9.1: Prelude to Coordination Chemistry I - Structure and Isomers
- Page ID
- 374474
What is a coordination compound?
Coordination compounds consist of one or more metals bound to one or more Lewis base ligands. For example, hexamineruthenium (3+) ion is a coordination complex in which six ammonia ligands coordinate a Co3+ ion, as shown in Scheme \(\sf{\PageIndex{1}}\).
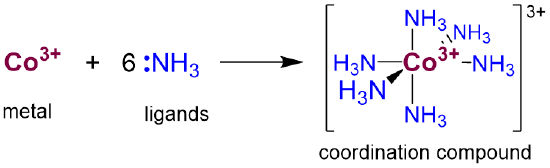
Such complexes are called coordination complexes because the ligand-metal bond may be thought of as a coordinate covalent bond in which both of the bonding electrons come from the ligand, which is then said to coordinate the metal. This coordinate covalent model is a very useful formalism for understanding the basic features of coordination chemistry, although it does not always accurately reflect the actual details of the bonding in every coordination complex. Nevertheless, even in those cases where the simple coordinate covalent bond model breaks down, the coordinate covalent bond concept supplies the language sophisticated models employ to describe the more complex bonding involved.
Additional important terms
Some of the common widely used terms that follow from the coordinate covalent model of bonding in coordination complexes include:
- Coordination compounds are also called coordination complexes, metal complexes, or just complexes. The term complex refers to coordination compounds' composite nature, in that they may be thought of as comprising multiple ligand and metal ion parts that can be restored by breaking the coordinate covalent bonds holding the complex together. This is in contrast to inorganic or organic molecules which are more commonly thought about as whole molecules held together by the sharing of electrons contributed by all the atoms.
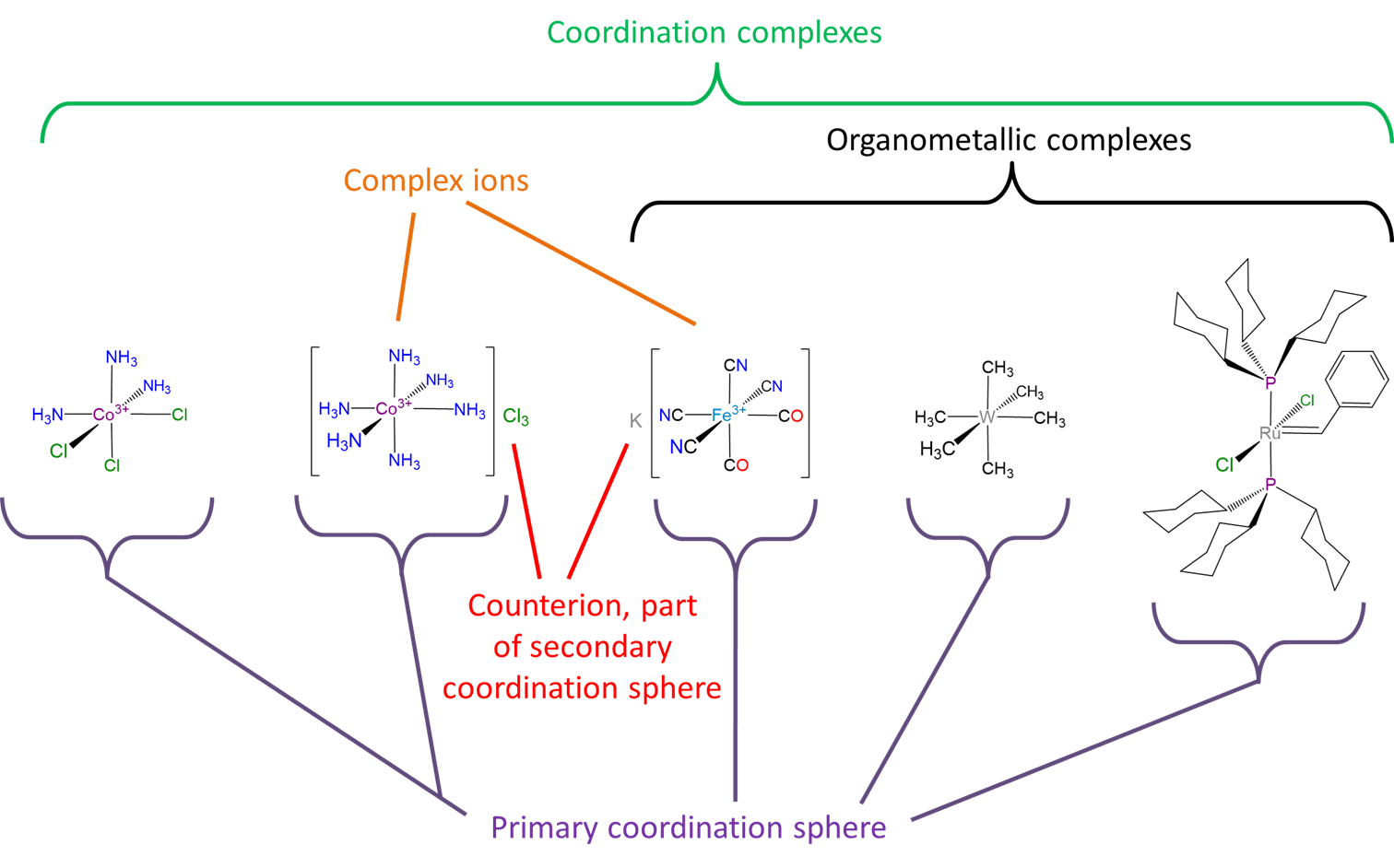
- Coordination complexes that are ions are called complex ions.
