8.2: Bioinorganic chemistry
- Page ID
- 125421
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Many biological reactions are known to involve metal ions. There are also metals recognized as essential elements, although their roles in living organisms are not clear. Bioinorganic chemistry, the study of the functions of metals in biological systems using the knowledge and methods of inorganic chemistry, has progressed remarkably in recent years.
The following list shows typical bioactive substances containing metals.
- Electron carriers. Fe: cytochrome, iron-sulfur protein. Cu : blue copper protein.
- Metal storage compound. Fe : ferritin, transferrin. Zn : metallothionein.
- Oxygen transportation agent. Fe: hemoglobin, myoglobin. Cu: hemocyanin.
- Photosynthesis. Mg: chlorophyll.
- Hydrolase. Zn: carboxypeptidase. Mg: aminopeptidase.
- Oxidoreductase. Fe: oxygenase, hydrogenase. Fe, Mo: nitrogenase.
- Isomerase. Fe: aconitase. Co: vitamin B12 coenzyme.
The basis of chemical reactions of metalloenzymes are
- Coordinative activation (coordination form, electronic donating, steric effect),
- Redox (metal oxidation state),
- Information communication, and, in many cases, reaction environments are regulated by biopolymers such as proteins, and selective reactions are performed.
Examples of actions of metals other than by metalloenzymes include
- Mg: MgATP energy transfer
- Na/K ion pumping,
- Ca: transfer of hormone functions, muscle contraction, nerve transfer, blood coagulation, are some of the important roles of metals.
(a) Oxidation
Oxidation reactions in living systems are fundamental to life, and many studies of these systems have been performed. In particular, the mechanisms of oxygen gas transportation by hemoglobin and mono-oxygen oxidation by the iron porphyrin compounds named P-450 have been studied at length. Oxygen gas transportation, which has been studied for many years, is described below. Iron porphyrins hemoglobin and myoglobin and the copper compound hemocyanin are involved in the transportation of oxygen gas in air to cells in living organisms. The basis of this function is reversible bonding and dissociation of dioxygen to iron or copper ions. In order to perform these functions, metals must be in oxidation states and coordination environments suitable for the reversible coordination of dioxygen. The iron porphyrin compound hemoglobin is found in red bloods of human beings and other animals.
Hemoglobin has the structure of heme iron with four iron porphyrin units combined with a globin protein. Dioxygen is transported in blood by being coordinated to ferrous ions in the hem iron unit. The Fe (II) ion is penta-coordinate with four nitrogen atoms of porphyrin and a nitrogen atom of the polypeptide histidine, and becomes hexa-coordinate when a dioxygen coordinates to it. The spin state of Fe (II) changes from high spin to low spin upon the coordination of dioxygen. The high spin Fe(II) is above the plane of porphyrin because it is too large to fit in the available space. When the Fe(II) ion becomes low spin upon dioxygen coordination, the size of the iron ion decreases and it just fits into the hole of the porphyrin molecule.
This molecular-level movement has attracted interest as an allosteric effect because it affects the whole protein through the histidine coordinate bond and governs the specific bond of a dioxygen molecule. Oxidation of the Fe(II) ion of a hem molecule is prevented by a macromolecular protein, and if the hem iron is taken out of the prote in, Fe(II) ion is oxidized to Fe(III), and two porphyrin rings are bridged by a peroxide \(\mu\)-O22-, which finally changes to a bridging \(\mu\)-O2-structure.
When the hem is in this state, it loses the ability to coordinate to the dioxygen molecule. Based on this phenomenon, a synthetic porphyrin that is able reversibly to coordinate to a dioxygen by suppressing dimerization of the iron porphyrin has been developed, and was named the picket fence porphyrin after its three dimensional form.
(b) Nitrogen fixation
The reaction which converts the nitrogen in air into ammonia is basic to all life. Nitrogen fixation, the reaction to fix atmospheric nitrogen to form ammonia, is carried out by Rhizobium in the roots of legumes or by bacteria in algae in an anaerobic atmosphere. All animals and plants, including mankind, were depended on biological nitrogen fixation as a source of nitrogen for protein and other compounds containing nitrogen before the invention of the Harber-Bosch process.
\[N_{2} + 8 H^{+} + 8 e^{-} + 16 MgATP \rightarrow 2 NH_{3} + H_{2} + 16 MgADP + 16 Pi \quad \text{(where Pi is an inorganic phosphate)} \nonumber \]
An enzyme named nitrogenase catalyzes this reaction. Nitrogenase contains iron-sulfur and iron-molybdenum sulfur proteins, and reduces dinitrogen by coordination and cooperative proton and electron transfers, while using MgATP as an energy source. Because of the importance of this reaction, attempts to clarify the structure of nitrogenase and to develop artificial catalysts for nitrogen fixation have continued for many years. Recently, the structure of an active center in nitrogenase called iron-molybdenum cofactor was clarified by single crystal X-ray analysis (Figure \(\PageIndex{2}\)). According to this analysis, its structure has Fe3MoS4 and Fe4S4 clusters connected through S.
