1.5: Characteristics of the Diels-Alder Reaction
- Page ID
- 500351
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- determine whether or not a given compound would behave as a reactive dienophile in a Diels-Alder reaction.
- predict the stereochemistry of the product obtained from the reaction of a given diene with a given dienophile.
- recognize that in order to undergo a Diels-Alder reaction, a diene must be able to assume ans-cis geometry, and determine whether or not a given diene can assume this geometry.
The Dienophile
In general, Diels-Alder reactions proceed fastest with electron-withdrawing groups on the dienophile (diene lover). Ethylene reacts slowly while propenal, ethyl propenoate, and other molecules shown below are highly reactive in a Diels-Alder reaction.
In much the same manner as electron-withdrawing substituents on a benzene ring, these are typically a double or triple bond in conjugation with the double bond in the dienophile. A resonance form can be drawn which places a positive charge in the dienophile double bond. This results in the double bond being less electron rich (greater electron density shown in Red/Orange) than ethylene. Electrostatic potential maps in Figure \(\PageIndex{1}\) show that the electron-withdrawing groups pull electron density away from the double bond.
Stereochemistry of Diels-Alder (dienophile)
The Diels-Alder reaction is enormously useful for synthetic organic chemists, not only because ring-forming reactions are useful in general but also because in many cases multiple new stereocenters are formed, and the reaction is inherently stereospecific. During a Diels-Alder reaction the stereochemistry of the dienophile is retained in the product. A cis dienophile will generate a cyclohexene ring with cis (syn) substitution on the two carbons from the dienophile. Likewise a trans dienophile will generate a cyclohexene ring with trans (anti) substitution on these two carbon.
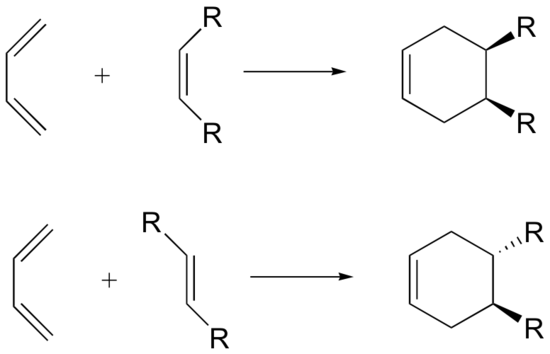
During a Diels-Alder reaction the stereochemistry of the dienophile is retained in the product. A cis dienophile will generate a cyclohexene ring with cis (syn) substitution on the two carbons from the dienophile. Likewise a trans dienophile will generate a cyclohexene ring with trans (anti) substitution on these two carbon.

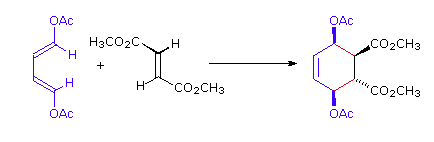
The retention of stereochemistry is due to the planar nature of both reactants and that the forming process is suprafacial (i.e. to or from the same face of each plane). This stereospecificity also confirms the concerted nature of the Diels-Alder mechanism. The drawing below illustrates this fact for the reaction of 1,3-butadiene with (E)-dicyanoethene. The trans relationship of the cyano groups in the dienophile is preserved in the six-membered ring of the adduct.
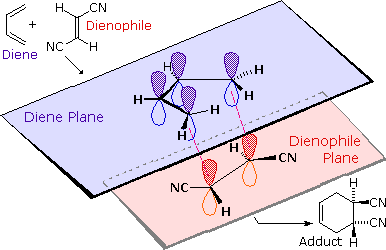
Formation of Bicyclic Structures
Another facet of the stereochemical retention of the dienophile is that only the endo product, rather than the alternative exo product, is formed. The words endo and exo are used to indicate relative stereochemistry when referring to bicyclic structures like substituted norbornanes. The endo position on a bicyclic structure refers to the position that is inside the concave shape of the larger (six-membered) ring. As you might predict, the exo position refers to the outside position.
Diels-Alder reactions with cyclic dienes favor the formation of bicyclic structures in which substituents are in the endo position. Preference of the endo position is also a facet of the suprafacial nature of the Diels-Alder reaction. The orbital overlap required for the reaction is greater when the dienophile lies directly underneath the diene.

