Extra Credit 14
- Page ID
- 82821
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Q17.2.3
For the cell notations in the previous problem, write the corresponding balanced reactions.
- Mg(s)│Mg2+(aq)║Cu2+(aq)│Cu(s)
- Ni(s)│Ni2+(aq)║Ag+(aq)│Ag(s)
S17.2.3
1. Answer:
\[Mg(s)+Cu^{_{2+}}(aq)\rightarrow Mg^{_{2+}}(aq)+Cu(s)\]
Solution:
We are asked to write the corresponding balanced reactions based on the cell notation.
First, we need to identify and sperate the half reactions. Note that the left side of the cell notation is the oxidation half reaction, and the right side of the cell notation is the reduction half reaction. Thus:
Oxidation half reaction:\(Mg(s)\rightarrow Mg^{_{2+}}(aq)\)
Reduction half reaction: \(Cu^{_{2+}}(aq)\rightarrow Cu(s)\)
Second, we need to balance each half reactions.
Each half equation is balanced by adjusting coefficients and adding H2O, H+, and e- in this order:
- Balance elements in the equation other than O and H.
- Balance the oxygen atoms by adding the appropriate number of water (H2O) molecules to the opposite side of the equation.
- Balance the hydrogen atoms (including those added in step 2 to balance the oxygen atom) by adding H+ ions to the opposite side of the equation.
- Add up the charges on each side. Make them equal by adding enough electrons (e-) to the more positive side.
- * If the equation is being balanced in a basic solution, through the addition of one more step, the appropriate number of OH- must be added to turn the remaining H+ into water molecules.
In this problem, as no O or H atoms are involved, we simply need to balance all elements, than adding e- to equal the changes.
Oxidation half reaction: \(Mg(s)\rightarrow Mg^{_{2+}}(aq)+2e^{_{-}}\)
Reduction half reaction:\(Cu^{_{2+}}(aq)+2e^{_{-}}\rightarrow Cu(s)\)
Finally, we adding two half equations together to from the final balanced reaction.
Half reactions are added together by this order:
- Making sure the numbers of e- in each half reactions are equal. If they are not equal, they must be multiplied by the lowest common multiple to be made the same.
- Adding to half equation together. Canceling out the electrons and common terms to form one balanced equation.
In this problem, numbers of e- in each half reactions are equal. Thus:
Balanced Equation : \(Mg(s)+Cu^{_{2+}}(aq)\rightarrow Mg^{_{2+}}(aq)+Cu(s)\)
Check the equation to make sure it is balanced.
2. Answer:
\[Ni(s)+2Ag^{_{+}}(aq)\rightarrow Ni^{_{2+}}(aq)+2Ag(s)\]
Solution:
We are asked to write the corresponding balanced reactions based on the cell notation.
First, we need to identify and sperate the half reactions. Note that the left side of the cell notation is the oxidation half reaction, and the right side of the cell notation is the reduction half reaction. Thus:
Oxidation half reaction: \(Mg(s)\rightarrow Mg^{_{2+}}(aq)\)
Reduction half reaction: \(Ag^{_{+}}(aq)\rightarrow Ag(s)\)
Second, we need to balance each half reactions.
In this problem, as no O or H atoms are involved, we simply need to balance all elements, than adding e- to equal the changes.
Oxidation half reaction: \(Ni(s)\rightarrow Ni^{_{2+}}(aq)+2e^{_{-}}\)
Reduction half reaction: \(Ag^{_{+}}(aq)+e^{_{-}}\rightarrow Ag(s)\)
Finally, we adding two half equations together to from the final balanced reaction.
Half reactions are added together by this order:
- Making sure the numbers of e- in each half reactions are equal. If they are not equal, they must be multiplied by the lowest common multiple to be made the same.
- Adding to half equation together. Canceling out the electrons and common terms to form one balanced equation.