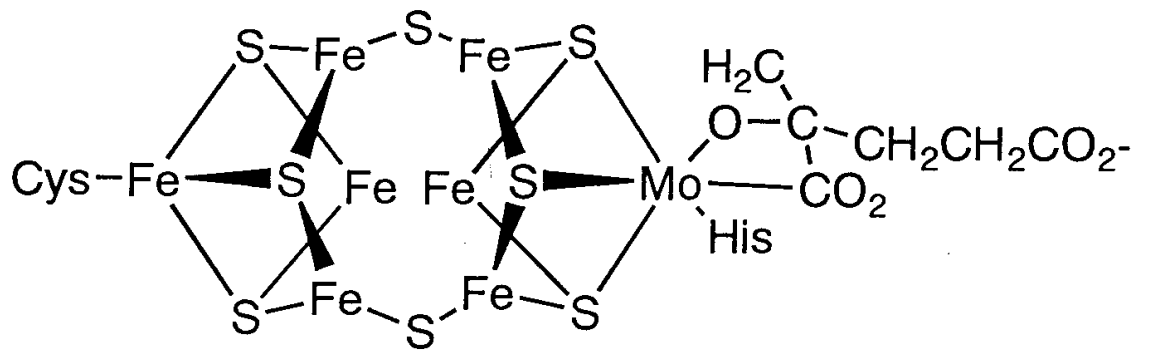
It is believed that dinitrogen is activated by coordination between the two clusters. On the other hand, the portion called P cluster consists of two Fe4S4 clusters. The roles and reaction mechanism of both parts are not yet clear.
(c) Photosynthesis
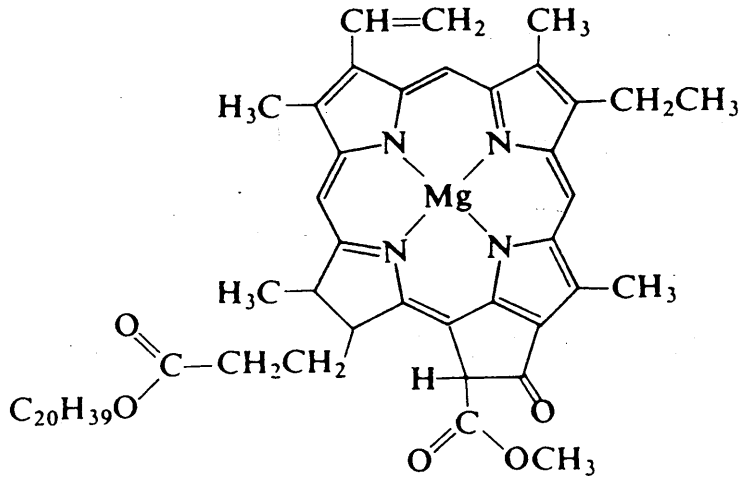
The formation of glucose and dioxygen by the reaction of carbon dioxide and water is a skillful reaction using photoenergy and in which chlorophyll (Figure \(\PageIndex{3}\)), which is a magnesium porphyrin and a manganese cluster complex, plays the central role. A chloroplast contains photosystem I (PSI) and photosystem II (PSII), which use light energy to reduce carbon dioxide and to oxidize water.
Chlorophyll is a fundamental component of PSI. Chlorophyll is a porphyrin complex of magnesium and is responsible for the green colors of leaves. It plays an important role in receiving light energy and transferring it to redox reaction systems. Chlorophyll is excited from the singlet ground state to the singlet excited state by light, the energy of the excited state is transferred to an acceptor within 10 ps, and the resultant energy reduces an iron-sulfur complex and is finally used for reduction of carbon dioxide in subsequent dark reactions. Since charge separation by photochemical excitation is the most important first stage, studies on photoinduced electron transfer are have been actively performed using various kinds of porphyrin compounds as models of chlorophylls. PSI, which obtains oxidizing energy by electron transfer, converts ADP to ATP.
On the other hand, the oxidized form of PSII oxidizes water through a chain of redox reactions of oxo cluster complexes of manganese, and generates oxygen. Since four electrons shift in the reduction of Mn(IV) to Mn (II) in this reaction, at least two manganese species are involved. Probably, a cluster complex which contains two Mn(II) and two Mn(IV) species mediates the electron transfer via four step reactions. However, the details of this reaction are as yet unclear because it is very difficult to isolate this cluster and to analyze its structure. The electron transfer stage is being studied at present by using various manganese complexes as model systems.
Photosynthesis is a very interesting research theme in bioinorganic chemistry as it involves a few metal ions, a porphyrin, sulfide and oxide clusters that constitute a cycle of subtle electron transfer and redox reactions, and generate oxygen gas by photolysis of water and produce carbohydrates from carbon dioxide by reductive dark reactions,. Recently, the reaction center of a photosynthetic bacteria was crystallized and J. Deisenhofer and his colleagues won a Nobel prize for its structural analysis (1988).
Give examples of small molecules that are fundamentally important for living things.
- Answer
-
- H2O
- O2
- N2
- CO2


