8.4: Oxides and Oxoacids
- Page ID
- 212661
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Oxides of Nitrogen
A summary of the physical properties of the oxides of nitrogen is given in Table \(\PageIndex{1}\).
| Oxide | Formula | Mp (°C) | Bp (°C) |
| Nitrous oxide | N2O | -90.8 | -88.5 |
| Nitric oxide | NO | -163.6 | -161.8 |
| Dinitrogen trioxide | N2O3 | -100.6 | 3.5 (dec.) |
| Nitrogen dioxide (dinitrogen tetroxide) | NO2/N2O4 | -11.2 (NO2) | 21.2 (N2O4) |
| Nitrogen pentoxide | N2O5 | 30 | 47 |
Nitrous oxide
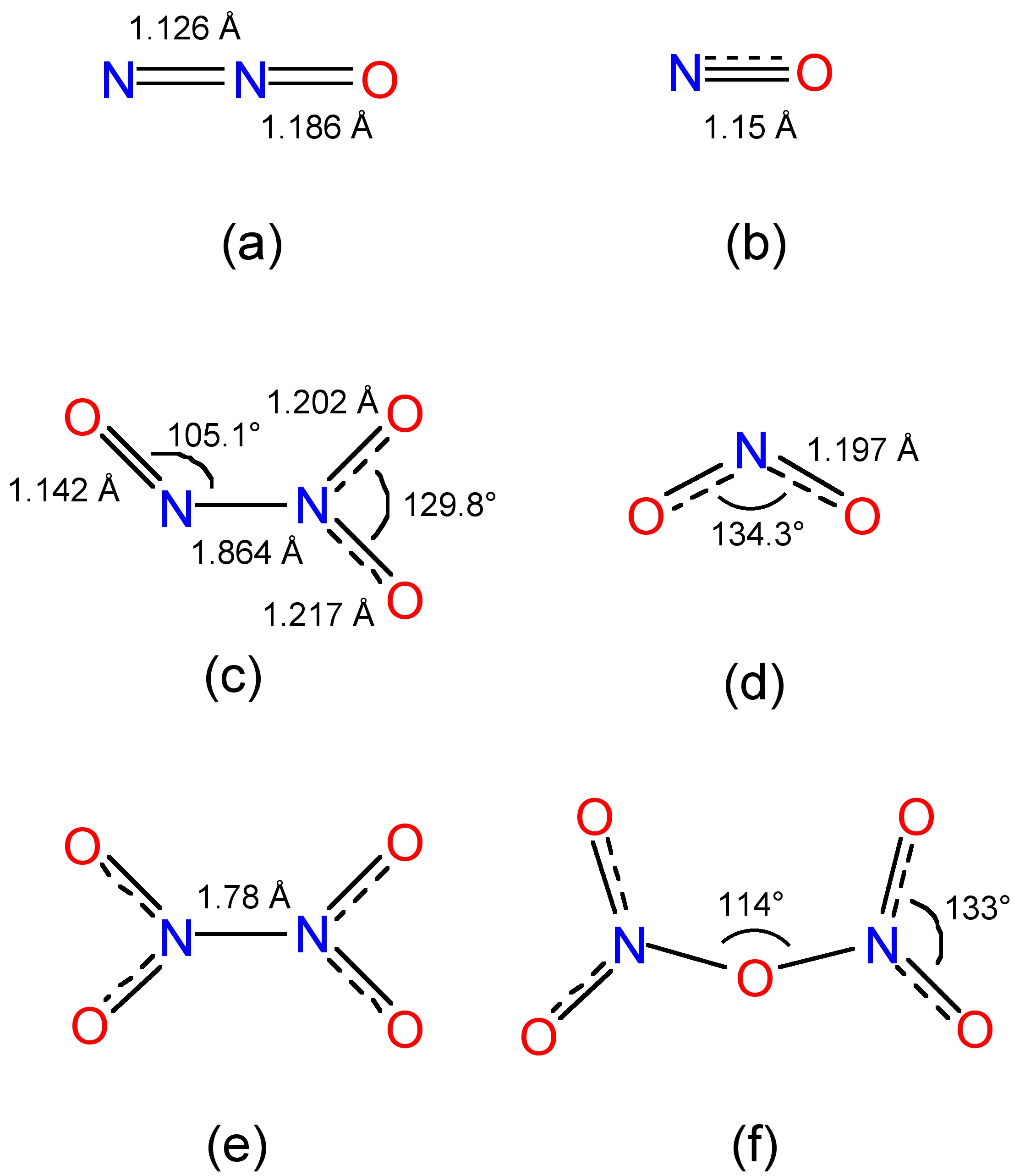
Gaseous nitrous oxide (N2O) is prepared by the careful thermal decomposition of ammonium nitrate (NH4NO2), (8.4.1). Nitrous oxide is a linear molecule (Figure \(\PageIndex{1}\)a) that is isoelectronic (and isostructural) with carbon dioxide. Despite its use as a power enhancement for automobiles, nitrous oxide is actually not very reactive and a major use is as an aerosol propellant.
\[ \rm NH_4NO_2 \xrightarrow{\Delta} N_2O + 2 H_2O \]
Nitrous oxide as an anesthetic drug
Nitrous oxide is known as "laughing gas" due to the euphoric effects of inhaling it, a property that has led to its recreational use as a hallucinogen. However, it is as a anesthetic that it has a legitimate application.
The first use of nitrous oxide as anesthetic drug was when dentist Horace Wells (Figure \(\PageIndex{2}\)) with assistance by Gardner Quincy Colton (Figure \(\PageIndex{3}\)) and John Mankey Riggs (Figure \(\PageIndex{4}\)), demonstrated insensitivity to pain from a dental extraction in December 1844. Wells subsequently treated 12-15 patients, and according to his own record it only failed as an anesthetic in two cases. In spite of these results, the method was not immediately adopted, probably because during his first public demonstration was only partly successful.

The method did not come into general use until 1863, when Colton successfully used it for more than 25,000 patients. As such, the usage of nitrous oxide rapidly became the preferred anesthetic method in dentistry. Because the gas is mild enough to keep a patient in a conscious and conversational state, and yet in most cases strong enough to suppress the pain caused by dental work, it remains the preferred gas anesthetic in today's dentistry.
