Halide Ions as Reducing Agents
- Page ID
- 3700
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page examines the redox reactions involving halide ions and concentrated sulfuric acid, using these reactions to discuss the trend in reducing ability of the ions from fluoride to iodide. Two types of reactions might occur when concentrated sulfuric acid is added to a solid ionic halide like sodium fluoride, chloride, bromide or iodide. The concentrated sulfuric acid can act as both an acid and an oxidizing agent.
Concentrated sulfuric acid acting as an acid
Concentrated sulfuric acid transfers a proton to the halide ion to produce a gaseous hydrogen halide, which immediately escapes from the system. If the hydrogen halide is exposed to moist air, steam fumes are formed. For example, concentrated sulfuric acid reacts with solid sodium chloride at low temperatures to produce hydrogen chloride and sodium bisulfate, as in the following equation:
\[ NaCl + H_2SO_4 \rightarrow HCl + NaHSO_4 \nonumber \]
All the halide ions behave similarly.
Concentrated sulfuric acid acting as an oxidizing agent
With fluoride or chloride
Concentrated sulfuric acid is not a strong enough oxidizing agent to oxidize fluoride or chloride. In those cases, only the steamy fumes of the hydrogen halide—hydrogen fluoride or hydrogen chloride—are produced. In terms of the halide ions, fluoride and chloride are not strong enough reducing agents to reduce the sulfuric acid. This is not the case for bromides and iodides.
With bromide
Bromide is a strong enough reducing agent to reduce sulfuric acid. Bromide is oxidized to bromine in the process, as in the half-equation below:
\[ 2Br^- \rightarrow Br_2 + 2e^- \nonumber \]
Bromide reduces sulfuric acid to sulfur dioxide gas, decreasing the oxidation state of sulfur from +6 to +4. The half-equation for this transition is as follows:
\[ H_2SO_4 + 2H^+ + 2e^- \rightarrow SO_2 + 2H_2O \nonumber \]
These two half-equations can be combined into the overall ionic equation for the reaction:
\[ H_2SO_4 + 2H^+ + 2Br^- \rightarrow Br_2 +SO_2 +2H_2O \nonumber \]
In practice, this reaction is confirmed by the steamy fumes of hydrogen bromide contaminated with the brown color of bromine vapor. The sulfur dioxide is a colorless gas, so its presence cannot be directly observed.
With Iodide
Iodide is a stronger reducing agent than bromide, and it is oxidized to iodine by the sulfuric acid:
\[ 2I^- \rightarrow I_2 +2e^- \nonumber \]
The reduction of the sulfuric acid is more complicated than with bromide. Iodide is powerful enough to reduce it in three steps:
- sulfuric acid to sulfur dioxide (sulfur oxidation state = +4)
- sulfur dioxide to elemental sulfur (oxidation state = 0)
- sulfur to hydrogen sulfide (sulfur oxidation state = -2).
The most abundant product is hydrogen sulfide. The half-equation for its formation is as follows:
\[H_2SO_4 + 8H^+ + 8e^- \rightarrow H_2S + 4H_2O \nonumber \]
Combining these two half-equations gives the following net ionic equation:
\[ H_2SO_4 + 8H^+ + 8I^- \rightarrow 4I_2 + H_2S + 4H_2O \nonumber \]
This is confirmed by a trace of steamy fumes of hydrogen iodide and a large amount of iodine. The reaction is exothermic: purple iodine vapor is formed, with dark gray solid iodine condensing around the top of the reaction vessel. There is also a red color where the iodine comes into contact with solid iodide salts. The red color is due to the I3- ion formed by reaction between I2 molecules and I- ions. Hydrogen sulfide gas can be detected by its "rotten egg" smell, but this gas is intensely poisonous.
- Fluoride and chloride cannot reduce concentrated sulfuric acid.
- Bromide reduces sulfuric acid to sulfur dioxide. In the process, bromide ions are oxidized to bromine.
- Iodide reduces sulfuric acid to a mixture of products including hydrogen sulfide. Iodide ions are oxidized to iodine.
- The reducing ability of halide ions increases down the group.
Explaining the trend
An over-simplified explanation
The following explanation is only (partially) accurate if fluoride is neglected. When a halide ion acts as a reducing agent, it transfers electrons to something else. That means that the halide ion itself loses electrons. The larger the halide ion, the farther the outer electrons are from the nucleus, and the more they are shielded by inner electrons. It therefore gets easier for the halide ions to lose electrons down the group because there is less attraction between the outer electrons and the nucleus. This argument seems valid, but it is flawed. The energetics of the change must be examined.
A more detailed explanation
Enthalpy change variation between halogens
The amount of heat evolved or absorbed when a solid halide (like sodium chloride) is converted into an elemental halogen must be considered. Taking sodium chloride as an example, the following energetic quantities are important:
- The energy required to break the attractions between the ions in the sodium chloride (the lattice enthalpy).
- The energy required to remove the electron from the chloride ion. This is the reverse of the electron affinity of the chlorine (the electron affinity can be acquired from a data table and negated).
