Enamine Reactions
- Page ID
- 5133
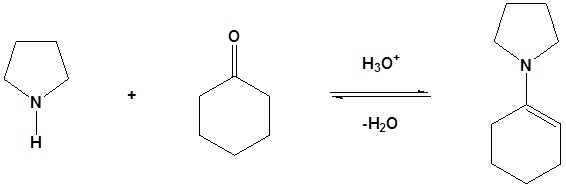
As previously seen, aldehydes and ketones react with 2o amines to reversibly form enamines.
Example
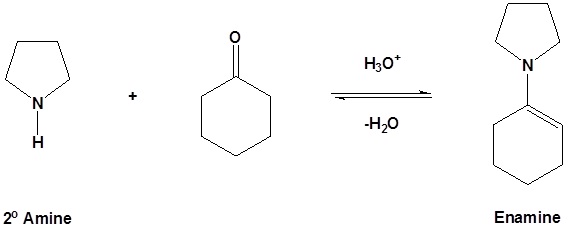
Reversible
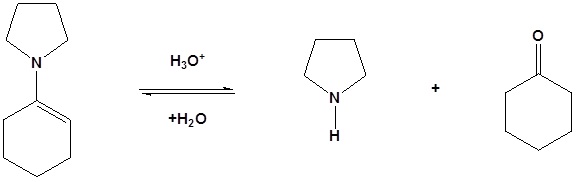
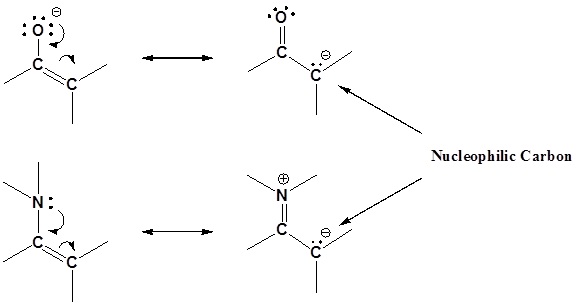
Enamines act as nucleophiles in a fashion similar to enolates. Because of this enamines can be used as synthetic equivalents as enolates in many reactions. This process requires a three steps:
- Formation of the enamine,
- Reaction with an eletrophile to form an iminium salt,
- Hydrolysis of the iminium salt to reform the aldehyde or ketone.
Some of the advantages of using an enamine over and enolate are enamines are neutral, easier to prepare, and usually prevent the overreaction problems plagued by enolates. These reactions are generally known as the Stork enamine reaction after Gilbert Stork of Columbia University who originated the work.
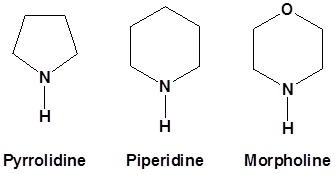
Typically we use the following 2o amines for enamine reactions
Alkylation of an Enamine
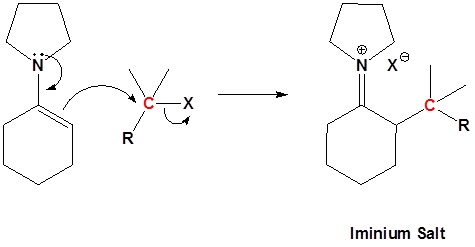
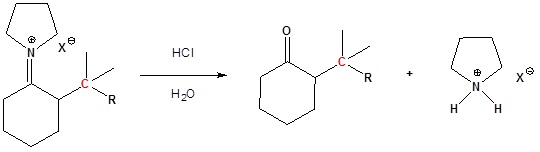
Enamined undergo an SN2 reaction with reactive alkyl halides to give the iminium salt. The iminium salt can be hydrolyzed back into the carbonyl.
Step 1: Formation of an enamine
Step 2: SN2 Alkylation
Step 3: Reform the carbonyl by hydrolysis
All three steps together:
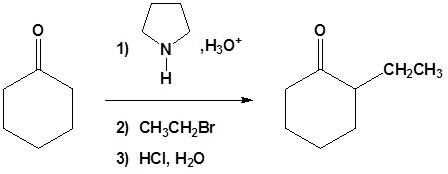
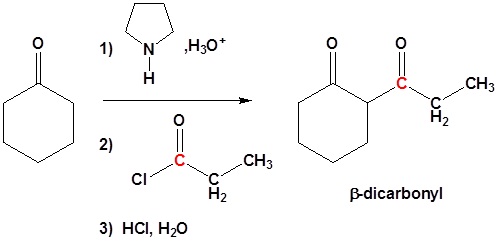
Acylation of Enamines
Enamine can react with acid halides to form β-dicarbonyls
1) Formation of the enamine
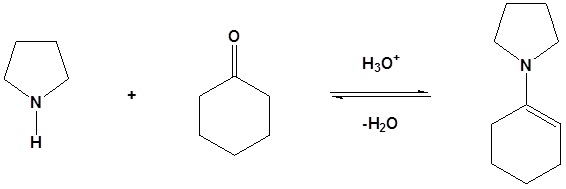
2) Nucleophilic attack
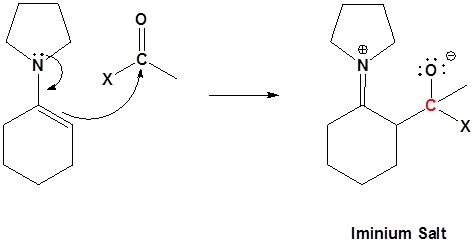
3) Leaving group removal
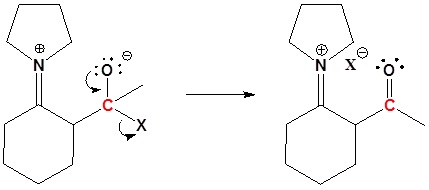
4) Reform the carbonyl by hydrolysis
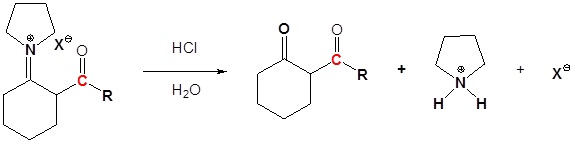
All three steps together:
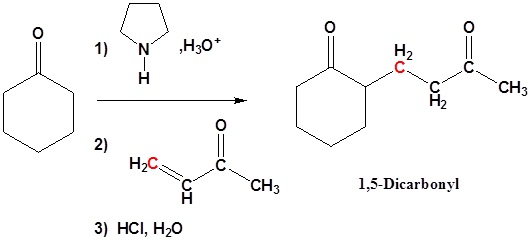
Michael Addition using Enamines
Enamines, like other weak bases, add 1,4 to enones. The end product is a 1,5 dicarbonyl compound.
Contributors
Prof. Steven Farmer (Sonoma State University)