Nitrous: the secret to more power.
In motorsports, nitrous oxide (often referred to as nitrous or NOS) allows the engine to burn more fuel, resulting in a more powerful combustion, and hence greater horsepower. The gas itself is not flammable, but it delivers more oxygen (33%) than atmospheric air (21%) by breaking down at elevated temperatures. When N2O breaks down in during fuel combustion, the decomposition of nitrous is exothermic, contributing to the overall power increase.
Nitrous oxide is stored as a compressed liquid (Figure \(\PageIndex{5}\)); the evaporation and expansion of liquid nitrous oxide in the intake manifold causes a large drop in intake charge temperature, resulting in a denser charge, further allowing more air/fuel mixture to enter the cylinder. Nitrous oxide is sometimes injected into (or prior to) the intake manifold, whereas other systems directly inject right before the cylinder (direct port injection) to increase power.
One of the major problems of using nitrous oxide in a reciprocating engine is that it can produce enough power to damage or destroy the engine. Very large power increases are possible, and if the mechanical structure of the engine is not properly reinforced, the engine may be severely damaged or destroyed during this kind of operation.
Nitric oxide
Nitric oxide (NO) is formed by the high temperature oxidation of nitrogen, (8.4.2), or the platinum catalyzed oxidation of ammonia at 800 °C, (8.4.3).
\[ \rm N_2 + O_2 \rightleftharpoons 2 NO\]
\[ \rm 4 NH_3 + 5 O_2 \xrightarrow{Pt} 4 NO + 6 H_2O \]
Nitric oxide (Figure \(\PageIndex{6}\)b) is electronically equivalent to dinitrogen (N2) plus an electron, and as a consequence it is paramagnetic with one unpaired electron. The location of the unpaired electron in the π* orbital (Figure \(\PageIndex{6}\)a) results in a bond order of 2.5 rather than the triple bond observed for N2 (Figure \(\PageIndex{6}\)b). The N-O distance of 1.15 Å is intermediate between the triple bond distance in NO+ (1.06 Å) and the typical double bond distance (ca. 1.20 Å). Furthermore, because of the location of the electron it is easy to oxidize nitric oxide to the nitrosonium ion (NO+), (8.4.4).
\[ \rm NO \rightarrow NO^+ + e^- \]
\[ \rm 2 NO + O_2 \rightarrow 2 NO_2\]
Nitric oxide is unstable to heat, (8.4.6), and oxidation, (8.4.5). It will also react with halogens to form the nitrosyl halides, XNO.
\[ \rm 3 NO \rightarrow N_2O + NO_2\]
Dinitrogen trioxide
Dinitrogen trioxide (N2O3) is formed from the stoichiometric reaction between NO and O2 or NO and N2O4. Dinitrogen trioxide has an intense blue color in the liquid phase and a pale blue color in the solid state. Thermal dissociation of N2O3, (8.4.7), occurs above -30 °C, and some self-ionization of the pure liquid is observed, (8.4.8). The asymmetric structure of N2O3 (Figure \(\PageIndex{1}\)c) results in a polar molecule (Figure \(\PageIndex{7}\)).
\[ \rm N_2O_3 \rightleftharpoons NO + NO_2 \]
\[ \rm N_2O_3 \rightleftharpoons NO^+ + NO_2^-\]

Nitrogen dioxide (and tetroxide)
Formed from the oxidation of nitric oxide, (8.4.9), brown nitrogen dioxide is actually in equilibrium with its colorless dimeric form, nitrogen tetroxide (N2O4), (8.4.10).
\[ \rm 2 NO + O_2 \rightleftharpoons 2 NO_2\]
\[ \rm \underset{brown\\paramagnetic}{2NO_2} \rightleftharpoons \underset{colorless\\diamagnetic}{N_2O_4}\]
Nitrogen dioxide (Figure \(\PageIndex{1}\)d) is electronically equivalent to the nitrate anion (NO2-) less one electron, and as such it is paramagnetic with one unpaired electron. The location of the unpaired electron in a nitrogen sp2 orbital, and a consequently it forms a dimer through a N-N bond (Figure \(\PageIndex{1}\)e). Furthermore, it is easy to oxidize nitrogen dioxide to the nitronium ion (NO2+), (8.4.11).
\[ \rm NO_2 \rightarrow NO_2^+ + e^-\]
Nitrogen dioxide dissolves in water to form a mixture of nitric and nitrous acids, (8.4.12). Nitrogen dioxide acts as an oxidizing agent with the formation of nitrate anion, (8.4.13).
\[ \rm 2 NO_2 + H_2O \rightarrow NO_3^- + NO_2^- + 2H^+\]
\[ \rm 2 NO_2 + 2 I^- \rightarrow I_2 + 2 NO_2^-\]
The most common structural form of N2O4 (Figure \(\PageIndex{1}\)e) is planar with a long N-N bond (1.78 Å) that is significantly longer than observed in hydrazine (1.47 Å). Rationalization of this structural effect is obtained from a consideration of the molecular orbitals, which show that the electrons in the σ-bond are actually delocalized over the whole molecule. The rotation about the N-N bond is 9.6 kJ/mol.
Nitrogen pentoxide
The dehydration of nitric acid, with P2O5, yields nitrogen pentoxide, (8.4.14), which is an unstable solid at room temperature (Table \(\PageIndex{1}\)). In the solid state nitrogen pentoxide is actually nitronium nitrate (NO2+NO3-), however, in the vapor phase it exists as a molecular species (Figure \(\PageIndex{1}\)f) with a bent N-O-N unit. Nitrogen pentoxide is a very powerful nitrating and oxidation agent.
\[ \rm 2 HNO_3 \rightleftharpoons N_2O_5 + H_2O \]
Nitrogen oxides as precursors to smog and acid rain
Nitrogen oxides (NOx) emissions are estimated to be in the range of 25 - 100 megatonnes of nitrogen per year. Natural sources are thought to make up approximately 1/3 of the total. The generations of NOx (primarily a mixture of NO2 and NO) is the main source of smog and a significant contribution to atmospheric pollution; however, NOx is also responsible for much of the acidity in acid rain.
