6.2: Fatty acyls
- Page ID
- 432913
- Understand the basic structural features of fatty acids, the notations reflecting them, and their relationship to the melting points and health effects.
- Define essential fatty acids, omega fatty acids, and their importance.
- Understand the basic structure features, nomenclature, and functions of prostaglandins.
- Understand the structures and uses of important waxes.
Fatty acyls are a group of lipids that contain a fatty acid or its derivative.
Fatty acids
Fatty acids are carboxylic acids with an alkyl chain that is usually unbranched and containing an even number of \(\ce{C's}\), usually between 8 to 20 \(\ce{C's}\).
Figure \(\PageIndex{1}\) shows some examples of fatty acids that have 18 \(\ce{C's}\) in the chain. Carboxylic acid group (\(\ce{-COOH}\) is hydrophilic, and the alkyl chain is hydrophobic, making fatty acids amphiphilic. The hydrophobic property of the long alkyl chain dominates, making fatty acids insoluble in water.

- The alkyl chain can be saturated with all \(\ce{C-C}\) \(\sigma\)-bonds, called saturated fatty acid (SFA), e.g., steric acid;
- or may contain some double bonds (\(\ce{C=C}\)), called unsaturated fatty acid (USFA)., e.g., oleic acid and linoleic acid Figure \(\PageIndex{1}\).
- The unsaturated fatty acid may have one \(\ce{C=C}\) bond called monounsaturated fatty acid (MUFA), e.g., oleic acid;
- or more than one \(\ce{C=C}\) bonds called polyunsaturated fatty acid (PUFA), e.g., linoleic acid.
Notations for and labelling \(\ce{C's}\) in fatty acids
Short notations
Carboxylic acids are usually referred to by their trivial names. Short notation is used to indicate # of \(\ce{C's}\):# of \(\ce{C=C}\) bonds, e.g., steric acid Figure \(\PageIndex{1}\) is 18:0 meaning eighteen carbons and zero double bonds. Similarly, oleic acid is 18:1 meaning eighteen carbons and one double bond, and linoleic acid is 8:2 meaning eighteen carbons and two double bonds.
Labelling \(\ce{C's}\)
Systematic IUPAC nomenclature starts numbers from carbonyl-carbon (\(\ce{C=O}\)) as shown in Figure \(\PageIndex{2}\), red numbers. Trivial nomenclature uses Greek alphabets starting from \(\ce{C}\)#2 as \(\alpha\), next one as \(\beta\), than \(\gamma\), and so on, as shown in Figure \(\PageIndex{2}\), black numbers. IUPAC short notation shows the position labels of the double bonds' first \(\ce{C's}\) in a bracket, separated by commas, next to the regular short notation. The numbering begins from (\(\ce{-COOH}\) end. For example, steric acid 18:0, i.e., eighteen carbons and zero double bonds, oleic acid is 18:1(9), i.e., eighteen carbons and one double bond at \(\ce{C}\)# 9, and linoleic acid is 18:2(9, 12), i.e., eighteen carbons and bonds, one at \(\ce{C}\)# 9 and the other at \(\ce{C}\)# 12.
Omega-minus (\(\omega\)-#) labeling
In omega-minus (\(\omega\)-#) labeling the last \(\ce{C}\), i.e., the \(\ce{-CH3}\) at the end opposite to carboxyl (\(\ce{-COOH}\)) group is labeled \(\omega\)-1, the \(\ce{C}\) next to it as \(\omega\)-2, than \(\omega\)-3 and so on, as as shown in Figure \(\PageIndex{2}\), in blue fonts. Recall that \(\omega\) is the last alphabet in Greek, so, \(\omega\)-1 is the last carbon. The "omega-#" (\(\omega\)-#) label is usually used to refer to the position of the first \(\ce{C}\) of the first \(\ce{C=C}\) bond from the \(\omega\)-end, i.e., from the \(\ce{-CH3}\) end, opposite to the \(\ce{-COOH}\) end. For example, \(\alpha\)-linolenic acid in Figure \(\PageIndex{2}\) is an \(\omega\)-3 fatty acid.

