above the liquid at this temperature will be richer in the more volatile substance B. If you condense that vapor, it will give a liquid of the composition M1. If you reboil that, it will boil at a temperature T2. The vapor over that liquid will have a composition M2, still richer in B. If you go on doing that, reboiling and recondensing, then the vapor becomes richer and richer in B until it eventually becomes pure B. When you finally get to that point and condense the vapor, then you will have pure B liquid.
- You have to be sure that only the vapor of the more volatile of the two liquids passes into the condenser. That means that the thermometer has to read exactly the boiling point of the more volatile liquid. If it is below that, then nothing is going to pass out into the condenser. If it is above that, then your distillate will still contain some of the less volatile component.
- B is the more volatile liquid; A is the less volatile one. The vapor over the boiling liquid in the flask will be richer in B than the original liquid is. That vapor will pass up the column until the temperature falls enough for it to condense to give a liquid richer in B than the one in the flask (equivalent to M1 in the diagram). This will start to trickle down the column. Hot vapor coming up from the flask will reboil the condensed liquid, giving a vapor which will be even richer in B (M2 on the diagram). This will condense to a liquid, trickle down the column and then be reboiled. This continuous process will go on until the vapor is entirely B. The column is heated so that this is finally complete right at the top of the column. Meanwhile, the liquids trickling down the column get richer and richer in A as the B is removed and carried up the column. Eventually, the liquid in the flask will end up as pure A.