- The energy recovered when the chlorine atoms convert to diatomic chlorine. Energy is released when these bonds are formed. Chlorine is simple because it is a gas. In bromine and iodine, heat is also released during condensation to a liquid or a solid, respectively. To account for this, it is simpler to think in terms of atomization energy rather than bond energy. The value of interest is the reverse of atomization energy.
Atomization energy is the energy needed to produce 1 mole of isolated gaseous atoms starting from an element in its standard state (gas for chlorine, and liquid for bromine, for example - both of them as X2). The figure below shows how this information fits together:
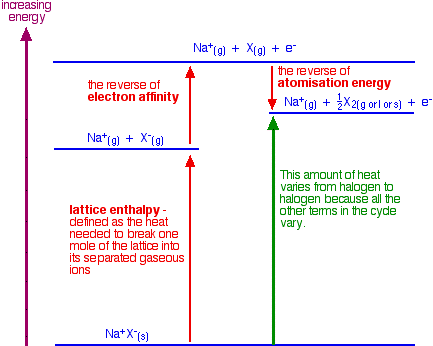
The enthalpy change shown by the green arrow in the diagram for each of the halogens must be compared. The diagram shows that the overall change involving the halide ions is endothermic (the green arrow is pointing up toward a higher energy).
This is not the total enthalpy change for the whole reaction. Heat is emitted when the changes involving the sulfuric acid occur. That is the same irrespective of the halogen in question. The total enthalpy change is the sum of the enthalpy changes for the halide ion half-reaction and the sulfuric acid half-reaction.
The table below shows the energy changes that vary from halogen to halogen. The process is assumed to start from the solid sodium halide. The values for the lattice enthalpies for other solid halides would be different, but the pattern would be the same.
| heat needed to break up NaX lattice (kJ mol-1) |
heat needed to remove electron from halide ion (kJ mol-1) |
heat released in forming halogen molecules (kJ mol-1) |
sum of these (kJ mol-1) |
|
|---|---|---|---|---|
| F | +902 | +328 | -79 | +1151 |
| Cl | +771 | +349 | -121 | +999 |
| Br | +733 | +324 | -112 | +945 |
| I | +684 | +295 | -107 | +872 |
The overall enthalpy change for the halide half-reaction:
The sum of the enthalpy changes, in the final column, is decreasingly endothermic down the group. The total change in enthalpy (including the sulfuric acid) is also less positive.
The amount of heat produced in the half-reaction involving the sulfuric acid must be great enough to make the reactions with the bromide or iodide feasible, but not enough to compensate for the more positive values produced by the fluoride and chloride half-reactions.
Exploring the changes in the various energy terms
In this section, the individual energy terms in the table that are most important in making the halogen half-reaction less endothermic down the group are determined.
Chlorine to iodine
From chlorine to iodine, the lattice enthalpy changes most, decreasing by 87 kJ mol-1. By contrast, the energy required to remove the electron decreases by only 54 kJ mol-1. Both of these terms matter, but the decrease in lattice enthalpy is the more significant. This quantity decreases because the ions are getting larger. That means that they are farther away from each other, and so the attractions between positive and negative ions in the solid lattice are lessened.
The simplified explanation mentioned earlier is misleading because it concentrates on the less-important decrease in the amount of energy needed to remove the electron from the ion.
Fluorine
Fluoride ions are very difficult to oxidize to fluorine. The table above shows that this has nothing to do with the amount of energy required to remove an electron from a fluoride ion. It actually takes less energy to remove an electron from a fluoride ion than from a chloride ion. The generalization that an electron becomes easier to remove as the ion becomes larger does not apply here.
Fluoride ions are so small that the electrons experience strong repulsion from each other. This outweighs the effect of their closeness to the nucleus and makes them easier to remove than the simplified argument predicts.
There are two important reasons why fluoride ions are so difficult to oxidize. The first is the comparatively high lattice enthalpy of the solid fluoride. This is due to the small size of the fluoride ion, which means that the positive and negative ions are very close together and therefore strongly attracted to each other. The other factor is the small amount of heat that is released when the fluorine atoms combine to make fluorine molecules (see the table above). This is due to the low bond enthalpy of the F-F bond. The reason for this low bond enthalpy is discussed on a separate page.
What if the halide ions were in solution rather than in a solid?
This discussion has focused on the energetics of the process starting from solid halide ions because that is the standard procedure when using concentrated sulfuric acid. Halides could also be oxidized in solution with another oxidizing agent.
The trend is exactly the same. Fluoride is difficult to oxidize and it becomes easier down the group toward iodide; in other words, fluoride ions are not good reducing agents, but iodide ions are.
The explanation starts from the hydrated ions in solution rather than solid ions. In a sense, this has already been done on another page. Fluorine is a very powerful oxidizing agent because it very readily forms its negative ion in solution. It is therefore energetically difficult to reverse the process. By contrast, for energetic reasons, iodine is relatively reluctant to form its negative ion in solution. Therefore, it is relatively easy for it to revert back to iodine.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


