Electrophilic Substitution at Oxygen
- Page ID
- 739
Because of its enhanced acidity, the hydrogen atom of a hydroxyl group is easily replaced by other substituents. A simple example is the facile reaction of simple alcohols with sodium (and sodium hydride), as described in the first equation below. Another such substitution reaction is the isotopic exchange that occurs when mixing an alcohol with deuterium oxide (heavy water). This exchange, which is catalyzed by acid or base, is rapid under normal conditions because it is difficult to avoid traces of these catalysts in most experimental systems.
2 R–O–H + 2 Na  2 R–O(–)Na(+) + H2 2 R–O(–)Na(+) + H2 |
R–O–H + D2O  R–O–D + D–O–H R–O–D + D–O–H |
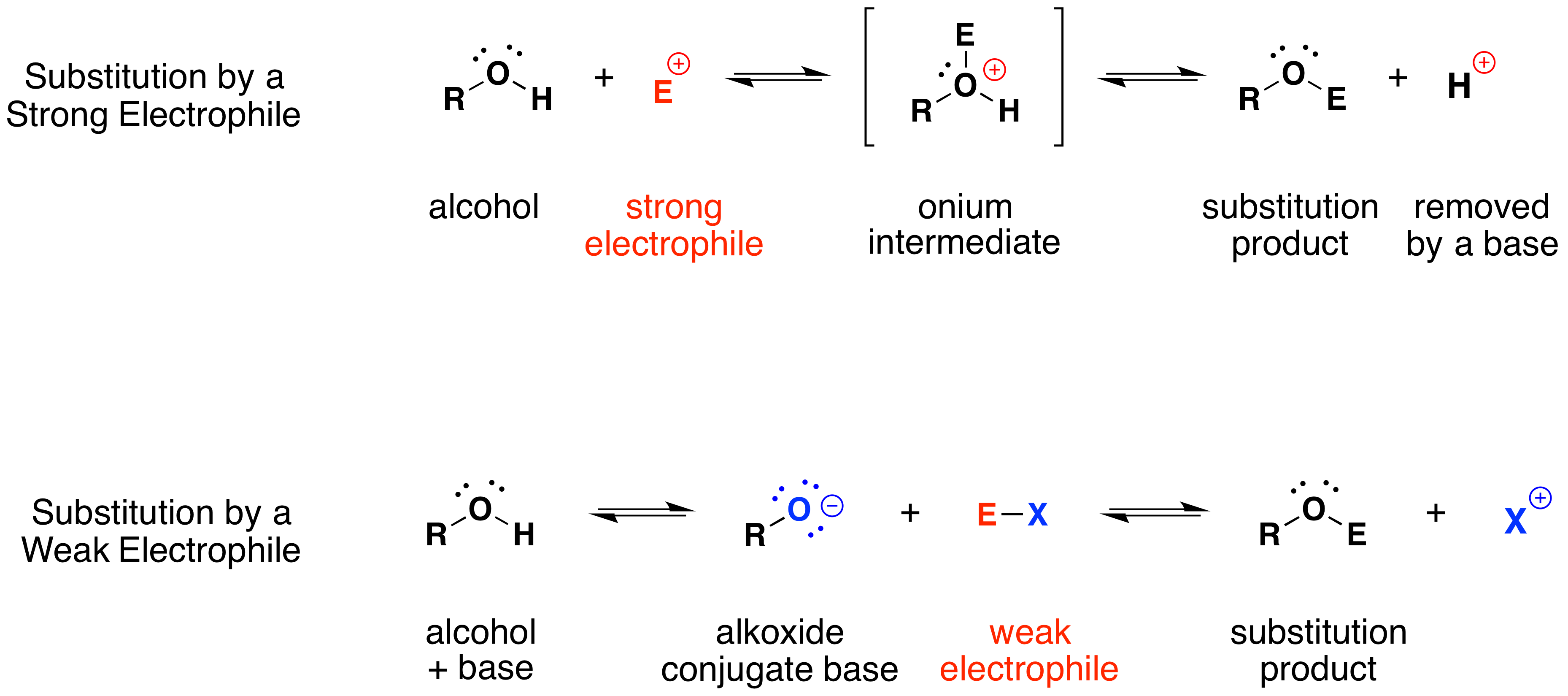
The mechanism by which these substitution reactions proceed is straightforward. The oxygen atom of an alcohol is nucleophilic; therefore, it is prone to attack by electrophiles. The resulting "onium" intermediate then loses a proton to a base, forming the substitution product. If a strong electrophile is not present, then the nucleophilicity of the oxygen may be enhanced by conversion to its conjugate base (an alkoxide). This powerful nucleophile then attacks the weak electrophile. These two variations of the substitution mechanism are illustrated in the following diagram.
The preparation of tert-butyl hypochlorite from tert-butyl alcohol is an example of electrophilic halogenation of oxygen, but this reaction is restricted to 3º alcohols; 1º and 2º hypochlorites lose HCl, forming aldehydes and ketones. In the following equation the electrophile may be regarded as Cl(+).