Atmospheric reactions leading to acid rain
In the dry atmosphere, nitric oxide reacts is oxidized rapidly in sunlight by ozone, (8.4.15). The nitrogen dioxide reacts with the hydroxide radical, formed by the photochemical decomposition of ozone, (8.4.16) and (8.4.17), in the presence of a non-reactive gas molecule such as nitrogen to form nitric acid vapor, (8.4.18). The conversion rate for NOx to HNO3 is approximately ten times faster than the equivalent reaction for sulfur dioxide. For example, conversion is essentially complete for a NOx plume by the time it transverses the North Sea from the UK to Scandinavia.
\[ \rm NO + O_2 \rightarrow NO_2 + O_2 \]
\[ \rm O_3 + h\nu \rightarrow O* + O_2\]
\[ \rm O* + H_2O \rightarrow 2HO \cdot\]
\[ \rm NO_2 + \cdot OH + N_2 \rightarrow HNO_3 + N_2 \]
At night, conversion takes place via the formation of a nitrate radical, (8.4.19), which subsequently photochemically unstable under sunlight forming nitrogen pentoxide, (8.4.20), that reacts with water on the surface of aerosol particles to form nitric acid, (8.4.21).
\[ \rm NO_2 + O_3 \rightarrow NO_3 + O_2 \]
\[ \rm NO_2 + NO_3 + N_3 \rightarrow N_2O_5 + N_2 \]
\[ \rm N_2O_5 + H_2O \rightarrow 2 HNO_3\]
Both NO and NO2 are only slightly soluble in water and it is therefore more probable that the nitric acid content of rain is more likely due to the dissolution of nitric acid vapor into raindrops, (8.4.22), rather than a separate reaction.
\[ \rm N_2O_5 + H_2O_{(l)} \rightarrow 2 HNO_{3(aq)}\]
Oxoacids of Nitrogen
Nitrous acid
In the gas phase nitrous acid can be made by the following reaction:
\[ \rm NO + NO_2 + H_2O \rightleftharpoons 2 HNO_2\]
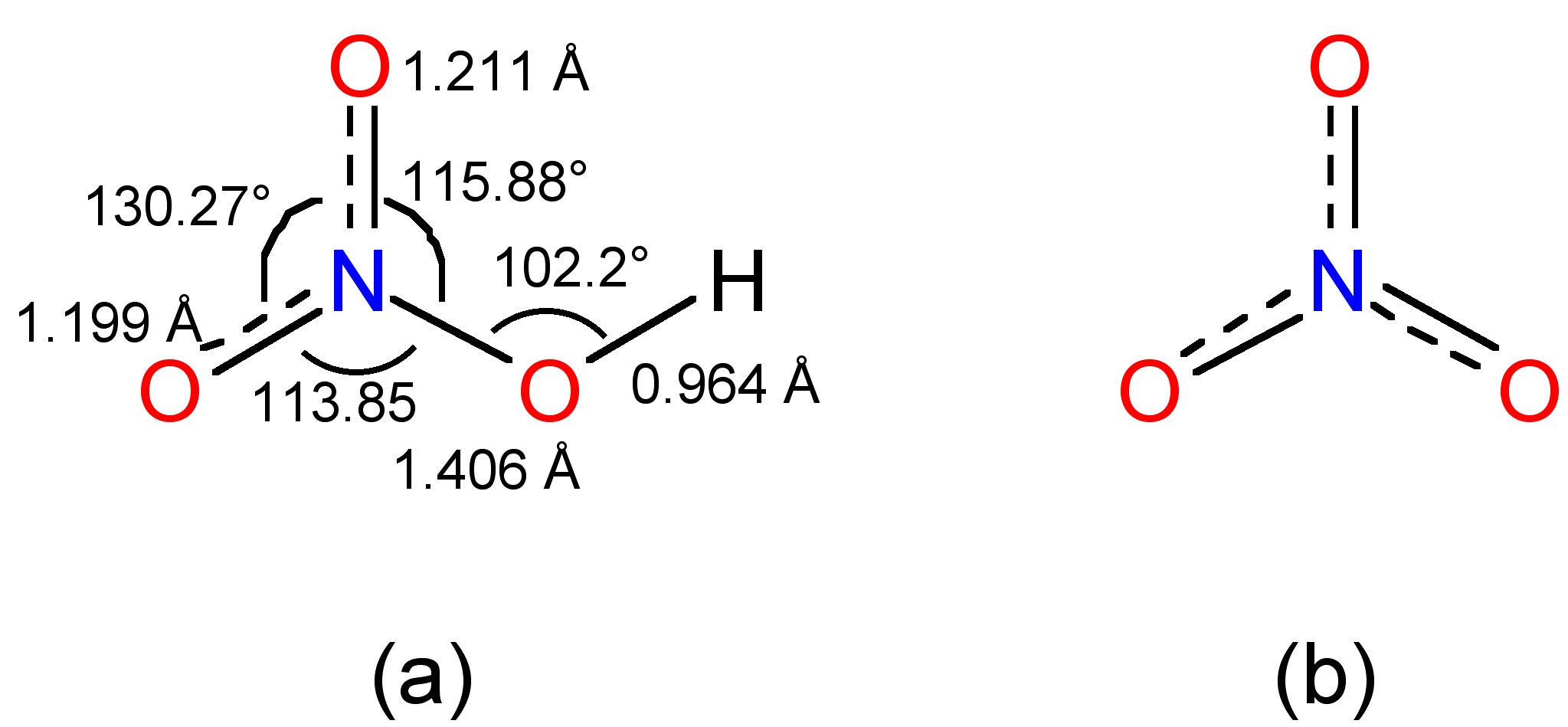
The gas phase structure as determined by IR spectroscopy is shown in Figure \(\PageIndex{8}\)a, in which the nitrogen is planar with sp2 hybridization.
