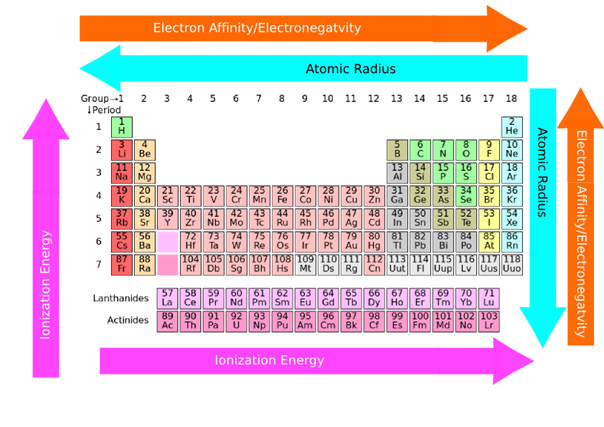
2.6: The periodic trends in properties of the elements
- Page ID
- 372868
Properties of elements generally show a periodic trend that correlates with their position in the periodic table. The properties and their periodic trends are described below.
Valence electrons
Electrons in the outermost shell are the valence electrons. Fig. 2.5.8 shows the valence electrons of the first twenty elements in red fonts.
All elements in a group have the same number of valence electrons equal to the first digit of their group number. For example, 1st group of hydrogen and alkali metals has one valence electron, 2nd group of alkali metals has two valence electrons, halogens in 17th group have seven valence electrons, and noble gases in 18th group have eight valence electrons.
The transition metals in groups 3 to 12, and inner-transition metals, i.e., lanthanoids and actinoids -the two rows of elements placed below the periodic table, are the exception to the general trend of valence electrons described above. Valence electron configurations of transition metals and inner-transition metals are not described here; it is beyond the scope of this book.
The valence electrons mainly determine the chemical properties of the elements. The elements in the same group have similar chemical properties because they have the same valence shell electron configuration. The elements in a row show a gradual change in chemical properties because their valence shell electron configuration changes gradually along the row.
Lewis symbols
- Lewis symbols show the valence electrons as dots around the symbol of an element. One dot represents one valence electron, e.g.,
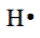 .
. - The dots are shown on any of the four sides of the symbol.
- A single dot on the top, bottom, left, or right is shown four valence electrons. Then start pairing the dots beyond four valence electrons, as shown in Fig. 2.6.1 for the first twenty elements.
- Helium is an exception that has only two valence electrons, but they are shown paired.
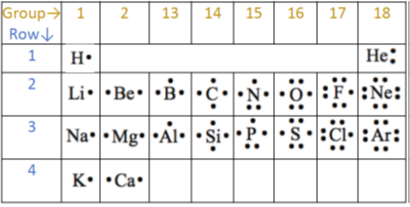
The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make.
Generally, each unpaired dot can make one bond.
For example, a hydrogen atom with one unaired dot can make one bond as in H-H. A bond is represented by a line between the bonded atoms. A bond is formed by sharing unpaired valence electrons. It is called a covalent bond. Carbon, nitrogen, oxygen, and fluorine with 4, 3, 2, and 1 unpaired dot can make 4, 3, 2, and 1 bond, e.g., in the following molecules: 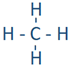 ,
, 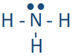 ,
, 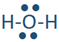 , and
, and 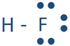 .
.
- Each line in these molecules represents a bonding electron pair, and
- the pair of dots represent valence electrons that are not involved in bonding, called lone pair of electrons.
Atomic size
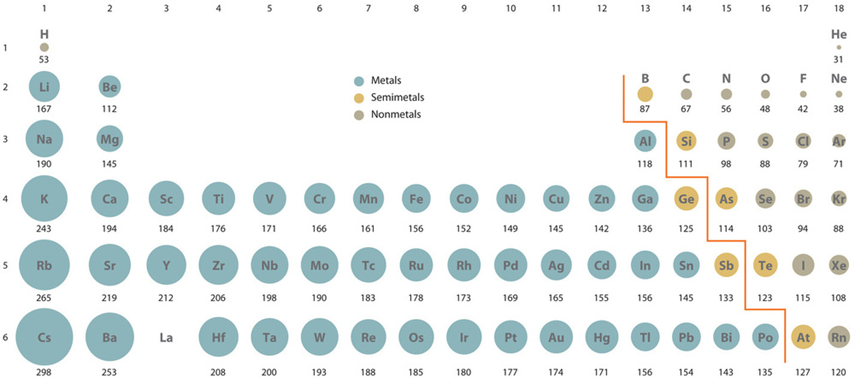
Electrons exist around the nucleus in a cloud-like appearance with no clearly defined boundaries. Therefore, the atomic size generally refers to the covalent radius of an atom that is one-half of the distance between the nuclei covalently bonded in a homonuclear molecule, like Cl2, I2, H2, as illustrated in Fig. 2.6.2.