In this problem, numbers of e- in each half reactions are not equal. To make it equal, the reduction half reaction need to be multiply by two. Then adding two equations together. Thus:
Oxidation half reaction: \(Ni(s)\rightarrow Ni^{_{2+}}(aq)+2e^{_{-}}\)
Reduction half reaction: \(2Ag^{_{+}}(aq)+2e^{_{-}}\rightarrow 2Ag(s)\)
Balanced Equation : \(Ni(s)+2Ag^{_{+}}(aq)\rightarrow Ni^{_{2+}}(aq)+2Ag(s)\)
Check the equation to make sure it is balanced.
Q19.1.12
How many cubic feet of air at a pressure of 760 torr and 0 °C is required per ton of Fe2O3 to convert that Fe2O3 into iron in a blast furnace? For this exercise, assume air is 19% oxygen by volume.
S19.1.12
Answer:
Solution:
In the process of converting Fe2O3 into Fe in a blast furnace ( see The Extraction of Iron), 3 steps of reaction occurs:
\[O_2(g)+C(s)\rightarrow CO_2(g)\]
\[CO_2(g)+C(s)\rightarrow CO(g)\]
\[Fe_2O_3(s)+3CO(g)\rightarrow 2Fe(s)+3CO_2(g)\]
Thus, the overall reaction is:
\[2Fe_2O_3(s)+6C(s)+3O_2(g)\rightarrow 4Fe(s)+6CO_2(g)\]
So, for 1 mole of Fe2O3, we need 3/2 mole of O2.
M(Fe2O3) = 1 ton/ (159.69 g/mole)=1 ton* (1000000 g/ton) / (159.69 g/mole) =6262.1mole
M(O2) =3/2*M(Fe2O3) = 9393.2mole
\[0.19n(air) = M(O2)\]
\[n(air) = 49438 mole\]
As the air is at a pressure of 760 torr and 0 °C, based on idea gas law PV=nRT.
In this problem, \(P=760\), \(torr=1 atm\), \(n = 49438mole\), \(T=0 °C+273.15=273.15K\), \(R = 0.08206 L atm mol^{_{-}}K^{_{-}}\).
\[PV=nRT \]
\[1 atm*V= 49438 mole *273.15K *0.08206 L atm mol^{_{-}}K^{_{-}} \]
\[ V = 1.1081*10^{_{6}L = 1.1081*10^{_{6}L*0.0353147 cubic feet L^{_{-}} =39134 cubic feet \]
Thus, 39134 cubic feet of air is needed.
Q19.3.4
The solid anhydrous solid CoCl2 is blue in color. Because it readily absorbs water from the air, it is used as a humidity indicator to monitor if equipment (such as a cell phone) has been exposed to excessive levels of moisture. Predict what product is formed by this reaction, and how many unpaired electrons this complex will have.
S19.3.4
Answer: [Co(H2O)6]Cl2 with three unpaired electrons.
Solution: The electron configuration of Co is [Ar]4s23d7. In this problem, as Cl has a charge of -1, and H2O has a has of 0, the Co has a charge of +2. Thus, Co2+has a electron configuration of [Ar]3d7.
The spectrochemical series is a list that orders ligands on the basis of their field strength.
(weak) I− < Br− < S2− < SCN− < Cl− < NO3− < N3− < F− < OH− < C2O42− ≈ H2O <
NCS− < CH3CN < py < NH3 < en < bipy < phen < NO2− < PPh3 < CN− ≈ CO (strong)
As H2O is a weak field ligand, the d-orbital energy-level diagram is at high spin. This is a ochahedral complex as Co has 6 ligands connect to ie.
Thus, we can construct a diagram:

Thus, there is 3 unpaired electron.