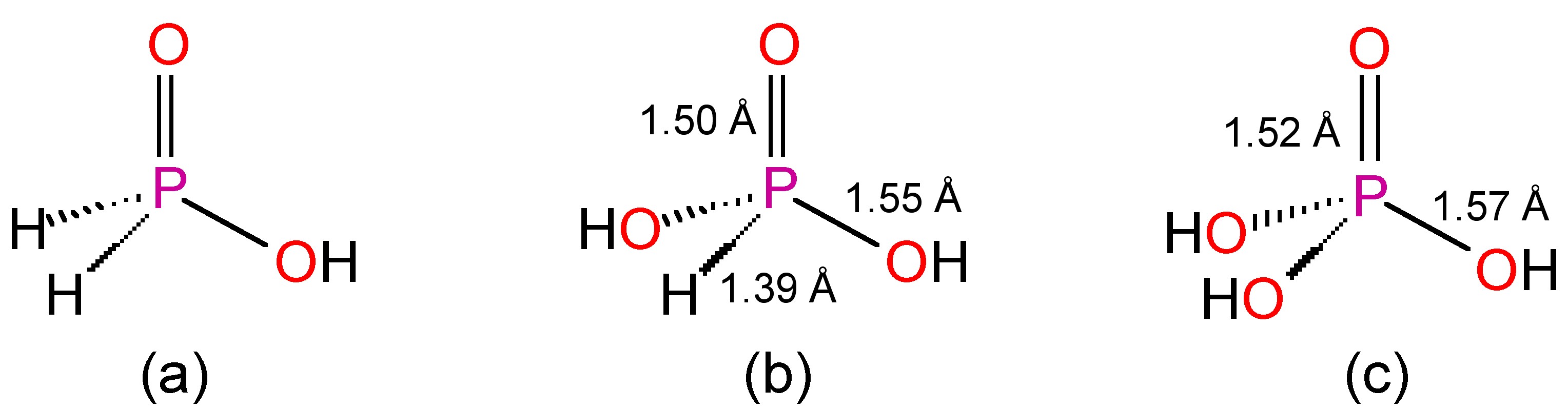 Figure \(\PageIndex{8}\)
Figure \(\PageIndex{8}\)In basic aqueous solution the same reaction results in the formation of the nitrite ion, (8.4.24), which can be precipitated as the Ba2+ salt. After separation the addition to sulfuric acid yields a solution of nitrous acid. However, it is not possible to concentrate by heating since decomposition occurs, (8.4.25).
\[ \rm NO + NO_2 + 2 OH^- \rightleftharpoons 2 NO_2^- + H_2O\]
\[ \rm 3 HNO_2 \rightarrow H_3O^+ + NO_3^- + 2 NO\]
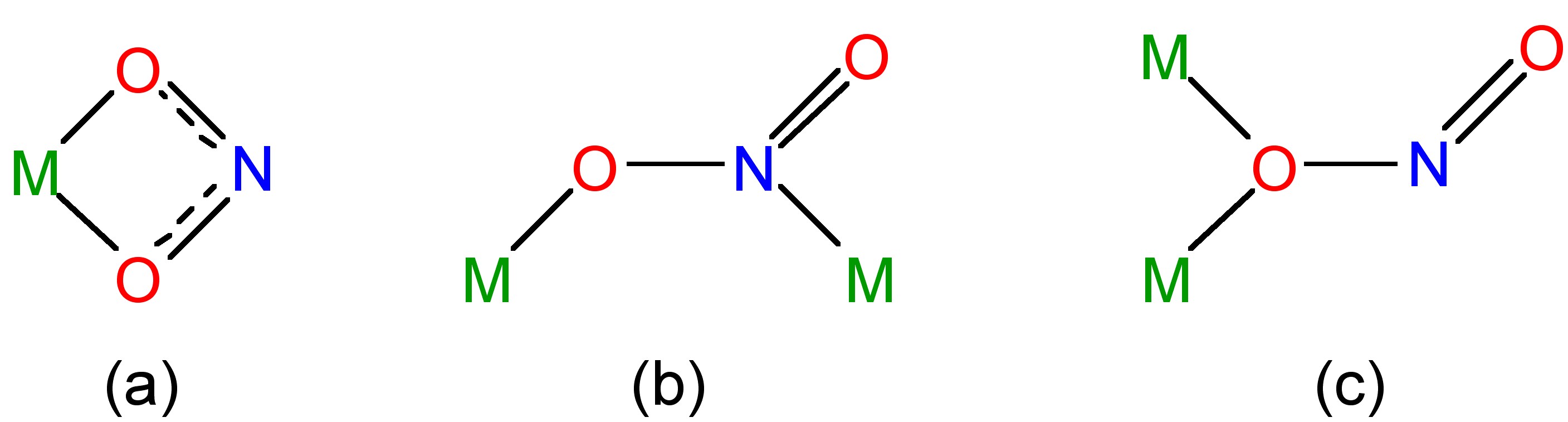
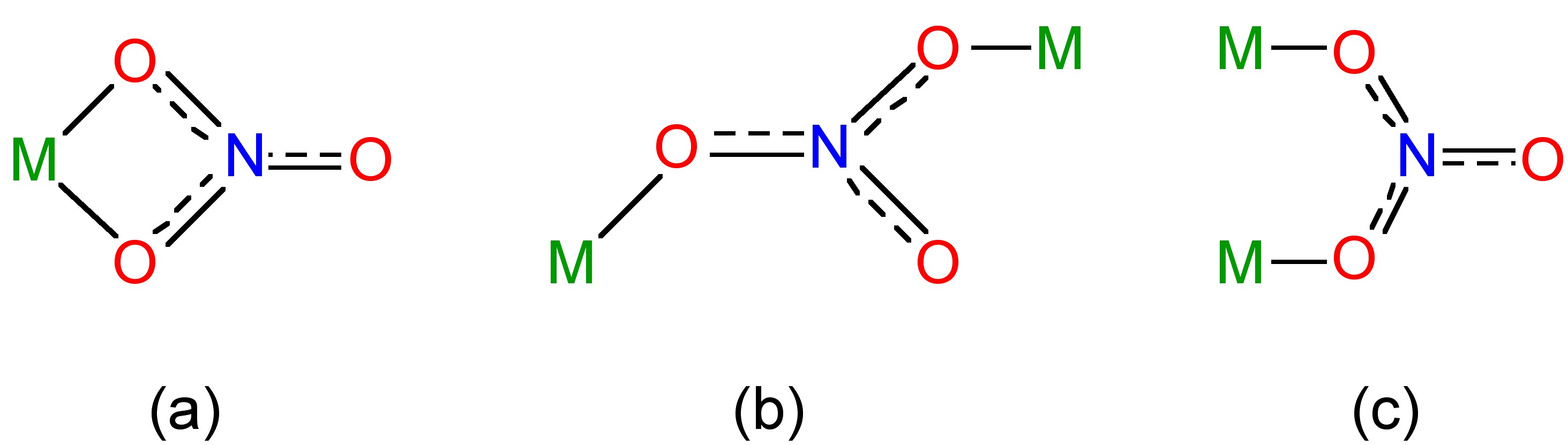
Nitrous acid is a fairly weak acid in water (pK = 5.22); however, many salts are known of the nitrite ion, NO2- (Figure \(\PageIndex{8}\)b). Complexes of the nitrite ion can be monodentate with bonding via nitrogen (nitro) or an oxygen (nitrito). Both isomers can be isolated in the case of an inert metal, i.e., substitutionally inert d6 octahedral complexes (Figure \(\PageIndex{9}\)). Bidentate and bridging modes of coordination are also known for the nitrite anion (Figure \(\PageIndex{10}\)).


