Chemistry of Cadmium
- Page ID
- 633
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This module is created to provide information about Cadmium, which can be very helpful to know as this transition metal is poisonous and can harm those who do not handle it with caution.
Introduction
Cadmium, a transition metal, has the chemical symbol of Cd. Cadmium is located in the d-block and 12 group of the periodic table possessing an atomic number of 48 and an atomic mass of 112.411g. Cadmium was discovered in Germany in 1817, by a German scientist, Friedrich Strohmeyer. This element was very commonly used as a sacrificial anode to protect iron and steel from corroding and is also used in nickel-cadmium batteries. Cadmium is highly toxic so it should be used and handled with great caution.
General Properties of Cadmium
- Chemical Symbol: Cd
- Atomic Number: 48
- Atomic Mass: 112.411
- Group Number: 12
- Period Number: 5
- Electronegetivity: 1.69
- Standard State: Solid
- Standard Color: Silvery Grey Metallic
- Density: 8.65 g/cm3
- Melting Point: 320.9 oC
- Boiling Point: 767oC
- Most common Oxidation State: +2
- Heat of Fusion: 6.192 kJ mol-1
- Heat of Vaporization: 99.87 kJ mol-1
Atomic Structure
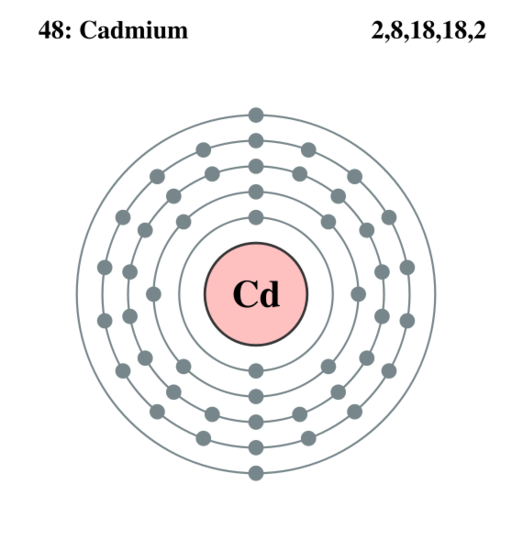
Electron Configuration of Cadmium
Cd: 1s22s22p63s23p64s23d104p65s2 4d10 or [Kr] 4d105s2
Common Isotopes
Cadmium has a total of eight naturally occuring isotopes. These are 106Cd, 108Cd, 110Cd, 111Cd, 112Cd, 113Cd,114Cd, 116Cd. Of these eight istopes, 114Cd and 112Cd are the most common with an abundance of 29% and 24% respectively.
Metallic Characteristics
Cadmium metal is soft and malleable when pure and is used mostly for electroplating steel (corrosion resistance) and in the manufacture of bearings. Cadmium compounds are also found in oil paints where they provide some brilliant colors (e.g., "cadmium yellow"). In its solid state, this metal is a silvery grey color. Cadmium is a very good electricity conductor, as well as very resistance to corrosion, making it an excellent sacrificial anode for metals such as iron and steel.
Like zinc and tin, cadmium metal "cries" or emits a high pitched sound when bent rapidly. Most cadmium today is recovered as a by-product of the zinc refining process.
Natural Occurrences
Cadmium never occurs in nature in its elemental form. It is always found in a compound with another element. The most common natural compounds of Cadmium are Cadmium Sulfide, Cadmium Carbonate, and Carbon Oxide. Carbon Sulfide, also known as the mineral Greenockite, is the most well known source of Cadmium. Cadmium is also found in the Earth's Crust in amounts of about 0.1 to .2 ppm, a very smallamount. Most of the Cadmium that is obtained is through a process known as Zinc refinement, which uses their differing melting points.
Common Reactions with Cadmium
Cadmium forms several important compounds in nature:
- Cadmium Hydroxide (Cd(OH)2): This compound is seen in the Nickel-Cadmium battery and forms precipitate in the presense of a Nitric Acid solution.
- Cadmium Oxide (CdO): This compound in used in a variety of reactions including acting as a catalyst in redox reactions, hydrogenation reactions, polymerization and cleavage. Cadmium Oxide is heat resistant so it can be very useful in heat-resistant plastics, as well as other coatings for plastic. This compound is all seen in certain enamels and batteries.
- Cadmium Sulfate (CdSO4): This compound, like many other compounds of Cadmium is used in electroplating, pigments, fluorescents, and batteries, specifically the Weston Cell.
These three compounds have the most significant use in society; however, a few other compounds that exist in nature are Cadmium Cyanide, Cadmium Chloride, Cadmium Carbonate, and Cadmium Nitrate.
Common Uses
Cadmium's s most known to be used in Nicad, or Nickel-Cadium rechargeble batteries, which is the second most widely used battery, seen in airplanes, telephones, power tools, cd players, computers, and even radar stations. In this battery, Cadmium takes the form of Cadmium hydroxide. Cadmium is also used in coatings, pigments, plastics, and in alloys. The most commonly formed alloys are with silver, copper, and zinc mainly because Cadmium lowers the melting point allowing them to be suitable for the hard soldering of metals. In past years, Cadmium was used in electroplating of steel to protect from corrosion; however, that is not as common.
Hazards and Toxicity
Cadmium is a very toxic metal, therefore needs to be handled with great caution. Cadmium can enter the human body through smoking cigarettes, contaminated water due to landfills, certain foods such as shellfish, and mostly through handling the metal itself. This metal when inhaled or digested can cause many health affects, including vomiting, nausea, diarrhea, dry throat, and headaches. More serious affects are still being investigated, but is thought to cause high blood pressure, cancer, heart and liver disease.
References
- Schulte-Schrepping, K.-H. and Piscator, M. 2000. Cadmium and Cadmium Compounds. Ullmann's Encyclopedia of Industrial Chemistry.Print.
- Newton, David E. "Cadmium." Chemical Elements: From Carbon to Krypton. Vol. 1. Detroit: U.X.L., 1999. 81-86. Print.
- Young, Jay A. "Cadmium." Journal of Chemical Education 2005 82 (4), 521. Print.
Problems
- What is the electron configuration of Cd2+? Draw the orbital filled diagram for this ion.
- What is an example of a balanced chemical reaction for the formation of Cadmium hydroxide? Don't forget to include phases.
- Electroplating was a common use of Cadmium, explain what electroplating means and how is it beneficial?
- Explain the term of an isotope in regards to Cadmium.
- What are the characteristics of a metal? Does Cadmium show these characteristics?
Solutions to Problems
- 1s22s22p63s23p64s23d104p64d10
- Cd(NO3)2 + 2 NaOH → Cd(OH)2 + 2 NaNO3
- Electroplating is the using of one metal to fully cover or plate another metal to protect it from corrosion. For example, the use of Cadmium to electroplate steel to protect it from corroding.
- An isotope is a naturally occuring form of Cadmium where there is a differing numbers of neutrons, however, the same number of protons.
- The characteristics of a metal include good conductors of heat and electricity, tendency to lose electrons, high melting points, are ductile and malleable. Cadmium does posses these characteristics and is a good example of a metal.