Q12.4.4
Pure ozone decomposes slowly to oxygen, 2O3(g)⟶3O2(g)2O3(g)⟶3O2(g). Use the data provided in a graphical method and determine the order and rate constant of the reaction.
| Time (h) | 0 | 2.0 × 10−3 | 7.6 × 10−3 | 1.00 × 10−2 |
|---|---|---|---|---|
| [O3] (M) | 1.00 × 10−5 | 4.98 × 10−6 | 2.07 × 10−6 | 1.66 × 10−6 |
| Time (h) | 1.23 × 10−2 | 1.43 × 10−2 | 1.70 × 10−2 | |
| [O3] (M) | 1.39 × 10−6 | 1.22 × 10−6 | 1.05 × 10−6 |
S12.4.4
Answer:

To identify how the concentrations changes a function of time, requires solving the appropriate differential equation (i.e., the differential rate law).
The zero-order rate law predicts in a linear decay of concentration with time
The 1st-order rate law predicts in an exponential decay of concentration with time
The 2nd-order rate law predicts in an reciprocal decay of concentration with time
![Time vs [O3].png](https://chem.libretexts.org/@api/deki/files/112236/Time_vs_%255BO3%255D.png?revision=1&size=bestfit&width=564&height=409)
The plot is not linear, so the reaction is not zero order.
![Time vs ln[O3].png](https://chem.libretexts.org/@api/deki/files/112237/Time_vs_ln%255BO3%255D.png?revision=1&size=bestfit&width=570&height=414)
The plot is not linear, so the reaction is not first order.
![Time vs 1over[O3].png](https://chem.libretexts.org/@api/deki/files/112240/Time_vs_1over%255BO3%255D.png?revision=1&size=bestfit&width=608&height=441)
The plot is nicely linear, so the reaction is second order.
To a second order equation, \( 1/[A] \ = k*t + 1/[A_0] \)
Thus, the value of K is the slope of the graph Time vs 1/[O3],
k = 50.3*10^6 L mol−1 h−1
Q12.7.7
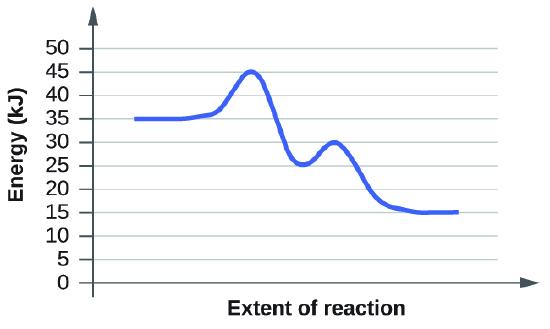
For each of the following reaction diagrams, estimate the activation energy (Ea) of the reaction:
(a)
(b)
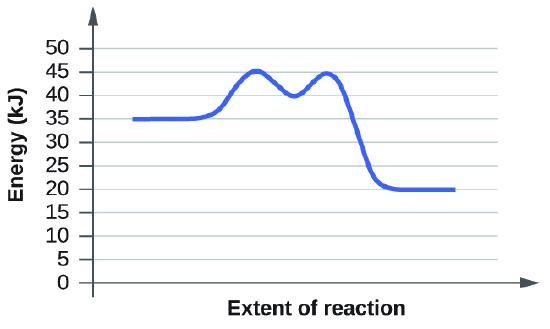
S12.7.7
Answer: (a) 10 kJ; (b) 10 kJ
Solution: The activation energy is the minimum energy that reacting species needed to undergo a reaction. More specifically, the activation energy is energy needed for a reacting specie go from its initial state to its transition state. Thus, the activation energy, equals to the absolute difference between energy at initial state and energy at transition state.
\[Ea = Et - Ei \]
On a reaction diagram, the energy at initial state is the energy value at the start point. The energy at transition state is the energy at its first maximum value. At the maximum value, energy starts to decline, showing that the reaction begins. Thus, the first maximum value is at the transition state.