Table 1 lists common fatty acids with their IUPAC short notations, \(\omega\)-#, and melting points.
| Common name | Condensed formula | IUPAC notation* | Melting point (oC) | \(\omega\)-# | Found in |
|---|---|---|---|---|---|
| Saturated fatty acids | |||||
| Caprylic acid | CH3(CH2)6COOH | 8:0 | 17 | Milk | |
| Capric acid | CH3(CH2)8COOH | 10:0 | 32 | Coconut | |
| Lauric acid | CH3(CH2)10COOH | 12:0 | 44 | Coconut | |
| Myristic acid | CH3(CH2)12COOH | 14:0 | 55 | Nutmeg | |
| Palmitic acid | CH3(CH2)14COOH | 16:0 | 63 | Palm | |
| Stearic acid | CH3(CH2)16COOH | 18:0 | 69 | Animal fat | |
| Arachidic acid | CH3(CH2)18COOH | 20:0 | 75 | Corn | |
| Monounsaturated fatty acids | |||||
| Myristoleic acid | CH3(CH2)3CH=CH(CH2)7COOH | 14:1(9) | 0 | \(\omega\)−5 | Butter |
| Oleic acid | CH3(CH2)7CH=CH(CH2)7COOH | 18:1(9) | 14 | \(\omega\)-9 |
Olives, pecan, grapeseed |
| Elaidic acid | CH3(CH2)7CH=CH(CH2)7COOH | 18:1(9t) | 45 | \(\omega\)-9 | Milk |
| Erucic acid | CH3(CH2)7CH=CH(CH2)11COOH | 22:1(13) | 34 | \(\omega\)-9 | Rapeseed |
| Polyunsaturated fatty acids | |||||
| Linoleic acid | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | 18:2(9,12) | -5 | \(\omega\)−6 |
Soybean, safflower, sunflower |
| \(\alpha\)-Linolenic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH | 18:3(9,12,15) | -11 | \(\omega\)−3 | Corn |
| Arachidonic acid | CH3(CH2)4CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOH | 20:4(5,8,11,14) | -50 | \(\omega\)−6 |
Meat, eggs, fish |
| Eicosapentaenoic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOH | 20:5(5,8,11,14,17) | -65 | \(\omega\)−3 | Fish |
| Docosahexaenoic acid | CH3CH3CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)2COOH | 22:6(4,7,10,13,16,19) | -44 | \(\omega\)−3 | Fish |
| * IUPAC notation shows Numbers of \(\ce{C's}\):number of double bonds(position of cis-double bond, except when there is symbol t for trans-configuration) | |||||
\(\alpha\)-Linolenic acid (18:3(9,12,15)) -an \(\omega\)−3 fatty acid, and linoleic acid (18:2(9,12)) - an \(\omega\)−6 fatty acid are essential fatty acids (EFAs) because humans and other animals cannot synthesize them. They must be included in the diet as they are needed for good health. Although eicosapentaenoic acid (20:5(5,8,11,14,17)) and docosahexaenoic acid (22:6(4,7,10,13,16,19)), called long-chain unsaturated fatty acids, can be synthesized from \(\alpha\)-linolenic acid, the conversion efficiency is low, and it is recommended that these should also be included in the diet. Arachidonic acid (20:4(5,8,11,14)) is another long-chain unsaturated fatty acid that can be synthesized from linoleic acid.
Shapes and physical properties of fatty acids
Shapes
The saturated fatty acids are cylindrical that pack nicely in the solid state with a large contact area between the molecules. The double bonds in unsaturated fatty acids are usually in the cis configuration. A cis-\(\ce{C=C}\) introduces a kink in the structure that does not let molecules pack with a large contact area between neighboring molecules, as illustrated in Figure \(\PageIndex{3}\). Some unsaturated fatty acids have trans-double bonds and are cylindrical, like saturated fatty acids, but they are rare.