Nitrite can act as either an oxidizing agent, (8.4.26), or a reducing agent, (8.4.27).
\[ \rm NO_2^- + 2H^+ + Fe_{2+} \rightarrow NO + H_2O + Fe^{3+}\]
\[ \rm NO_2^- + H_2O + 2 Ce^{4+} \rightarrow NO_3^- + 2 H^+ + 2 Ce^{3+}\]
The most important use of nitrous acid is in the diazotization reactions in which nitrous acid is generated by acidifying nitrite solution.
Nitric acid
Nitric acid (HNO3), also known as aqua fortis and spirit of nitre, is made by the dissolution of nitrogen dioxide in water, (8.4.28). Nitric acid can be concentrated by distillation from concentrated sulfuric acid.
\[ \rm 3 NO_2 + H_2O \rightarrow 2 HNO_3 + NO\]
The structure of HNO3 in the gas phase is planar at the sp2 nitrogen (Figure \(\PageIndex{11}\)a).

Pure 100% nitric acid is a very corrosive liquid that is strongly acidic and protonates and dissolves organic species. In the liquid phase it is slightly dissociated, (8.4.29). It is also a powerful oxidizing agent, converting non-metal elements to either the oxide or oxoacid. In contrast with metals it forms either salts or complexes in which the metal is in its highest oxidation state. It is unstable and decomposes upon heating or photolysis.
\[ \rm 3 HNO_3 \rightleftharpoons H_3O^+ + NO_2^+ + 2 NO_3^-\]
The pure acid has the highest self ionization of pure liquid acids, (8.4.30). However, the loss of water results, (8.4.31), such that the overall reaction can be described by (8.4.32).
\[ \rm 2 HNO_3 \rightleftharpoons H_2NO_3^+ + NO_3^+\]
\[ \rm H_2NO_3^+ \rightleftharpoons H_2O + NO_2^+\]
\[ HNO_3 \rightleftharpoons NO_3^+ + NO_2^+ + H_2O\]
The common concentration of nitric acid is 70%. While the pure acid is colorless, samples often take on a yellow color due to the photochemical decomposition of nitric acid to give brown NO2.
\[ \rm 4 HNO_3 \xrightarrow{h\nu} 4 NO_2 + 2 H_2O + O_2\]
Note
The term the “acid test” is derived from the medieval practice of debasing of gold and silver currencies (often by the Monarchs who issued them) by debasing with copper. With ducats (Milan), livres (France), florins (Florence), maravedies (Spain) and bezants (Constantinople) in widespread use, and each with a nominal gold or silver content, it was important for a merchant to be able verify the worth of any particular coin. If a drop of dilute nitric acid was placed onto a silver coin adulterated with copper, it turned green, due to the formation of copper(II) nitrate. Conversely, if a gold coin reacted in any way with the nitric acid it was not pure. In both cases the coins failed the “acid test”.
Aqua regia (so called because it dissolves gold) is a mixture of 70% nitric acid and hydrochloric acid in a 1:3 ratio. Aqua regia is a very powerful oxidizing agent (it contains Cl2) and stablizes some metals as their chloro complexes (Table \(\PageIndex{2}\)). If HF is added in place of HCl, tantalum may be dissolved with the formation of [TaF2]-.
| Metal | Chloride salt formed |
| Au | [AuCl4]- |
| Pt | [PtCl6]2- |
Fuming nitric acid (100% nitric) is exceedingly corrosive and should not be used. In 100% sulfuric acid, nitric acid acts as a base and gets protonated, (8.4.34) and (8.4.35), and acts as a powerful nitrating agent.
\[ \rm HNO_3 + H_2SO_4 \rightarrow ON(OH)_2^+ + HSO_4^-\]
\[ \rm ON(OH)_2^+ + HSO_4^- + H_2SO_4 \rightarrow H_3O^+ + NO_2^+ + 2 HSO_4^-\]
In water dilute nitric acid is fully ionized, (8.4.36).
\[ \rm HNO_3 + H_2O \rightarrow H_3O^+ + NO_3^-\]
The nitrate ion is planar (Figure \(\PageIndex{11}\)b) and forms many salts and complexes. The nitrate anion is most commonly a monodentate ligand, but can also be a bidentate ligand (Figure \(\PageIndex{12}\)a) or a bridging ligand (Figure \(\PageIndex{12}\)b and 12c).
Phosphorous Oxides
Phosphorous trioxide
The stoichiometric oxidation of white phosphorus yields phosphorous trioxide, (8.4.37).
\[ \rm P_4 + 3 O_2 \rightarrow 2 P_2O_3\]
Several structural forms of phosphorous trioxide are known, but P4O6 is a stable molecule structure (Figure \(\PageIndex{13}\)), while the rest are polymers.

Phosphorous pentoxide
The reaction of white phosphorus with excess oxygen (or the oxidation of phosphorous trioxide) yields phosphorous pentoxide, (8.4.38).
\[ \rm P_4 + 5 O_2 \rightarrow 2 P_2O_5\]
The structure of hexagonal phosphorous pentoxide is actually that of the dimeric form, P4O10, and is based upon the structure of P4O6, but with P=O units instead of lone pairs (Figure \(\PageIndex{14}\)). The structure is maintained in the vapor phase; however, other crystalline and glassy forms comprise of a sheet-like structure (Figure \(\PageIndex{15}\)).