- Ligands bound to the coordination complex are said to reside in the primary or inner coordination sphere. These bound ligands are not readily exchangeable, in contrast to nearby counterions and solvent molecules, which are said to reside in the secondary or outer coordination sphere.
- The portion of the complex contributing the electron pairs is said to be the donor and the portion which receives them the acceptor. In conventional coordination compounds the ligand is the donor and the metal the acceptor. In these cases it would be equally convenient to refer to the ligand donor as the Lewis base and the metal acceptor as the Lewis acid. However, in more complex bonding scenarios there may be multiple electron pair donation and acceptance interactions taking place between each pair of atoms and donor-acceptor language will be more convenient.
- The number of ligand sites donating lone pairs to the central atom is referred to as the coordination number. For most complexes this will just be equal to the number of ligand atoms bound to the metal. In simple complexes it is just equal to the number of ligands. For instance, the cobalt in Scheme \(\sf{\PageIndex{1}}\) has a coordination number of six.
- Although technically compounds with metal-carbon bonds are coordination complexes, the term coordination complex is sometimes used to refer to complexes which do not possess metal-carbon bonds in their primary coordination sphere. Complexes which possess metal-carbon bonds are called organometallic compounds instead. The use of the terms organometallic and coordination to distinguish organometallic compounds from other types of coordination compounds is often convenient since many organometallic ligands engage in more than simple \(\sigma\) donor-acceptor coordinate covalent bond formation with the metal center. However, this is true of some wholly inorganic ligands too, so it should always be kept in mind that organometallic compounds are just a type of coordination compound and that inorganic ligands can in principle be tuned to interact with a metal center in much the same way an organic ligand does.
A summary of some of the concepts and terms used to describe coordination compounds is given in Scheme \(\sf{\PageIndex{2}}\).
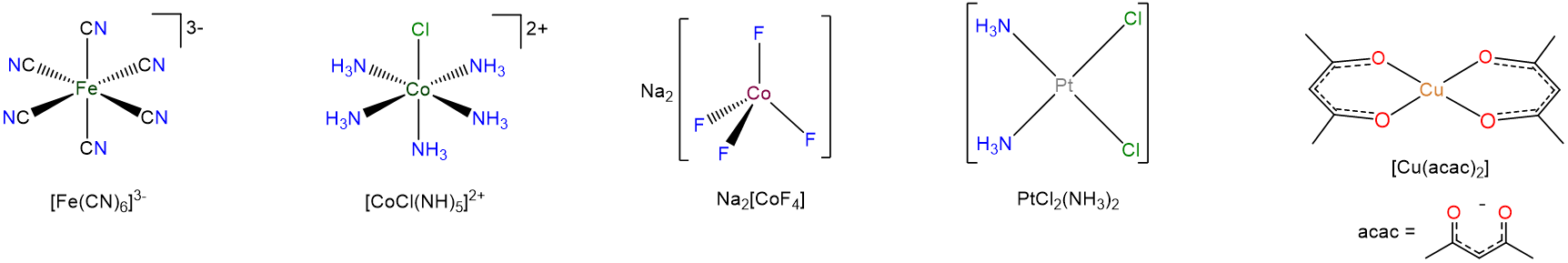
The formulae of coordination complexes
The way the formulae of coordination complexes are written reflects the fact that it is often convenient to think of coordination compounds as a composite of metals and ligands. When writing the formula of a complex
- the atoms of a ligand are not added to those of the rest of the compound. Instead, the ligand atoms are kept together, if necessary by enclosing the ligand formula in parentheses or giving an abbreviation for the ligand.
- For complex ions the metal and ligands are enclosed in square brackets. Sometimes this is also done for neutral coordination compounds as well. In either case the brackets enclose those parts of the compound which comprise the primary coordination sphere; anything else is in the secondary coordination sphere outside.
A careful perusal of the examples in Scheme \(\sf{\PageIndex{3}}\) should make the important features of this system clear.