For graph a), the energy at initial state is 35 kJ, and the energy at the transition state is round 45 kJ. Thus, the energy difference, which equals to the activation engegy.
\[Ea = 45 kJ - 35 kJ = 10 kJ. \]
For graph b), the energy at initial state is 30 kJ, and the energy at the transition state is round 40 kJ. Thus, the energy difference, which equals to the activation engegy.
\[Ea = 40 kJ - 30 kJ = 10 kJ \]
Q21.5.2
How does nuclear fission differ from nuclear fusion? Why are both of these processes exothermic?
S21.5.2
Answer:
For nuclear fission reaction, smaller atoms combine to form a larger atom. For nuclrea fusion reactoni, larger atoms disintegrate to from smaller products.
Both of these processes are exothermic because total binding energy of product nuclei is different from the total being energy of the reactant nucleus. The excess energy is released as a result of mass defect created during the reaction.
Q20.3.16
Write the half-reactions for each overall reaction, decide whether the reaction will occur spontaneously, and construct a cell diagram for a galvanic cell in which a spontaneous reaction will occur.
- Co(s) + Fe2+(aq) → Co2+(aq) + Fe(s)
- O2(g) + 4H+(aq) + 4Fe2+(aq) → 2H2O(l) + 4Fe3+(aq)
- 6Hg2+(aq) + 2NO3−(aq) + 8H+ → 3Hg22+(aq) + 2NO(g) + 4H2O(l)
- CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
S20.3.16
Answer:
Graph of Standard Reduction Potentials, see P2: Standard Reduction Potentials by Value and P1: Standard Reduction Potentials by Element
1.)
\[Co(s)+Fe^{_{2+}}(aq)\rightarrow Co^{_{2+}}(aq)+Fe(s)\]
In the redox reaction, the oxidation half of the reaction loses electron, and the reduction half of th reaction gain electron.
Oxidation Half Reaction: \(Co(s)+2e^{_{-}}\rightarrow Co^{_{2+}}(aq)\)
Reduction Half Reaction: \(Fe^{_{2+}}(aq)+2e^{_{-}}\rightarrow Fe(s)\)
To decide whether the reaction will occur spontaneously, we need to check whether E°cell > 0 or not. If E°cell > 0, the reaction is spontaneous.
Note that \(E°cell=E°cathode−E°anode\)
Substitute the value of standard reduction potential of E°cathode (reduction half reaction) and the value of standard reduction potential of E°anode (oxidation half reaction) base on the Standard Reduction Potential graph.
\( E°cell= -0.44V - (-0.28V) = -0.16V \)
Thus, this reaction is not spontaneous.
Thus, the spontaneous reaction will be \(Co^{_{2+}}(aq)+Fe(s)\rightarrow Co(s)+Fe^{_{2+}}(aq)\) Co2+(aq) + Fe(s) → Co(s) + Fe2+(aq)
Note: The anode (oxidation half reaction) always goes on the left and cathode (reduction half reaction) on the right. Separate changes in phase by | and indicate the the salt bridge with ||. The lack of concentrations indicates solutions are under standard conditions
In this problem, the oxidation half is \(Fe(s)+2e^{_{-}}\rightarrow Fe^{_{2+}}(aq)\), and the reduction half is \(Co^{_{2+}}(aq)+2e^{_{-}}\rightarrow Co(s)\).
The cell diagram is : Fe(s)│Fe2+(aq)║Co2++(aq)│Co(s)
2.) O2(g) + 4H+(aq) + 4Fe2+(aq) → 2H2O(l) + 4Fe3+(aq)
\[O_2(g)+4H^{_{+}}(aq)+4Fe^{_{2+}}(aq)\rightarrow 2H_2O(l) +4Fe^{_{3+}}\]
In the redox reaction, the oxidation half of the reaction loses electron, and the reduction half of th reaction gain electron.
Oxidation Half Reaction: \(Fe^{_{2+}}(aq)\rightarrow Fe^{_{3+}} +e^{_{-}}\)
Reduction Half Reaction:\(O_2(g)+4H^{_{+}}(aq)+4e^{_{-}}\rightarrow 2H_2O(l) \)
To decide whether the reaction will occur spontaneously, we need to check whether E°cell > 0 or not. If E°cell > 0, the reaction is spontaneous.