In the reaction of 1,3-cyclopentadiene with maleic anhydride, for instance, the following result is obtained:
Other Dienophiles
Alkynes can also serve as dienophiles in Diels-Alder reactions:


The Diene
In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups). The Diels-Alder reaction is a single step process, so the diene component must adopt an s-cis conformation in order for the end carbon atoms (#1 & #4) to bond simultaneously to the dienophile. For many acyclic dienes the s-trans conformer is more stable than the s-cis conformer (due to steric crowding of the end groups), but the two are generally in rapid equilibrium, permitting the use of all but the most hindered dienes as reactants in Diels-Alder reactions.
In the alternative s-trans conformation, the ends of the diene partner are too far apart to overlap with the dienophile p orbitals.
Predict the product of the following Diels–Alder reaction:
Strategy
Draw the diene so that the ends of its two double bonds are near the dienophile double bond. Then form two single bonds between the partners, convert the three double bonds into single bonds, and convert the former single bond of the diene into a double bond. Because the dienophile double bond is cis to begin with, the two attached hydrogens must remain cis in the product.
Solution
Unreactive Dienes
The dienes that can’t adopt an s-cis conformation, don’t undergo Diels–Alder reactions. Examples of fixed dienes in an s-trans arrangement and larger the substituents, more stable the s-trans geometry are shown in Figure \(\PageIndex{4}\). Those dienes can’t achieve an s-cis conformation and thus can’t undergo Diels–Alder reactions.

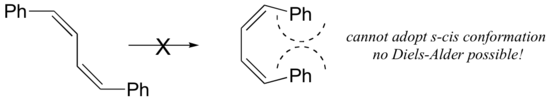
Figure \(\PageIndex{2}\): Examples of unreactive dienes: (a) Diene in an s-trans arrangement by a ring and (b) with large substituents
Examples are shown in Figure \(\PageIndex{3}\). In the bicyclic diene, the double bonds are rigidly fixed in an s-trans arrangement by geometric constraints of the rings. In (2Z,4Z)-2,4-hexadiene, steric strain between the two methyl groups prevents the molecule from adopting s-cis geometry.
Figure \(\PageIndex{3}\): Fixed dienes in an s-trans arrangement and Larger the substituents, more stable the s-trans geometry. The dienes that can’t achieve an s-cis conformation and thus can’t undergo Diels–Alder reactions.
Highly reactive dienes
In contrast to these unreactive dienes that can’t achieve an s-cis conformation, other dienes are fixed only in the correct s-cis geometry and are therefore highly reactive in Diels–Alder cycloaddition. 1,3-Cyclopentadiene, for example, is so reactive that it reacts with itself. At room temperature, 1,3-cyclopentadiene dimerizes. One molecule acts as diene and a second molecule acts as dienophile in a self-Diels–Alder reaction.

Stereochemistry of Diels-Alder (diene)
The 1 and 4 Carbons in the diene have the possibility of forming two new stereocenters in the cyclohexene product. Similarly to the effects of dienophile stereochemistry, the positioning of substituents on the 1 and 4 carbons in the diene determine the stereochemistry in the product. The diene substituents can be thought of as being either cis (both facing in or both facing out) or trans and the stereochemistry is retained to form a cis or trans cyclohexene product.
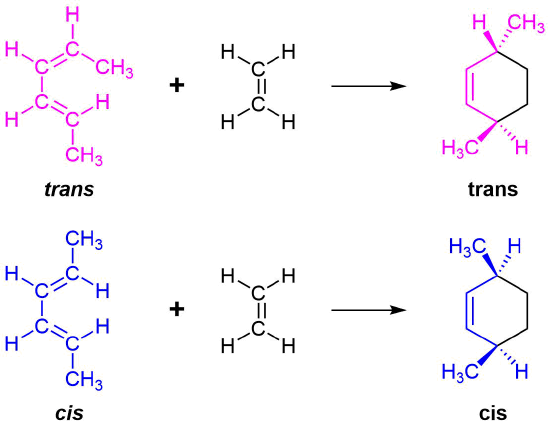

