15.1: Naming Aromatic Compounds
- Page ID
- 31562
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- draw the structure of each of the common aromatic compounds in Figure 16 (Common benzene derived compounds with various substituents), given their IUPAC-accepted trivial names.
- write the IUPAC-accepted trivial name for each of the compounds in Figure 16, given the appropriate Kekulé, condensed or shorthand structure.
- identify the ortho, meta and para positions in a monosubstituted benzene ring.
- use the ortho/meta/para system to name simple disubstituted aromatic compounds.
- draw the structure of a simple disubstituted aromatic compound, given its name according to the ortho/meta/para system.
- provide the IUPAC name of a given aromatic compound containing any number of the following substituents: alkyl, alkenyl or alkynyl groups; halogens; nitro groups; carboxyl groups; amino groups; hydroxyl groups.
- draw the structure of an aromatic compound containing any number of the substituents listed in Objective 6, above, given the IUPAC name.
- provide the IUPAC name of a given aromatic compound in which the phenyl group is regarded as a substituent.
- draw the Kekulé, condensed or shorthand structure of an aromatic compound in which the phenyl group is regarded as a substituent, given its IUPAC name.
Make certain that you can define, and use in context, the key terms below.
- arene
- benzyl group
- phenyl group
You should already know the names and structures of several of the hydrocarbons shown in Figure 15.1. A compound containing a benzene ring which has one or more alkyl substituents is called an arene.
A phenyl group consists of a benzene ring with one of its hydrogens removed.
You should memorize the structures and formulas shown in Figure 16. You will meet these compounds frequently throughout the remainder of this course.
Note that the ortho/meta/para system cannot be used when more than two substituents are present in the benzene ring. The “numbering system” can be used instead of the ortho/meta/para system in most cases when only two substituents are present.
Sources of Aromatic Compounds
Initially aromatic compounds were isolated from coal tar. Coal tar, which is a distillate obtained when heating coal at 1000 oC in the absence of air, is a source of an amazing number of aromatic compounds. Many simple aromatic compounds, some of which includes nitrogen, oxygen, and sulfur, as well as hydrocarbons are obtained.
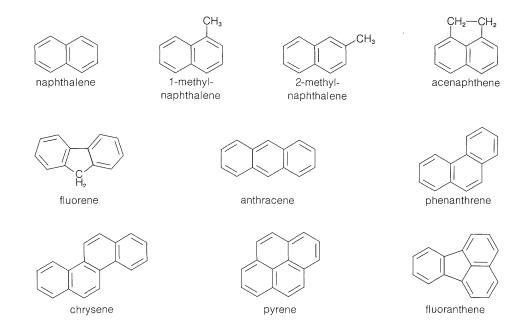
Some of the Aromatic Compounds Obtained from Coal Tar
Prior to World War II, coal tar was the only important source of aromatic hydrocarbons, but during the war the demand for benzene and toluene, a precursor to the explosive TNT, rose so sharply that other sources had to be found. Today, most of the benzene and almost all of the toluene produced in the United States are derived from petroleum. Although petroleum does contain some aromatic compound, it primarily made up of alkanes of various chain lengths. Aromatic compounds are synthesized from petroleum the by a process referred to in the petroleum industry as catalytic re-forming or hydroforming. This involves heating a C6-C10 alkane fraction of petroleum with hydrogen in the presence of a catalyst to modify the molecular structure of its components. Some amazing transformations take place, and the C6-C7 alkanes can be converted to cycloalkanes, which, in turn, are converted to arenes. Benzene, and methylbenzene (toluene) are produced primarily in this way.
Nomenclature of Mono-Substituted Benzenes
Unlike aliphatic organics, nomenclature of benzene-derived compounds can be confusing because a single aromatic compound can have multiple possible names (such as common and systematic names) be associated with its structure. Common names are often used in the nomenclature of aromatic compounds. IUPAC still allows for some of the more widely used common name to be used. A partial list of these common name is shown in Figure \(\PageIndex{2}\) and there are numerous others. These common names take the place of the benzene base name. Methylbenzene is commonly known with the base name toluene, hydroxyphenol is known as phenol etc. It is very important to be able to identify these structures as they will be utilized in the nomenclature of more complex compounds.