Note that \(E°cell=E°cathode−E°anode\)
Substitute the value of standard reduction potential of E°cathode (reduction half reaction) and the value of standard reduction potential of E°anode (oxidation half reaction) base on the Standard Reduction Potential graph.
\(E°cell=1.23V - 0.771V = 0.459V\)
Thus, this reaction is spontaneous.
Thus, the spontaneous reaction will be \(O_2(g)+4H^{_{+}}(aq)+4Fe^{_{2+}}(aq)\rightarrow 2H_2O(l) +4Fe^{_{3+}}\)
Note: The anode (oxidation half reaction) always goes on the left and cathode (reduction half reaction) on the right. Separate changes in phase by | and indicate the the salt bridge with ||. The lack of concentrations indicates solutions are under standard conditions
In this problem, as no solid reactants can be used as electrodes, we need to add Pt as electrodes.
The cell diagram is : Pt(s)│Fe2+(aq),Fe3+(aq)║O2(g)│H+(aq)│Pt(s)
3.)
\[6Hg^{_{2+}}(aq)+2NO_3^{_{-}}(aq) + 8H^{_{+}}\rightarrow 3Hg_2^{_{2+}}(aq)+2NO(g) +4H_2O(l) \]
In the redox reaction, the oxidation half of the reaction loses electron, and the reduction half of th reaction gain electron.
Oxidation Half Reaction: \(2Hg^{_{2+}}(aq) +2e^{_{-}}\rightarrow Hg_2^{_{2+}}(aq)\)
Reduction Half Reaction: \(NO_3^{_{-}}(aq) + 4H^{_{+}}+3e^{_{-}}\rightarrow NO(g) +2H_2O(l) \)
To decide whether the reaction will occur spontaneously, we need to check whether E°cell > 0 or not. If E°cell > 0, the reaction is spontaneous.
Note that \(E°cell=E°cathode−E°anode\)
Substitute the value of standard reduction potential of E°cathode (reduction half reaction) and the value of standard reduction potential of E°anode (oxidation half reaction) base on the Standard Reduction Potential graph.
\[E°cell=0.96V - 0.911V = 0.059V\]
Thus, this reaction is spontaneous.
Thus, the spontaneous reaction will be \(6Hg^{_{2+}}(aq)+2NO_3^{_{-}}(aq) + 8H^{_{+}}\rightarrow 3Hg_2^{_{2+}}(aq)+2NO(g) +4H_2O(l) \) 6Hg2+(aq) + 2NO3−(aq) + 8H+ → 3Hg22+(aq) + 2NO(g) + 4H2O(l)
Note: The anode (oxidation half reaction) always goes on the left and cathode (reduction half reaction) on the right. Separate changes in phase by | and indicate the the salt bridge with ||. The lack of concentrations indicates solutions are under standard conditions
In this problem, as no solid reactants can be used as electrodes, we need to add Pt as electrodes.
The cell diagram is : Pt(s)│Hg2+(aq) ,Hg22+(aq) ║2NO3−(aq)│2NO(g)│Pt(s)
4.) CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
\[CH_4(g)+2O_2(g) \rightarrow CO_2(g) + 2H_2O(l) \]
In the redox reaction, the oxidation half of the reaction loses electron, and the reduction half of th reaction gain electron.
Oxidation : \(CH_4(g) \rightarrow CO_2(g) \)
Reduction: \(O_2(g) \rightarrow 2H_2O(l) \)
Balance each reaction:
Oxidation :\(CH_4(g)+ 2H_2O \rightarrow CO_2(g)+8H^{_{+}} +8e^{_{-}} \)
Reduction: \(O_2(g)+4H^{_{+}} +4e^{_{-}}\rightarrow 2H_2O(l) \)
To decide whether the reaction will occur spontaneously, we need to check whether E°cell > 0 or not. If E°cell > 0, the reaction is spontaneous.