The Essential Characteristics of the Diels-Alder Cycloaddition Reaction:
- The reaction always creates a new six-membered ring.
- The diene component must be able to assume an s-cis conformation.
- Electron withdrawing groups on the dienophile facilitate reaction.
- Electron donating groups on the diene facilitate reaction.
- Steric hindrance at the bonding sites may inhibit or prevent reaction.
- The reaction is stereospecific with respect to substituent configuration in both the dienophile and the diene.
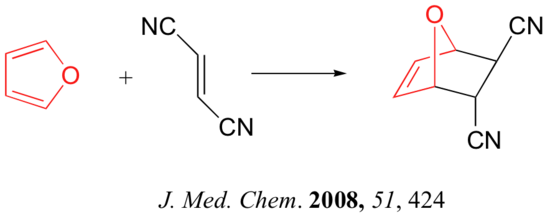
Predicting the Product of a Diels-Alder Reaction
Start by rotating the diene until it is in the s-cis conformation then point it towards the double bond of the dienophile. Remove the double bonds present in the diene and dieneophile. Connect carbons 1 and 4 of the the diene to a carbon in the dienophile double bond using a sigma bond to create a six-membered ring. Create a double bond between carbons 2 and 3 of diene.
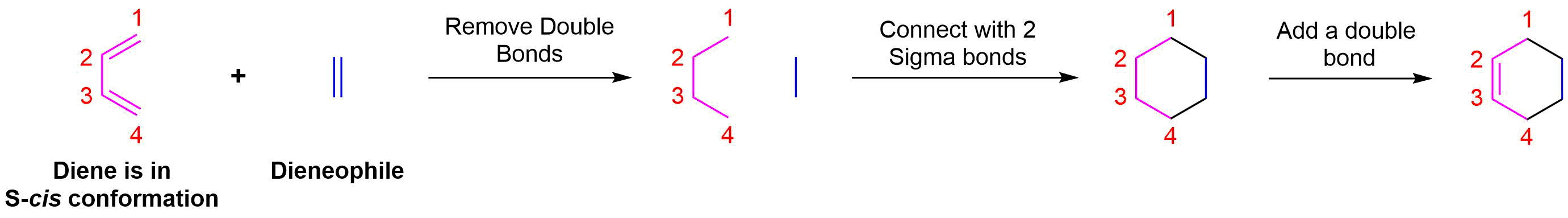
Determine if any substituents attached to either the double bond of the dieneophile or carbons 1 and 4 of the diene have a cis/trans conformation. If so, make sure the substituents have the same configuration as the cycloalkene product.

Predict the product of the following Diels–Alder reaction:

