17.7: Calorimetry
- Page ID
- 53875
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)At one time, calories in foods were measured with a bomb calorimeter. A weighed amount of the food would be placed in the calorimeter and the system was then sealed and filled with oxygen. An electric spark ignited the food-oxygen mixture. The amount of heat released when the food burned gave an idea of the calories present within the food. Today, calories are calculated from the protein, carbohydrate, and fat content of food (all determined by chemical analysis).
Calorimetry
Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. A calorimeter is an insulated container that is used to measure heat changes. The majority of reactions that can be analyzed in a calorimetry experiment are either liquids or aqueous solutions. A frequently used and inexpensive calorimeter is a set of nested foam cups fitted with a lid to limit the heat exchange between the liquid in the cup and the air in the surroundings (see figure below). In a typical calorimetry experiment, specific volumes of the reactants are dispensed into separate containers and the temperature of each is measured. They are then mixed into the calorimeter, which starts the reaction. The reactant mixture is stirred until the reaction is complete, while the temperature of the reaction is continuously monitored.
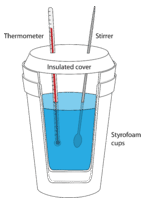
The key to all calorimetry experiments is the assumption that there is no heat exchange between the insulated calorimeter and the room. Consider the case of a reaction taking place between aqueous reactants: the water in which the solids have been dissolved is the surroundings, while the dissolved substances are the system. The temperature change that is measured is the temperature change that is occurring in the surroundings. If the temperature of the water increases as the reaction occurs, the reaction is exothermic. Heat was released by the system into the surrounding water. An endothermic reaction absorbs heat from the surroundings, so the temperature of the water decreases as heat leaves the surroundings to enter the system.
The temperature change of the water is measured in the experiment and the specific heat of water can be used to calculate the heat absorbed by the surroundings \(\left( q_\text{surr} \right)\).
\[q_\text{surr} = m \times c_p \times \Delta T\nonumber \]
In the equation, \(m\) is the mass of the water, \(c_p\) is the specific heat of the water, and \(\Delta T\) is \(T_f - T_i\). The heat absorbed by the surroundings is equal, but opposite in sign, to the heat released by the system. Because the heat change is determined at constant pressure, the heat released by the system \(\left( q_\text{sys} \right)\) is equal to the enthalpy change \(\left( \Delta H \right)\).
\[q_\text{sys} = \Delta H = -q_\text{surr} = - \left( m \times c_p \times \Delta T \right)\nonumber \]
The sign of \(\Delta H\) is positive for an endothermic reaction and negative for an exothermic reaction.
Example \(\PageIndex{1}\)
In an experiment, \(25.0 \: \text{mL}\) of \(1.00 \: \text{M} \: \ce{HCl}\) at \(25.0^\text{o} \text{C}\) is added to \(25.0 \: \text{mL}\) of \(1.00 \: \text{M} \: \ce{NaOH}\) at \(25.0^\text{o} \text{C}\) in a foam cup calorimeter. A reaction occurs and the temperature rises to \(32.0^\text{o} \text{C}\). Calculate the enthalpy change \(\left( \Delta H \right)\) in \(\text{kJ}\) for this reaction. Assume the densities of the solutions are \(1.00 \: \text{g/mL}\) and that their specific heat is the same as that of water.
Solution
Step 1: List the known quantities and plan the problem.
Known
- \(c_p = 4.18 \: \text{J/g}^\text{o} \text{C}\)
- \(V_\text{final} = 25.0 \: \text{mL} + 25.0 \: \text{mL} = 50.0 \: \text{mL}\)
- \(\Delta T = 32.0^\text{o} \text{C} - 25.0^\text{o} \text{C} = 7.0^\text{o} \text{C}\)
- Density \(= 1.00 \: \text{g/mL}\)
Unknown
The volume and density can be used to find the mass of the solution after mixing. Then calculate the change in enthalpy by using \(\Delta H = q_\text{sys} = -q_\text{surr} = - \left( m \times c_p \times \Delta T \right)\).
Step 2: Solve.
\[\begin{align*} m &= 50.0 \: \text{mL} \times \frac{1.00 \: \text{g}}{\text{mL}} = 50.0 \: \text{g} \\ \Delta H &= - \left( m \times c_p \times \Delta T \right) = - \left( 50.0 \: \text{g} \times 4.18 \: \text{J/g}^\text{o} \text{C} \times 7.0^\text{o} \text{C} \right) = -1463 \: \text{J} = -1.5 \: \text{kJ} \end{align*}\nonumber \]
Step 3: Think about the result.
The enthalpy change is negative because the reaction releases heat to the surroundings, resulting in an increase in the temperature of the water.
Summary
- Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process.
- A calorimeter is an insulated container that is used to measure heat changes.
- Calculations involving enthalpy changes are illustrated.