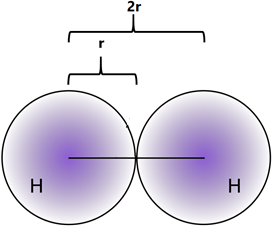
- The atomic size generally increases from top to bottom in a group because the valence electrons add in the higher shell in each consecutive member of a group down the column.
- The atomic size generally decreases from left to right in a row because the valence electrons are in the same shell while more protons add to the nucleus, increasing the pull on the valence electrons, as illustrated in Fig. 2.6.3.
Ionization energy
The positively charged nucleus attracts the negatively charged electrons. Therefore removal of an electron from the atom requires energy. The ionization produces a cation with fewer electrons than the parent neutral atom, i.e., cations.
Ionization energy is the energy needed to remove an electron from a neutral atom, as in the following reaction.
\[\mathrm{Na}+\text { ionization energy } \rightarrow \mathrm{Na}^{1+}+\mathrm{e}^{-}\nonumber\]
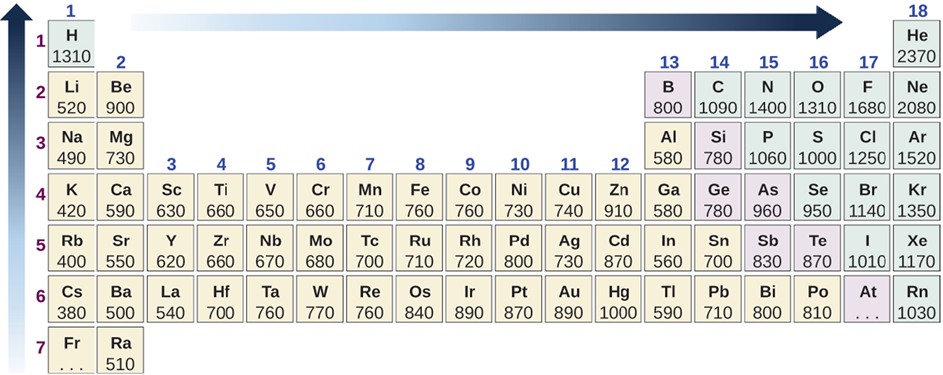
- The ionization energy generally decreases from top to bottom in a column because the valence electrons are further away and experience less pull to the nucleus down the column.
- The ionization energy generally increases from left to right in a row because the valence electrons are in the same shell while more protons add to the nucleus, which increases the pull on the valence electrons.
Fig. 2.6.4 illustrates the periodic trend in the ionization energy.
Electronegativity
Electronegativity is the ability of an atom in a compound to attract the bonding electron pair to itself. Electropositivity is the opposite of electronegativity. Electronegativity is a property of an atom in a compound, i.e., a bonded atom, not a feature of an individual atom.
There are several electronegativity scales. The most commonly used is the Pauling electronegativity scale. Fig. 2.6.5 shows the electronegativities values on Pauling’s electronegativity scale.
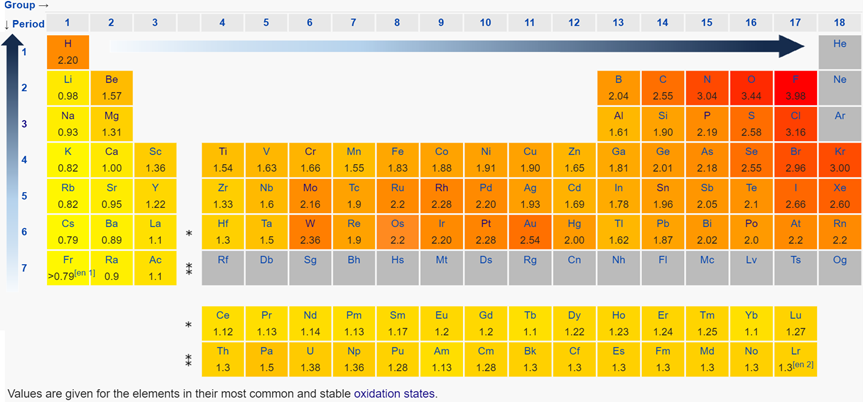
- The electronegativity generally decreases from top to bottom in a column because the atomic size increases down the column making the nucleus less effective in pulling the bonding electrons.
- The electronegative generally increases from left to right in a row because the atomic size decrease from left to right in a row, making the nucleus more effective in pulling the bonding electron pair to itself.
Metallic character
The metallic character relates to the ease of losing an electron in a chemical reaction. The metallic character trend is opposite to the trend of ionization energy.
- The metallic character generally increases from top to bottom in a column because the atomic size increases down the column, making the valence electrons less tightly held and easier to remove.
- The metallic character generally decreases from left to right in a row because the atomic size decreases from left to right, making the valence electrons more tightly held and difficult to remove.
Summary of the periodic trends
The ionization energy and the electronegativity generally increase from left to right in a row and from bottom to top in a column. The atomic size and the metallic character are opposite, i.e., they increase from right to left in a row and from top to bottom in a column. Fig. 2.6.6 summarizes the periodic trend in the properties of the elements.