Phosphorous pentoxide is an excellent drying agent below 100 °C. It reacts with water to form various phosphoric acids, and it will extract water from other ‘drying agents’, e.g., (8.4.39) and (8.4.40). Phosphorous pentoxide will dehydrate amides to give nitriles, (8.4.41).
\[ \rm 2 HNO_3 \xrightarrow[-H_2O]{P_4O_{10}}N_2O_5\]
\[ \rm H_2SO_4 \xrightarrow[-H_2O]{P_4O_{10}}SO_3\]
\[ \rm RC(O)NH_2 \xrightarrow[-H_2O]{P_4O_{10}} RC\equiv N\]
Oxoacids of Phosphorus
Phosphorous pentoxide (P2O5) is an excellent drying agent, and its action is a result of the formation of a range of oxoacids.
Hypophosphorous acid
Hypophosphorous acid, H3PO2, is easily prepared pure buy the reaction of white phosphorous with base, followed by acidification, (8.4.42). The pure acid is a solid (Mp = 27 °C) and very soluble in water.
\[ \rm P_4 + 4 OH^- + 4 H_2O \rightarrow \underset{\underset{H_3PO_2}{\downarrow \text{ acidify}}}{4 H_2PO_2^-} + 2 H_2\]
The structure of H3PO2 is determined by X-ray crystallography to be tetrahedral with two hydride ligands and a hydroxide (Figure \(\PageIndex{16}\)a). The presence of two hydrides is confirmed by NMR spectroscopy. The 1H NMR resonance shows an OH line and a doublet from the P-H with a large one-bond coupling constant to the 31P nucleus. The non-decoupled 31P NMR spectrum shows a triplet (δ = 13 ppm, JP-H = 530 Hz) due to the two hydrides.

In water hypophosphorous acid is a monobasic acid (pK = 1.2), (8.4.43), and it forms a wide range of salts. It is also a powerful reducing agent, but its reaction kinetics is slow.
\[ \rm H_2P(O)OH + H_2O \rightleftharpoons H_2P(O)_2^- + H_3O^+\]
Phosphorous acid
The reaction of P4O6 or PCl3 with water yields phosphorous acid, H3PO3; which like hypophosphorous acid is a solid (Mp = 70.1 °C) and very soluble in water. The structure is shown by X-ray crystallography to be comprised of a tetrahedral phosphorus with one hydride and two hydroxides (Figure \(\PageIndex{16}\)b). 31P NMR spectroscopy demonstrates the presence of a single hydride by the presence of a doublet as a consequence of the phosphorous center being split by a single hydride (δ = 4 ppm, JP-H = 700 Hz). The 1H NMR spectrum shows a doublet for the hydride and a single resonance of twice the intensity for the hydroxide.
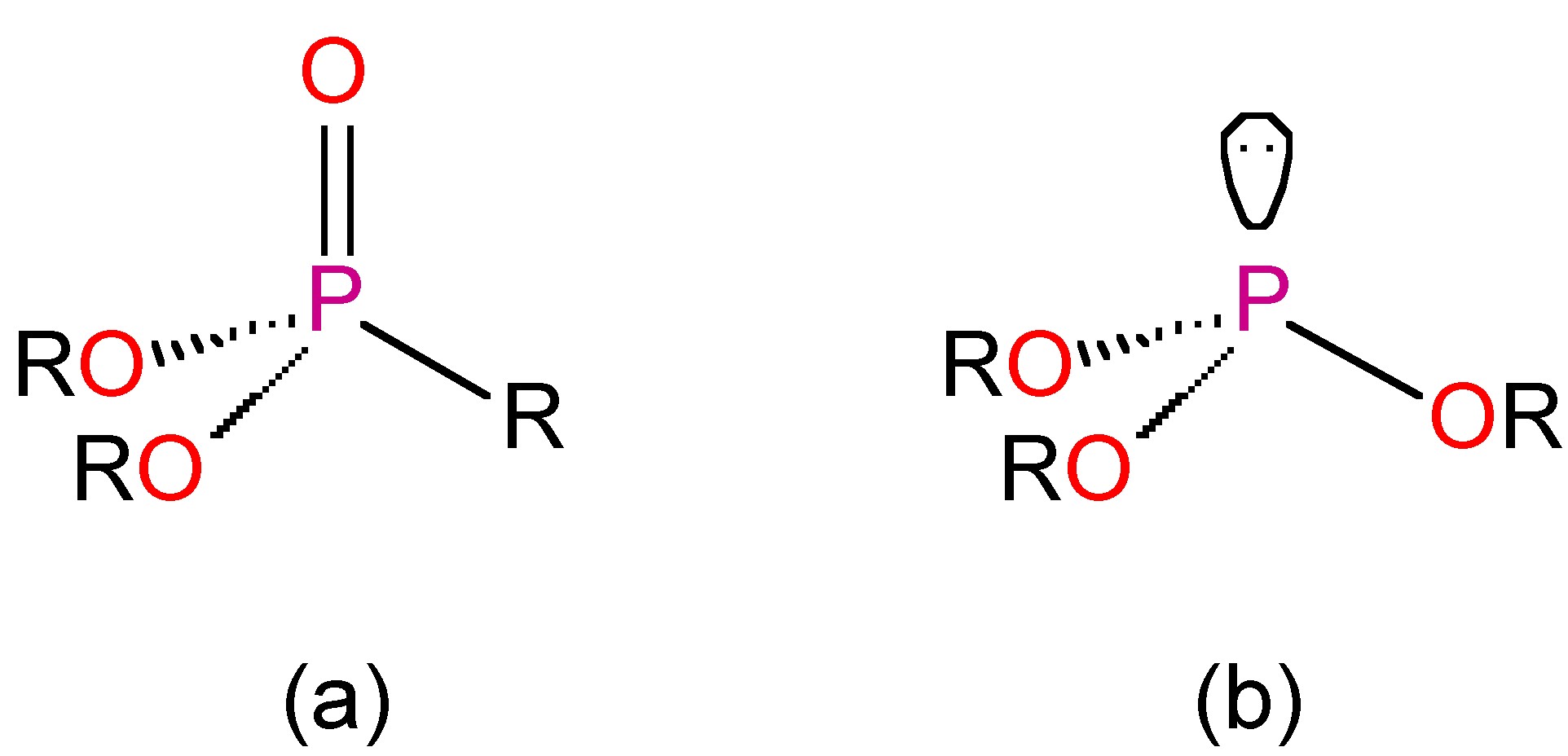
As expected, in water phosphorous acid is dibasic, (8.4.44). The acid (and the anions) are strong reducing agents, yielding phosphoric acid. They actually react very slowly, and it is thought that this may be due to the reaction being in the tautomeric form, P(OH)3. Although this has not been isolated, the trialkyl derivatives exist in both forms, i.e., esters of phosphoric acids (Figure \(\PageIndex{17}\)a) and trialkyl phosphates (Figure \(\PageIndex{17}\)b).
