8.16: Aldehydes and Ketones
- Page ID
- 49465
Drinking methanol is harmful, not because of the CH3OH molecules themselves, but rather because the human body converts these molecules into methanal (formaldehyde) molecules by combination with oxygen:
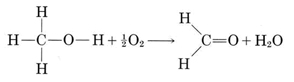
Formaldehyde, H2CO, is very reactive—in the pure state it can combine explosively with itself, forming much larger molecules. Consequently it is prepared commercially as a water solution, formalin, which contains about 35 to 40 percent H2CO. Formalin is made by combining methanol with air at 550°C over a silver or copper catalyst. It is used as a preservative for biological specimens, in embalming fluids, and as a disinfectant and insecticide—not a very good substance to introduce into your body. The biggest commercial use of formaldehyde is manufacture of Bakelite, melamine, and other plastics.
The 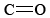 functional group found in formaldehyde is called a carbonyl group. Two classes of compounds may be distinguished on the basis of the location of the carbonyl group. In aldehydes it is at the end of a carbon chain and has at least one hydrogen attached. In ketones the carbonyl group is attached to two carbon atoms. Some examples are
functional group found in formaldehyde is called a carbonyl group. Two classes of compounds may be distinguished on the basis of the location of the carbonyl group. In aldehydes it is at the end of a carbon chain and has at least one hydrogen attached. In ketones the carbonyl group is attached to two carbon atoms. Some examples are

Chemical tests may be performed to determine the presence of a carbonyl group. In the video below, a solution of 2,4-dinitrophenylhydrazine is added to test tubes containing 2-propanol, an alcohol; 2-propanone (acetone), a ketone; and propionic acid a carboxylic acid. 2,4-dinitrophenylhydrazine only reacts with the carbonyl group of 2-propanone, forming an orange precipitate. The reaction that occurs is:
Another test can distinguish between aldehydes and ketones. Here is a video of the Silver Mirror Tollens Test for Aldehydes:
Tollens reagent is an aqueous solution of silver nitrate, sodium hydroxide, and a little ammonia. (In the video the ammonia comes from reaction of ammonium ions in ammonium nitrate and hydroxide ions from sodium hydroxide to form water and NH3.) If an aldehyde, in this case, glucose, is added to the solution, the Ag+ is reduced by the aldehyde, and the aldehyde is oxidized into a carboxylic acid. This produces silver metal, which coats the flask and creates the mirror. A similar reaction does not occur for ketones, so only aldehydes produce the silver mirror. The equation for the reaction in the video is:
The endings al and one signify aldehyde and ketone, respectively. The general formula for an aldehyde is 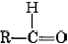 , while for a ketone it is
, while for a ketone it is 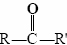 . Note that every ketone is isomeric with at least one aldehyde. Acetone, for example, has the same molecular formula (C3H6O) as propanal.
. Note that every ketone is isomeric with at least one aldehyde. Acetone, for example, has the same molecular formula (C3H6O) as propanal.
Aldehyde and ketone molecules cannot hydrogen bond among themselves for the same reason that ethers cannot—they do not contain hydrogens attached to highly electronegative atoms. The carbonyl group is rather polar, however, since the difference between the electronegativities of carbon (2.5) and oxygen (3.5) is rather large, and there are usually no other dipoles in an aldehyde or ketone molecule to cancel the effect of C==O.
Therefore the boiling points of aldehydes and ketones are intermediate between those of alkanes or ethers on the one hand and alcohols on the other. Acetaldehyde, CH3CH2CHO, boils at 20.8°C midway between propane (–42°C) and ethanol (78.5°C). The boiling points of propanal and acetone are compared with other organic compounds in the table of the boiling points of comparable organic compounds which shows the same trend.
Of all the aldehydes and ketones, formaldehyde and acetone are of greatest commercial importance. Uses of formaldehyde have already been mentioned. Like other ketones, acetone is mainly useful as a solvent, and you may have used it for this purpose in the laboratory. Acetone and other ketones are somewhat toxic and should not be handled carelessly.