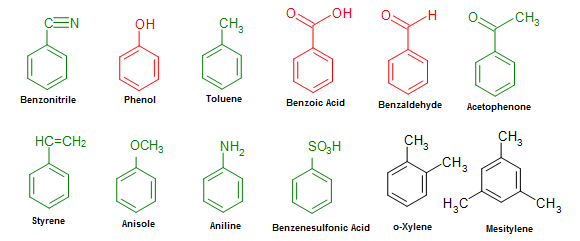
Mono-substituted benzene rings, with a substituent not on the list above, are named with benzene being the parent name. These compounds are named as such: Name of the substituent + Benzene.
The use of Phenyl and Benzyl in Nomenclature
If the alkyl group attached to the benzene contains seven or more carbons the compounds is named as a phenyl substituted alkane. The name phenyl (C6H5-)is often abbreviated (Ph) and comes from the Greek word pheno which means "I bear light". This name commemorates the fact that benzene was first isolated by Michael Faraday in 1825 from the residue left in London street lamps which burned coal gas. If the alkyl substituent is smaller than the benzene ring (six or fewer carbons), the compound is named as an alkyl-substituted benzene following the rules listed above.
The benzyl group (abbv. Bn), similar to the phenyl group and can be written as C6H5CH2-R, PhCH2-R, or Bn-R. Nomenclature of benzyl group based compounds are very similar to the phenyl group compounds. For example, a chlorine attached to a benzyl group would simply be called benzyl chloride, whereas an OH group attached to a benzyl group would simply be called benzyl alcohol.
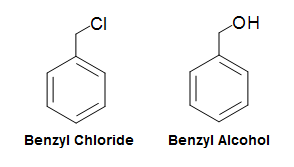
Nomenclature of Disubstituted Benzenes
With disubstituted benzenes there are three distinct positional isomers which can occur and must be identified in the compounds name. Although numbering can be used to indicate the position of the two subsituents it is much more common for the compounds to be named using prefixes. These prefixes are italicized and are often abbreviated with a single letter. They are defined as the following:
- ortho- (o-): 1,2- (next to each other in a benzene ring)
- meta- (m): 1,3- (separated by one carbon in a benzene ring)
- para- (p): 1,4- (across from each other in a benzene ring)
Nomenclature of Benzenes with Three or more Substituents
When three or more substituents are present the ortho, meta, para positional prefixes become inadequate and a numbering system for the ring must be applied. Here again it is important to check if any of the substituents are listed in Figure \(\PageIndex{2}\). If a substituent from Figure \(\PageIndex{2}\) is present it is given the parent name in the nomenclature. Also, this substituent is given position one in the numbering system. The other substituents are numbered such that they get the lowest possible sum. In the compound's name the subsituents are given their position number and listed alphabetically. Remember that di-, tri, tetra- prefixes are still used to indicate multiple of the same substituent being present but are ignored for alphabetical listing.
References
- Nicolaou, K. C., & Montagnon, T. (2008). Molecules That Changed the World. KGaA, Weinheim: Wiley-VCH. p. 54
- Pitman, V. (2004). Aromatherapy. Great Britain, UK: Nelson Thornes. p.135-136
- Burton, G. (2000). Chemical Ideas. Bicester, Oxon: Heinemann. p.290-292
- Vollhardt, K. P.C. & Shore, N. (2007). Organic Chemistry (5th Ed.). New York: W. H. Freeman. p. 667-669
- Schnaubelt, K. (1999). Medical Aromatherapy. Berkeley, CA: Frog Books. p. 211-213
- Patrick, G. L. (2004). Organic Chemistry. New York, NY: Taylor & Francis. p. 135-136
- Talbott, S. M. (2002). A Guide to Understanding Dietary Supplements. Binghamton, NY: Haworth Press. p. 616-619
- Lifton, R. J. (2000). The Nazi doctors. New York, NY: Basic Books. p. 255-261
- Myers, R. L., & Myers, R. L. (2007). The 100 most important chemical compounds. Westport, CT: Greenwood Publishing Group. p. 281-282
Exercises
(True/False) The compound above contains a benzene ring and thus is aromatic.