(CH3)3C–O–H + Cl2 + NaOH  (CH3)3C–O–Cl + NaCl + H2O
(CH3)3C–O–Cl + NaCl + H2O
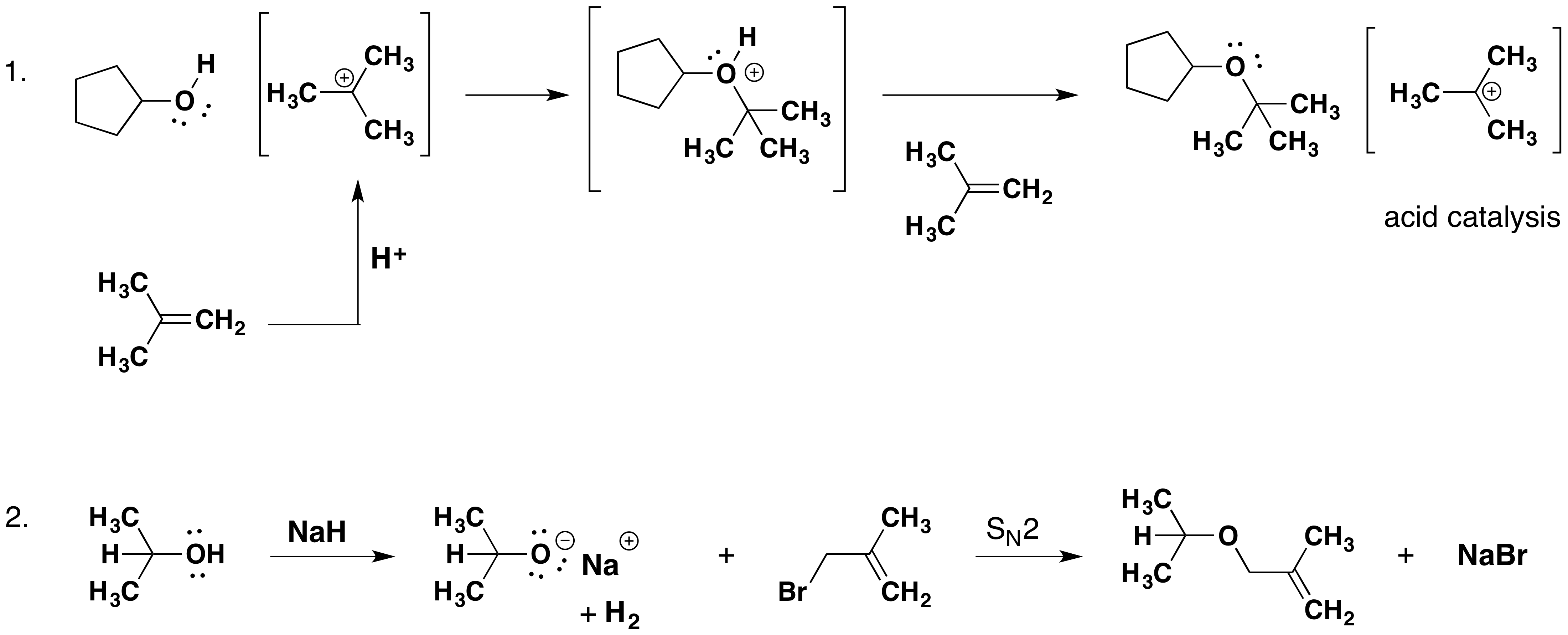
Alkyl substitution of the hydroxyl group creates ethers. This reaction provides examples of both strong electrophilic substitution (first equation below) and weak electrophilic substitution (second equation). The latter SN2 reaction is known as the Williamson ether synthesis and is generally used only with 1º alkyl halide reactants because the strong alkoxide base leads to E2 elimination of 2º and 3º alkyl halides.