\[ \rm HP(O)(OH)_2 \xrightleftharpoon[pK_1=1.8] [HP(O)_2OH]^- \xrightleftharpoons[pK_2=6.7] [HP(O)_3]^{2-}\]

Ortho phosphoric acid
Orthophosphoric acid (H3PO4) is the most common oxoacid of phosphorus (Figure c). The term acid phosphoric acid is commonly used. It is made from the reaction of phosphates with sulfuric acid (H2SO4), or from the hydrolysis of P4O10. The pure acid is a colorless crystalline solid (Mp = 42.35 °C) with extensive hydrogen bonding. The inter-phosphoric acid hydrogen bonding is partially maintained in aqueous solutions above 50% solutions.
Phosphoric acid is very stable and shows no oxidation chemistry below 350 °C. As expected, phosphoric acid is tribasic, (8.4.45). The anions H2PO4- and HPO42- have particular names, dihydrogen phosphate and monohydrogen phosphate, respectively.
\[ \rm PO(OH)_3 \xrightleftharpoons[pK_1=2.15] [PO_2(OH)_2]^- \xrightleftharpoons[pK_2=7.1] [PO_3(OH)]^{2-} \xrightleftharpoons[pK_3=12.4] [PO_4]^{3-}\]
Many salts of are known for all three anions; those with phosphate (PO42-) are often insoluble in water. Many coordination complexes are known, especially with M3+ and M4+ ions.
Phosphorous esters
The alkyl and aryl homologs of phosphoric acid (i.e., the phosphate triester O=P(OR)3) are prepared by the reaction of P4O10 with the appropriate alcohol. The triesters are good solvents and Lewis basic ligands with coordination via the oxide moiety. The diesters and monoesters can also be made, and they are important in biochemical processes.
Phosphite triesters, P(OR)3, may be made by the reaction of PCl3 with alcohols or phenols in the presence of an organic base as the acceptor for the HCl formed. Alternatively, they can be prepared directly from white phosphorous, (8.4.46).
