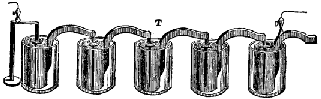16.7: Timeline of Battery Development
- Page ID
- 85099
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Although the development practical batteries largely paralelled the expansion of electrical technology from about the mid-19th century on, it is now thought that a very primitive kind of battery was apparently in use more than 2000 years ago. The brief popularity of electrically powered automobiles in the 1920's encouraged storage battery development. The widespread use of portable "personal" electrical devices has kept the search for better batteries very much alive.
|
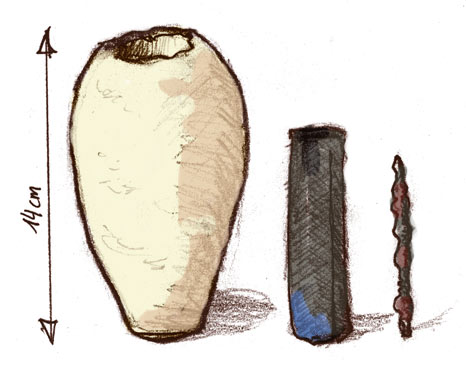
"Baghdad Battery" - 1000 BCE?
Drawing of the three pieces. (CC-BY-SA 2.5; Ironie) |
Earthenware jars containing an iron rod surrounded by a copper cylinder were discovered near Baghdad in 1938. They are believed to have been used by the Parthian civilization that occupied the region about 2000 years ago as a source of electricity to plate gold onto silver. |
|
Allesandro Volta 1782
|
His "Voltaic pile", a stack of zinc and silver disks separated by a wet cloth containing a salt or a weak acid solution, was the first battery known to Western civilization. |
| Sir Humphry Davy 1813 | Davy builds a 2000-plate battery that occupies 889 square feet in the basement of Britain's Royal Society. His earlier batteries provided power for the first public demonstration of electric lighting (carbon arc). |
| Michael Faraday, 1830's | Faraday discovered the fundamentals of galvanic cells and electrolysis that put electrochemistry on a firm scientific basis. |
|
1836 - Daniell cell (also known as a Crow's Foot or Gravity cell.)
|
John Daniell (English chemist and meterologist) developed the first modern storage cell based on Faraday's principles. This consists of a large glass jar with a copper star-shaped electrode in the bottom and a zinc "crow's foot" shaped electrode suspended near the top. The bottom of the jar was filled with a concentrated copper sulfate solution. On top of this was poured dilute sulfuric acid, whose lower density kept it on top. This was the first practical battery to find wide use to power telegraphs and railway signaling systems and home doorbells. |
|
1839 - William Grove (Welsh)
|
Grove was best known in the 19th century for his "nitric acid battery" which came into wide use in early telegraphy.
Now, however, he is most famous for his "gas voltaic battery" in which discovered "reverse electrolysis": the recombination of H2 and O2 following electrolysis of water at platinum electrodes. This was the first demonstration of what we now know as the hydrogen-oxygen fuel cell (see below.) |
|
1859 - Gaston Planté (French)
|
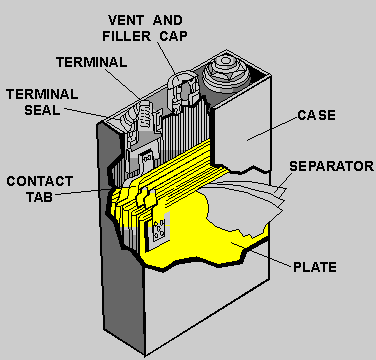
Invents the first lead-acid storage cell which consisted of two sheets of lead separated by a rubber sheet, rolled into a spiral and immersed in dilute sulfuric acid. |
|
1866 - Georges Leclanché (French)
|
By 1868 twenty thousand Leclanché cells were being used in telegraph systems. The original Leclanché cells were built in porous pots which were heavy and subject to breakage. Within twenty years other inventors had modified the design into what we now know as "dry cells" which became widely used in the first flashlights (1909) and in battery-powered radios of the 1920s. |
|
1881 - Faure and others
|
Development of the first practical lead-acid storage cell. The major improvement over Planté's design was the addition of a paste of PbSO4 to the positive plate. |
|
1905 Nickel-iron cell
|
Edison, who was as much a chemist as an all-around inventor, thought that the lead in Planté-type cells made them too heavy, and that having acid in contact with any metal was an inherently bad idea. After much experimentation, he developed a successful alkaline battery. The Edison cell uses an iron anode, nickel oxide cathode, and KOH electrolyte. This cell is extremely rugged and is still used in certain industrial applications, but it was never able to displace the lead-acid cell as Edison had hoped. |
| 1950s | A similar cell, employing a nickel anode instead of iron, was the first rechargeable cell that was small enough to be used in portable consumer devices. Its main disadvantage is that it is ruined by complete discharge. |
| 1949 - Alkaline dry cell - Lew Urry (Eveready Battery Co.) |
