Substitution of the hydroxyl group
- Page ID
- 786
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Reactions in which the hydroxyl group of a carboxylic acid is replaced by another nucleophilic group are important for preparing functional derivatives of carboxylic acids. The alcohols provide a useful reference chemistry against which this class of transformations may be evaluated. In general, the hydroxyl group proved to be a poor leaving group, and virtually all alcohol reactions in which it was lost involved a prior conversion of –OH to a better leaving group. This has proven to be true for the carboxylic acids as well.
Four examples of these hydroxyl substitution reactions are presented by the following equations. In each example, the new bond to the carbonyl group is colored magenta and the nucleophilic atom that has replaced the hydroxyl oxygen is colored green. The hydroxyl moiety is often lost as water, but in reaction #1 the hydrogen is lost as HCl and the oxygen as SO2. This reaction parallels a similar transformation of alcohols to alkyl chlorides, although its mechanism is different. Other reagents that produce a similar conversion to acyl halides are PCl5 and SOBr2.
The amide and anhydride formations shown in equations #2 & 3 require strong heating, and milder procedures that accomplish these transformations will be described in the next chapter.
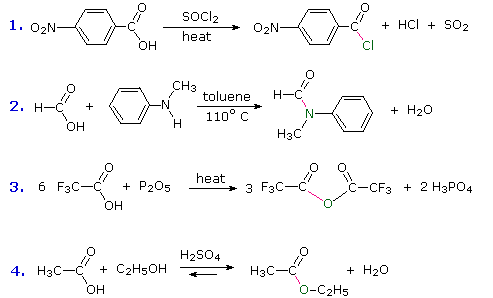
Reaction #4 is called esterification, since it is commonly used to convert carboxylic acids to their ester derivatives. Esters may be prepared in many different ways; indeed, equations #1 and #4 in the previous diagram illustrate the formation of tert-butyl and methyl esters respectively. The acid-catalyzed formation of ethyl acetate from acetic acid and ethanol shown here is reversible, with an equilibrium constant near 2. The reaction can be forced to completion by removing the water as it is formed. This type of esterification is often referred to as Fischer esterification. As expected, the reverse reaction, acid-catalyzed ester hydrolysis, can be carried out by adding excess water.
A thoughtful examination of this reaction (#4) leads one to question why it is classified as a hydroxyl substitution rather than a hydrogen substitution. The following equations, in which the hydroxyl oxygen atom of the carboxylic acid is colored red and that of the alcohol is colored blue, illustrate this distinction (note that the starting compounds are in the center).
H2O + CH3CO-OCH2CH3 |
H-substitution |
|
HO-substitution |
CH3CO-OCH2CH3 + H2O |
In order to classify this reaction correctly and establish a plausible mechanism, the oxygen atom of the alcohol was isotopically labeled as 18O (colored blue in our equation). Since this oxygen is found in the ester product and not the water, the hydroxyl group of the acid must have been replaced in the substitution. A mechanism for this general esterification reaction will be displayed on clicking the "Esterification Mechanism" button; also, once the mechanism diagram is displayed, a reaction coordinate for it can be seen by clicking the head of the green "energy diagram" arrow. Addition-elimination mechanisms of this kind proceed by way of tetrahedral intermediates (such as A and B in the mechanism diagram) and are common in acyl substitution reactions. Acid catalysis is necessary to increase the electrophilic character of the carboxyl carbon atom, so it will bond more rapidly to the nucleophilic oxygen of the alcohol. Base catalysis is not useful because base converts the acid to its carboxylate anion conjugate base, a species in which the electrophilic character of the carbon is reduced.
Since a tetrahedral intermediate occupies more space than a planar carbonyl group, we would expect the rate of this reaction to be retarded when bulky reactants are used. To test this prediction the esterification of acetic acid was compared with that of 2,2-dimethylpropanoic acid, (CH3)3CO2H. Here the relatively small methyl group of acetic acid is replaced by a larger tert-butyl group, and the bulkier acid reacted fifty times slower than acetic acid. Increasing the bulk of the alcohol reactant results in a similar rate reduction.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