Ester Formation
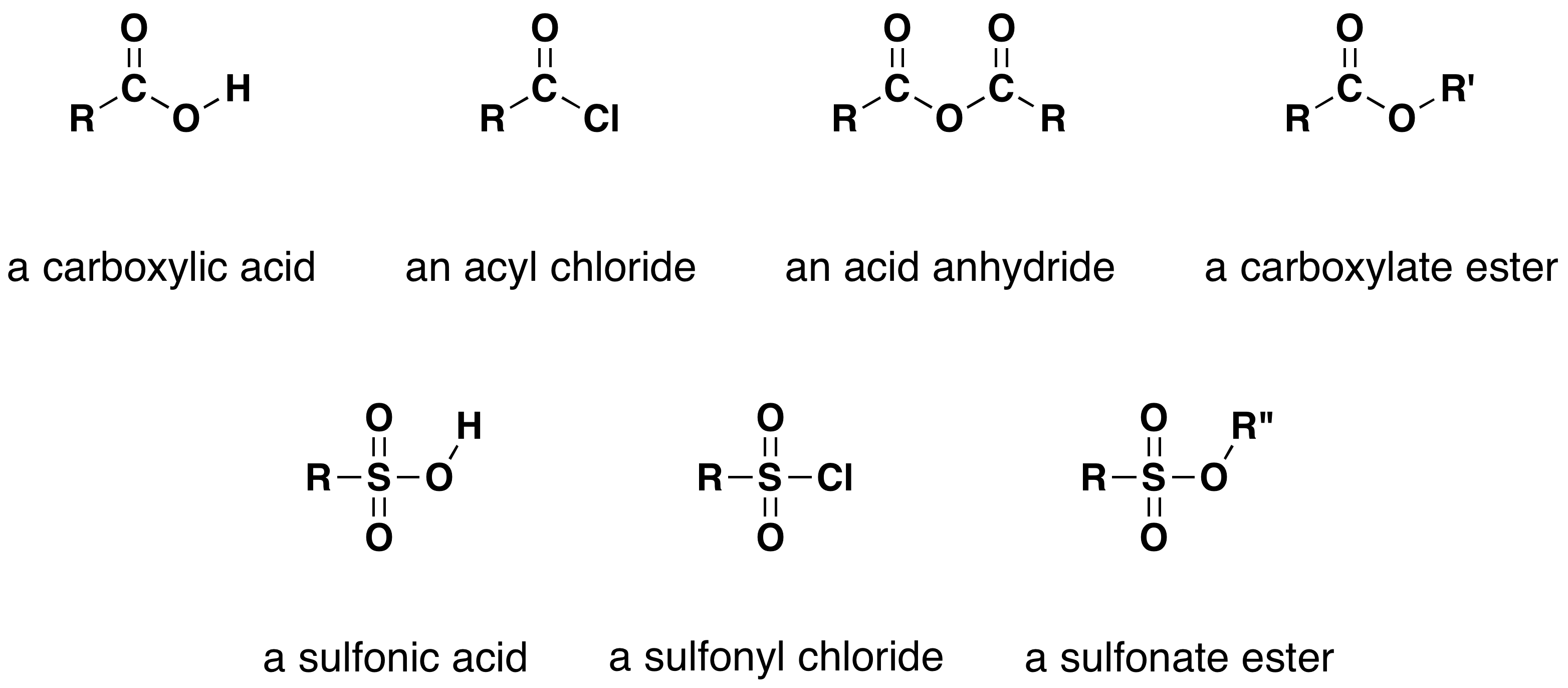
One of the most important substitution reactions at oxygen is ester formation, resulting from the reaction of alcohols with electrophilic derivatives of carboxylic and sulfonic acids. The following illustration displays the general formulas of these reagents and their ester products; the R'–O– group represents the alcohol moiety. The electrophilic atoms in the acid chlorides and anhydrides are colored red. Examples of specific esterification reactions are shown below.
Acyl Chloride Esterification
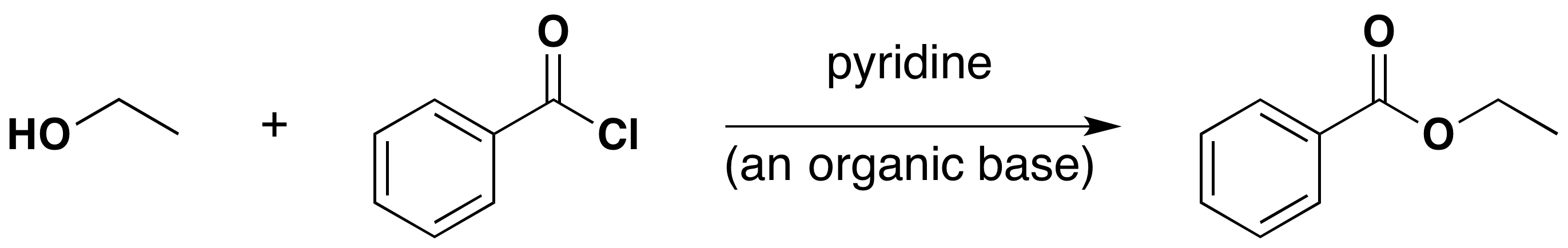
Anhydride Esterification

Methanesulfonate Formation
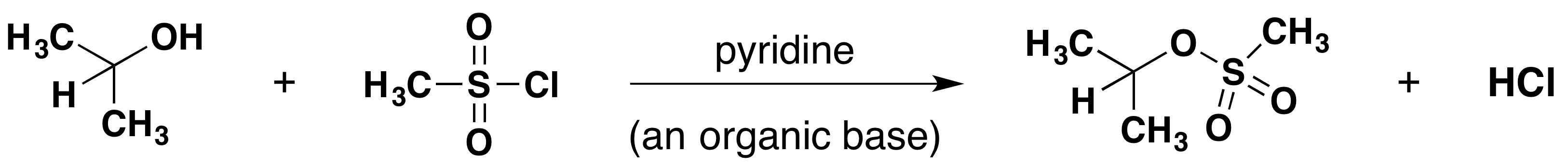
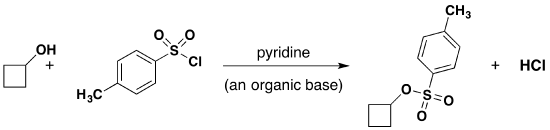
Tosylate Formation
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