- Answer
-
False, this compound does not contain a benzene ring in its structure.
Benzene unusual stability is caused by how many conjugated pi bonds in its cyclic ring? ____
- Answer
-
3
Menthol, a topical analgesic used in many ointments for the relief of pain, releases a peppermint aroma upon exposure to the air. Based on this conclusion, can you imply that a benzene ring is present in its chemical structure? Why or why not?
- Answer
-
No, a substance that is fragrant does not imply a benzene ring is in its structure. See camphor example (figure 1)
- Answer
-
No reaction, benzene requires a special catalyst to be hydrogenated due to its unusual stability given by its three conjugated pi bonds.
At normal conditions, benzene has ___ resonance structures.
- Answer
-
2
Which of the following name(s) is/are correct for the following compound?
- nitrohydride benzene
- phenylamine
- phenylamide
- aniline
- nitrogenhydrogen benzene
- All of the above is correct
- Answer
- b, d
Convert 1,4-dimethylbenzene into its common name.
- Answer
-
p-Xylene
TNT's common name is: ______________________________
- Answer
-
2,4,6-trinitrotoluene
Name the following compound using OMP nomenclature:
- Answer
-
p-chloronitrobenzene
Draw the structure of 2,4-dinitrotoluene.
- Answer
-
Name the following compound:
- Answer
-
4-phenylheptane
Which of the following is the correct name for the following compound?
- 3,4-difluorobenzyl bromide
- 1,2-difluorobenzyl bromide
- 4,5-difluorobenzyl bromide
- 1,2-difluoroethyl bromide
- 5,6-difluoroethyl bromide
- 4,5-difluoroethyl bromide
Solution
a
(True/False) Benzyl chloride can be abbreviated Bz-Cl.
- Answer
-
False, the correct abbreviation for the benzyl group is Bn, not Bz. The correct abbreviation for Benzyl chloride is Bn-Cl.
Benzoic Acid has what R group attached to its phenyl functional group?
- Answer
-
COOH
(True/False) A single aromatic compound can have multiple names indicating its structure.
- Answer
-
True. TNT, for example, has the common name 2,4,6-trinitrotoluene and its systematic name is 2-methyl-1,3,5-trinitrobenzene.
List the corresponding positions for the OMP system (o-, m-, p-).
- Answer
-
Ortho - 1,2 ; Meta - 1,3 ; Para - 1,4
A scientist has conducted an experiment on an unknown compound. He was able to determine that the unknown compound contains a cyclic ring in its structure as well as an alcohol (-OH) group attached to the ring. What is the unknown compound?
- Cyclohexanol
- Cyclicheptanol
- Phenol
- Methanol
- Bleach
- Cannot determine from the above information
- Answer
-
The correct answer is f). We cannot determine what structure this is since the question does not tell us what kind of cyclic ring the -OH group is attached on. Just as cyclohexane can be cyclic, benzene and cycloheptane can also be cyclic.
Which of the following statements is false for the compound, phenol?
- Phenol is a benzene derived compound.
- Phenol can be made by attaching an -OH group to a phenyl group.
- Phenol is highly toxic to the body even in small doses.
- Phenol can be used as a catalyst in the hydrogenation of benzene into cyclohexane.
- Phenol is used as an antiseptic in minute doses.
- Phenol is amongst one of the three common names retained in the IUPAC nomenclature.
- Answer
-
d
State wither the following is para, meta, or ortho substituted.
- Answer
-
A – meta; B – para; C – ortho
Name the following compounds.
- Answer
-
a. 1,3-Dibromobenzene
b. 1-phenyl-4-methylhexane
c. 1,4-Dichloro-2,5-dimethylbenzene
d. 2-methyl-1,3,5-trinitrobenzene. (Also known as trinitrotoluene, or TNT)
Draw the following structures
- p-chloroiodobenzene
- m-bromotoluene
- p-chloroaniline
- 1,3,5-trimethylbenzene
- Answer
-