Note that \(E°cell=E°cathode−E°anode\)
Substitute the value of standard reduction potential of E°cathode (reduction half reaction) and the value of standard reduction potential of E°anode (oxidation half reaction) base on the Standard Reduction Potential graph.
In this problem, we couldn't find the direct SRP value from the table.
But, because this reaction is a combustion reaction, we assume that this reaction is spontaneous
Thus, the spontaneous reaction will be \(CH_4(g)+2O_2(g) \rightarrow CO_2(g) + 2H_2O(l) \)
Note: The anode (oxidation half reaction) always goes on the left and cathode (reduction half reaction) on the right. Separate changes in phase by | and indicate the the salt bridge with ||. The lack of concentrations indicates solutions are under standard conditions
In this problem, as no solid reactants can be used as electrodes, we need to add C as electrodes.
The cell diagram is : C(s)│CH4(g)CO2(g)║O2(g),CO2(g),2H2O(g)│C(s)
Q20.5.29
You have constructed a cell with zinc and lead amalgam electrodes described by the cell diagram Zn(Hg)(s)∣Zn(NO3)2(aq)∥Pb(NO3)2(aq)∣Pb(Hg)(s). If you vary the concentration of Zn(NO3)2 and measure the potential at different concentrations, you obtain the following data:
| Zn(NO3)2 (M) | Ecell (V) |
|---|---|
| 0.0005 | 0.7398 |
| 0.002 | 0.7221 |
| 0.01 | 0.7014 |
- Write the half-reactions that occur in this cell.
- What is the overall redox reaction?
- What is E°cell? What is ΔG° for the overall reaction?
- What is the equilibrium constant for this redox reaction?
S20.5.29
Answer:
1.) Based on the cell diagram, the left side of the cell notation is the oxidation half reaction, and the right side of the cell notation is the reduction half reaction. Thus:
Oxidation Half Reaction: \(Zn(s)+2NO_3^{_{-}}(aq)\rightarrow Zn(NO_3)_2(aq)+2e^{_{-}}\)
Reduction Half Reaction: \(Pb(NO_3)_2(aq) + 2e^{_{-}}\rightarrow Pb(s)+2NO_3^{_{-}}(aq)\)
2.) To get the overall redox reaction, we need to balance both half equations, than add them together, cancelling other common elements. Detailed procedure can be found at S17.2.3
Overall redox reaction: \(Zn(s)+(Pb(NO_3)_2(aq)\rightarrow Zn(NO_3)_2(aq)+Pb(s)\)
3.)
\(E°cell=E°cathode−E°anode\)
Substitute the value of standard reduction potential of E°cathode (reduction half reaction) and the value of standard reduction potential of E°anode (oxidation half reaction) base on the Standard Reduction Potential graph.( See S20.3.16)
\[E°cell=-0.13V - (-0.79V) = 0.63V\]
ΔG°=-nFE°. n is the number of electron transferred in overall reaction. F, the Faraday constant, equals to 96485 C/mole. E° is the cell potential.
In this problem, n=2, and E°cell =0.63V.
\[ΔG°=-nFE°= - 2 mole* 96485 C/mole * 0.63 V = -121571 J=-1.2*10^{_{5}}J.\]
4.) ΔG°=-RTIn(k). R, the idea gas constant, equals to 8.31453 J/(mole*K). T is the temperature in Kelvin. K is the equilibrium constant.
In this problem, ΔG°=-1.2*105J. As T is not given, we assumed at the reaction is under standard condition, in which T= 298K.
\[ΔG°=RTInk\]
\[-1.2*10^{_{5}}J = -8.31453*298* \natural(k)\]
\[k = 1.1*10^{_{21}}\]
Note: If use ΔG° = -121571 J, K = 2.0*1021,