First commercial alkaline dry cell. These substitute KOH for the corrosive NH4Cl used in the older dry cells and last 5-8 times longer. |
| 1947 - Mercury cell (Ruben and Mallory, 1950's) |
This was one of the first "button"-type cells which were widely used in cameras and hearing aids. The constancy of the 1.34 v output made them popular for use in sensitive instruments and cardiac pacemakers. The net cell reaction is Zn(s) + HgO(s) → ZnO(s) + Hg(l) Most countries have outlawed sales of these cells in order to reduce mercury contamination of the environment. |
|
Nickel-Cadmium (NiCad) cells
|
The NiCad cell quickly become one of the most popular rechargeable batteries for small consumer devices. They can deliver high current and undergo hundreds of charge/discharge cycles. Because cadmium is an environmental toxin, their use is being discouraged. |
| 1959 - Fuel cell - Francis Bacon (UK) | The first practical fuel cell was developed by British engineer Francis Bacon (1904-1992). This hydrogen-oxygen cell used an alkaline electrolyte and inexpensive nickel electrodes. |
|
Late 1960's - Nickel-metal hydride cells
|
The hydride ion H– would be an ideal cathode material except for the fact that its oxidation product H2 is a gas. The discovery that certain compounds such as LiNi5 and ZrNi2 can act as "hydrogen sponges" made it practical to employ metal hydrides as a cathode material. One peculiarity of Ni-MH cells is that recharging them is an exothermic process, so that proper dissipation of heat must be allowed for. These batteries are widely used in cell phones, computers, and portable power tools. The electrode reactions take place in a concentrated KOH electrolyte: Cathode (+): NiOOH + H2O + e–→ Ni(OH)2 + OH– Anode (-): (1/x) MHx + OH–→ (1/x) M + H2O + e– |
|
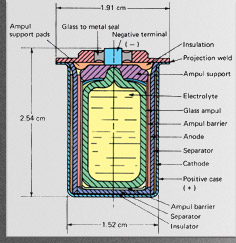
1990s - Lithium cells (Sony Corp.)
|
Lithium is an ideal anode material owing to its low density and high reduction potential, making Li-based cells the most compact ways of storing electrical energy. Lithium cells are used in wristwatches, cardiac pacemakers and digital cameras. Both primary (non-rechargeble) and rechargeable types have been available for some time. More recent applications are in portable power tools and— perhaps most importantly, in electric-powered or hybrid automobiles. Modern lithium cells operate by transporting Li+ ions between electrodes into which the ions can be inserted or intercalated. Cathodes are lithium transition-metal oxides such as LiCoO3, while anodes are lithium-containing carbon, LiC6. The species that undergoes oxidation-reduction is not lithium, but the transition metal, e.g. Co(III)-Co(IV). |
Lithium batteries as incendiary devices
There have been numerous reports of fires and explosions associated with lithium batteries. In 2006, the Dell Corporation had to recall 4.1 million Sony batteries that had been shipped with Dell's laptop computers and were judged to be at risk owing to a manufacturing defect. This illustrates the difficulty of concentrating a large amount of chemical energy into a small package, which is of course the goal of all battery developers eager to meet commercial demands ranging from consumer personal electronics to electrically-powered cars. The fully-charged Li+-deficient lithium cobalt oxide cathodes are inherently unstable, held in check only by a thin insulating membrane which, if accidentally breached, can lead to thermal runaway involving gaseous oxygen, carbon, organic solvents, and (in some cases) lithium chlorate— all the components necessary for a fierce fire.
Much research has gone into the development of fail-safe membranes. In one type, made by ExxonMobil and targeted at the automotive market, the pores are designed to close up and thus inhibit the passage of lithium ions when the temperature rises above a safe level.
Biological Batteries
Finally, we should mention the biological batteries that are found in a number of electric fish. The "electric organs" of these fish are modified muscle cells known as electrocytes which are arranged in long stacks. A neural signal from the brain causes all the electrocytes in a stack to become polarized simultaneously, in effect creating a battery made of series-connected cells. Most electric fish produce only a small voltage which they use for navigation, much in the way that bats use sound for echo-location of prey. The famous electric eel, however, is able to produce a 600-volt jolt that it employs to stun nearby prey.