Melting points
The melting point of fatty acids is affected by two factors, as shown in Table 1:
- an increase in the number of \(\ce{C's}\) increases the melting point because it increases London dispersion forces holding the molecules together, and
- the presence of cis-\(\ce{C=C}\) bond decreases the melting point due to the kink in the structure that reduces the contact area between molecules and, consequently, London dispersion forces are less.
Fats containing saturated fatty acids are considered unhealthy as they tend to deposit in arteries forming plaque, i.e., atherosclerosis. It can lead to high blood pressure, rupture of arteries, or heart attack. Saturated fatty acids are more common in animal fats, while unsaturated fatty acids, considered healthier, are more common in vegetable oils.
It has been observed that people from Alaska eat more unsaturated fats and have a low occurrence of atherosclerosis and heart attack. Fats in Alaskan food is primarily from fish, such as salmon, tuna, and herring, as shown in Figure \(\PageIndex{4}\). Vegetable oils have higher contents of \(\omega\)-6 fatty acids, such as linoleic acid and arachidonic acid. Fish oil mainly contains \(\omega\)-3 fatty acids, such as linolenic acid, eicosapentaenoic acid, and docosahexaenoic acid.
 salmon
salmon tuna
tuna herring
herring
Prostaglandins
Prostaglandins are lipids containing 20 \(\ce{C's}\) that are derived from arachidonic acid (20:4(5, 8, 11, 14)) and act like hormones. They are found in small concentrations in almost all body tissues and cells. Their activities include blood pressure regulation, blood clotting, inflammation, gastric secretions, kidney functions, and reproductive activities. They are usually short-lived and produced locally where they perform their function.
Prostaglandins synthesis
Prostaglandins synthesis starts from arachidonic acid using enzymes like cyclooxygenase (COX-1 and COX-2) and prostaglandin synthase, like PGE-synthase and PGF-synthase, as illustrated in Figure \(\PageIndex{5}\). The prostaglandins have a five-membered ring and two side chains. The prostaglandin PGG2 is synthesized from arachidonate and quickly converts to PGH2. PGH2 regulates constriction and dilation of blood vessels and stimulates platelet aggregation. PGH2 converts to PGE2 and PGF2 which induce labor by stimulating uterine contraction. PGF2 also lowers blood pressure by relaxing smooth muscle cells in blood vessels. PGH2 is also a precursor of several other Prostaglandins and related biomolecules.
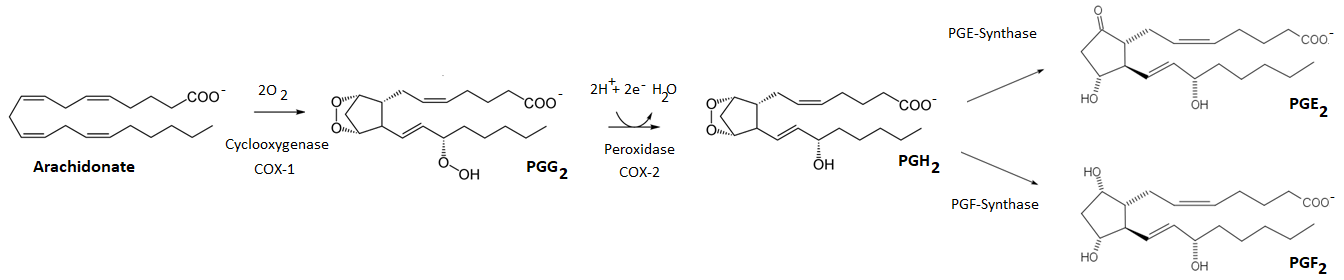
Prostaglandins names
The names of prostaglandins are abbreviated as three letters followed by a subscript, where the first two letters, i.e., PG, stands for prostaglandin, the 3rd letter indicates the substitution pattern, and the subscript tells the number of \(\ce{C=C}\) bonds in the side chains. For example, the 3rd letter G has endoperoxide in the ring and a hydroperoxide group at \(\ce{C's}\) 15, H has endoperoxide in the ring, and a hydroxyl group at \(\ce{C's}\) 15, E has a \(\beta\)-hydroxyketone, and F has a 1,3-diol groups in the ring. The subscript 2 shows two \(\ce{C=C}\) bonds in the side chains. The letter E in PGE2 also indicates that it is soluble in ether and F in PGF2 indicates it is soluble in phosphate buffer.
When tissues are injured, arachidonic acid is converted into prostaglandins that cause inflammation and pain.



Waxes
Lipid waxes are esters of one long-chain fatty acid with unbranched long-chain alcohol containing 14 to 30 \(\ce{C's}\) per chain. For example, myricyl palmitate found in beeswax can be obtained by the esterification of palmitic acid and myricyl alcohol:
\[\ce{CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-OH +HO{-}(CH2)29CH3 -> CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)29CH3 + H2O}\nonumber \]

Functions of waxes
Waxes form a waterproof coating on plants' leaves, fruits, and stems to minimize water evaporation and protect against parasites. Examples include palm trees and jojoba wax from jojoba bushes. The wax coating on animals' skin, fur, and feathers protects water birds from water wetting and rainwater and shelters them from rainwater. Bees use wax to construct protection and housing for honey, as illustrated below. Sperm whales use spermaceti wax in their heads as a part of their eco analysis, i.e., navigation system.
 Honeycomb made of beeswax: \[\ce{CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)29CH3 }\nonumber \]
Honeycomb made of beeswax: \[\ce{CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)29CH3 }\nonumber \] Carnauba wax coating on palm tree leaves: \[\ce{CH3(CH2)24{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)29CH3}\nonumber \]
Carnauba wax coating on palm tree leaves: \[\ce{CH3(CH2)24{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)29CH3}\nonumber \] Jojoba wax on leaves and fruits of jojoba bush: \[\ce{CH3(CH2)18{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)19CH3}\nonumber \]
Jojoba wax on leaves and fruits of jojoba bush: \[\ce{CH3(CH2)18{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)19CH3}\nonumber \] Sperm whale has spermaceti wax in the head as a part of their eco-analysis syterm: \[\ce{CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)15CH3}\nonumber \]
Sperm whale has spermaceti wax in the head as a part of their eco-analysis syterm: \[\ce{CH3(CH2)14{-}\!\!{\overset{\overset{\huge\enspace\!{O}}|\!\!|\enspace}{C}}\!\!-O{-}(CH2)15CH3}\nonumber \]Beeswax and carnauba wax protective coatings on cars, furniture, floors, etc. Jojoba wax and spermaceti are used to make candles and cosmetics. A mixture of waxes obtained from wool is used in lotions for face and skin softening.
Petroleum-derived waxes or paraffin are also called waxes, but they are not esters but mixtures of long-chain hydrocarbons. Earwax is a mixture of glycerol esters, cholesterol, phospholipids, etc., from dead skin and secretions of cerumen glands. Like other lipid waxes, it protects the ear canal against bacteria, fungi, and water. Excess earwax can cause blockage in the ear canal and hearing loss.