\[ \rm P_4 + 6 OR^- + 6 CCl_4 + 6 ROH \rightarrow 4 P(OR)_3 + 6 CHCl_3 + 6 Cl^-\]
Phosphite triesters are readily oxidized to the appropriate phosphate triester, (8.4.47). They also react with alkyl halides to form the dialkyl phosphonate via the Michaelis-Arbusov reaction (8.4.48).
\[ \rm 2 P(OR)_3 + O_2 \rightarrow 2 \text{O=P}(OR)_3 \]
\[ \rm P(OR)_3 + R'X \rightarrow \text{O=P}(OR)_2R' + RX\]
Polyphosphates
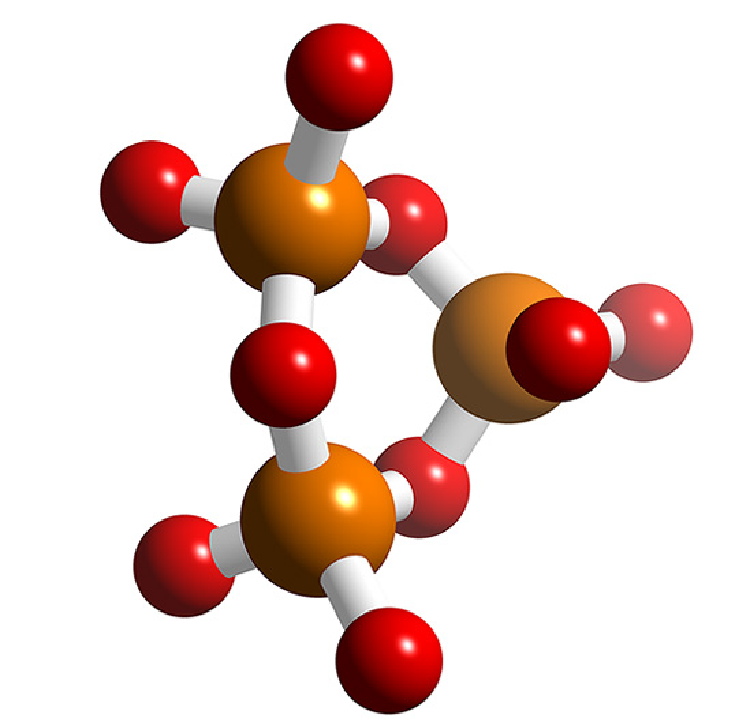
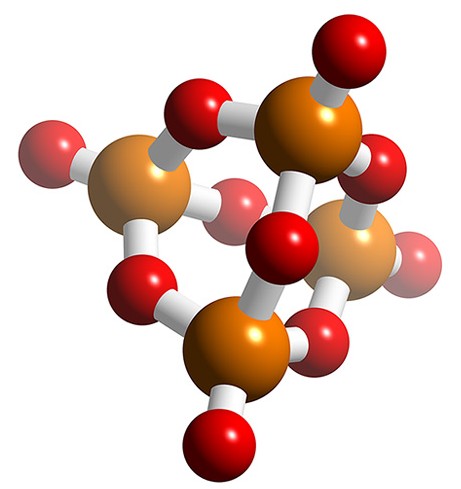
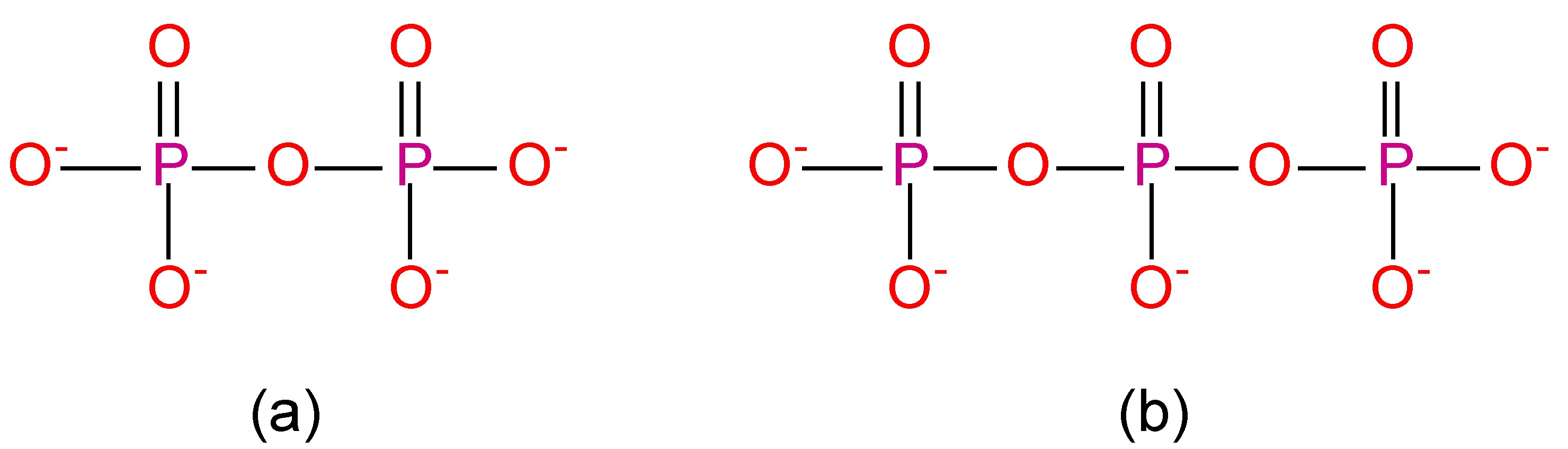
Polyphosphates contain the PO4 unit, and the simplest example (pyrophosphate or diphosphate, (Figure \(\PageIndex{18}\)a) can be considered as condensation products of monohydrogen phosphate, (8.4.49). Longer chains can be formed, e.g., the triphosphate P3O105- (Figure \(\PageIndex{18}\)b). The formation and reverse (hydrolysis) reaction are slow, but are readily catalyzed, e.g., by enzymes.
\[ \rm 2 HPO_4^{2-} \xrightarrow{\Delta} [O_3P\text{-O-}PO_3]^{4-} + H_2O\]
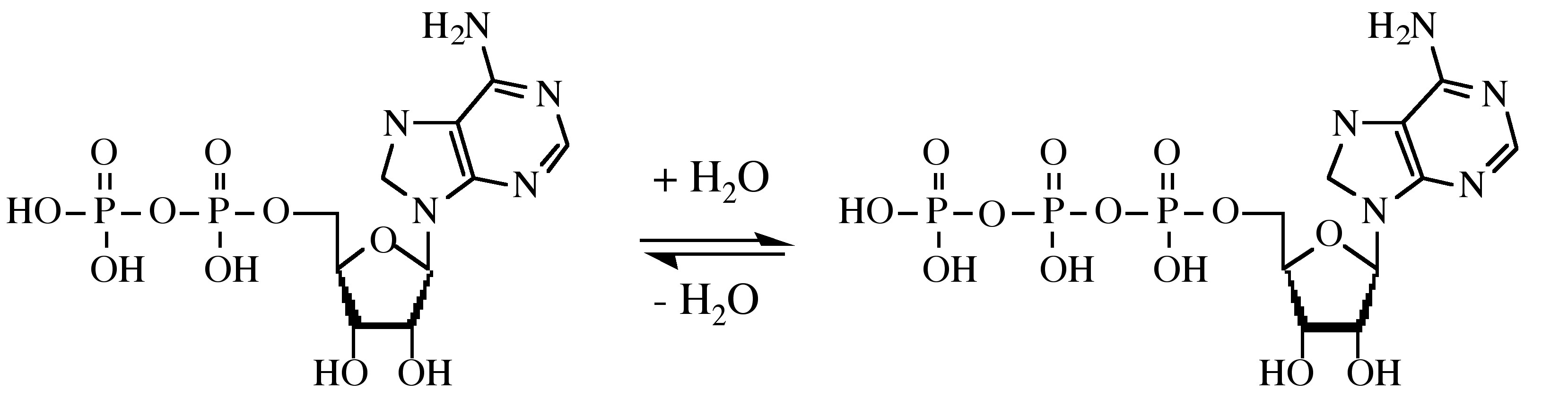
The monoesters of the diphosphate and triphosphate are very important in biological processes. In particular the conversion of the triphosphate ATP (adenosine triphosphate) to the diphosphate ADP (adenosine diphosphate) by the transfer of a phosphate group is important in the energetics of biological reactions.
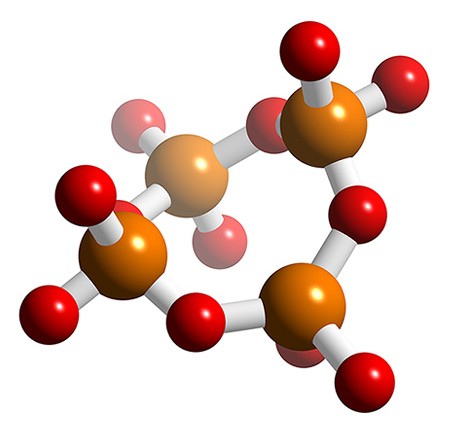
Cyclic polyphosphates
Also known as meta phosphates, cyclic phosphates are also made from fused PO4 units. The simplest, P3O93-, is shown in Figure \(\PageIndex{19}\). Slow addition of water to P4O10 results in the formation of the tetrameric polyphosphate, P4O124- (Figure \(\PageIndex{20}\)), which is known as calgon due to its ability to complex Ca2+ as well as other metals.