Strategy
The diene is locked into an s-cis configuration which will promote the reaction. The ring portion of the diene will act as electron donating groups which will also promote the reaction. Form two single bonds between the partners, convert the three double bonds into single bonds, and convert the former single bond of the diene into a double bond.
Because the diene already contained a ring the product will be bicyclic. The dienophile has two nitriles attached to it both of which are electron withdrawing. Since the two nitriles in the dieneophile are cis to each other the the two nitriles will be cis to each other in the product.
Solution
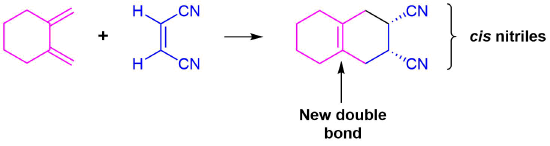
Which of the following alkenes would you expect to be good Diels–Alder dienophiles?
(a) (b)
(c)
(d)
(e)
- Answer
-
Good dienophiles: (a), (d)
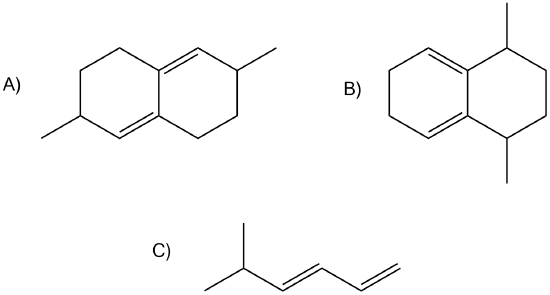
Of the following dienes, which are S-trans and which are s-cis? Of those that are s-trans, are they able to rotate to become s-cis?
- Answer
-
A) s-trans, unable to rotate to become s-cis. B) s-cis. C) s-trans, can rotate to become s-cis.
Predict the product of the following reactions:
(a) 
(b)
(c)
- Answer
-
(a)
.png?revision=1&size=bestfit&width=594&height=148) (b)
(b)(c)
Draw the major product of each following Diels-Alder reaction:
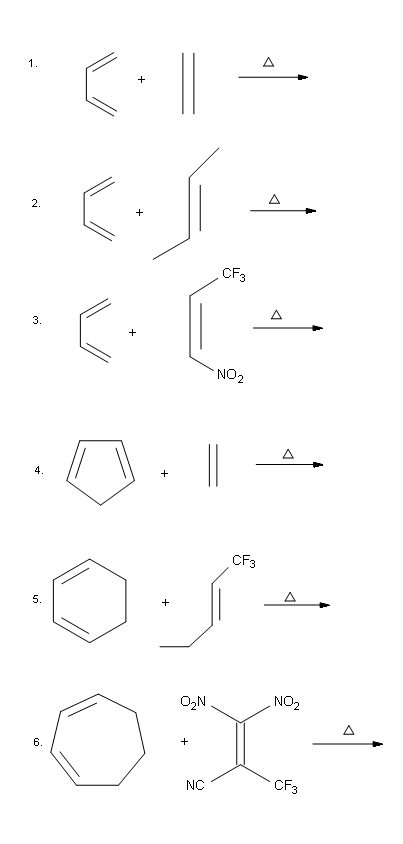
- Answer
-
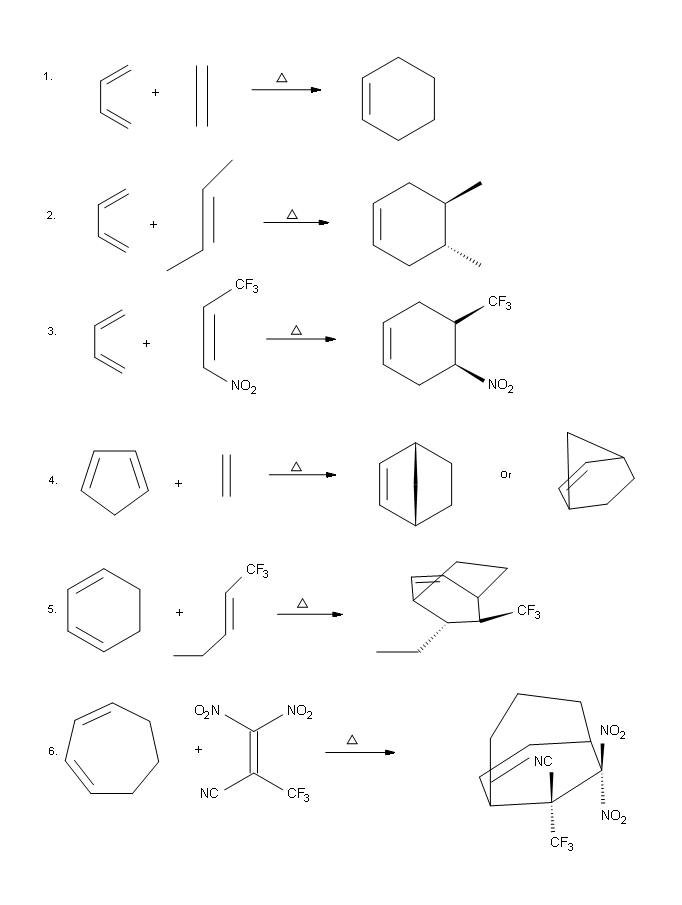.png?revision=1&size=bestfit&width=590&height=782)
Ensure to understand that the s-cis and s-trans forms of a diene such as 1,3-butadiene are conformers, not isomers. Note that some textbooks can confuse the issue further by referring to a compound such as (2Z, 4Z)-hexadiene as cis, cis-2,4-hexadiene, and saying that the most stable form of this compound is its s-trans conformer!
In fulfilling Objective 2, above, you must recognize that the Diels-Alder reaction is stereospecific.
Finally, note reaction B in the reading shows 1,3-cyclopentadiene reacting with another molecule of 1,3-cyclopentadiene. When the same compound acts as both diene and dienophile in a Diels-Alder reaction to couple it is a dimerization.


