9.11: Conversion of Alcohols to Alkyl Halides with HX
- Page ID
- 28205
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)When alcohols react with a hydrogen halide, a substitution takes place producing an alkyl halide and water. This conversion can be achieved in basic solution with thionyl chloride and one equivalent of pyridine as outlined in the previous video.
Scope of HX Reaction with Alcohols
- The order of reactivity of alcohols is 3° > 2° > 1° methyl.
-
The order of reactivity of the hydrogen halides is HI > HBr > HCl (HF is generally unreactive).
The reaction is acid catalyzed. Alcohols react with the strongly acidic hydrogen halides HCl, HBr, and HI, but they do not react with nonacidic NaCl, NaBr, or NaI.
Mechanisms of the Reactions of Alcohols with HX
Secondary, tertiary, allylic, and benzylic alcohols appear to react by a mechanism that involves the formation of a carbocation, in an \(S_N1\) reaction with the protonated alcohol acting as the substrate.
The \(S_N1\) mechanism is illustrated by the reaction tert-butyl alcohol and aqueous hydrochloric acid (\(H_3O^+\), \(Cl^-\) ). The first two steps in this \(S_n1\) substitution mechanism are protonation of the alcohol to form an oxonium ion. Although the oxonium ion is formed by protonation of the alcohol, it can also be viewed as a Lewis acid-base complex between the cation (\(R^+\)) and \(H_2O\). Protonation of the alcohol converts a poor leaving group (OH-) to a good leaving group (\)H_2O\_), which makes the dissociation step of the \(S_N1\) mechanism more favorable.
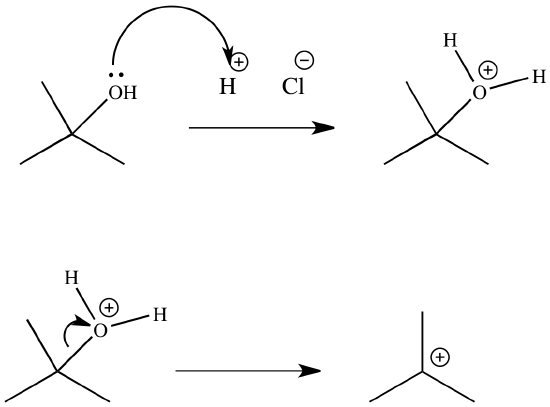
In step 3, the carbocation reacts with a nucleophile (a halide ion) to complete the substitution.
When we convert an alcohol to an alkyl halide, we carry out the reaction in the presence of acid and in the presence of halide ions, and not at elevated temperature. Halide ions are good nucleophiles (they are much stronger nucleophiles than water), and since halide ions are present in high concentration, most of the carbocations react with an electron pair of a halide ion to form a more stable species, the alkyl halide product. The overall result is an \(S_n1\) reaction.
Primary Alcohols
Not all acid-catalyzed conversions of alcohols to alkyl halides proceed through the formation of carbocations. Primary alcohols and methanol react to form alkyl halides under acidic conditions by an SN2 mechanism.
In these reactions the function of the acid is to produce a protonated alcohol. The halide ion then displaces a molecule of water (a good leaving group) from carbon; this produces an alkyl halide:
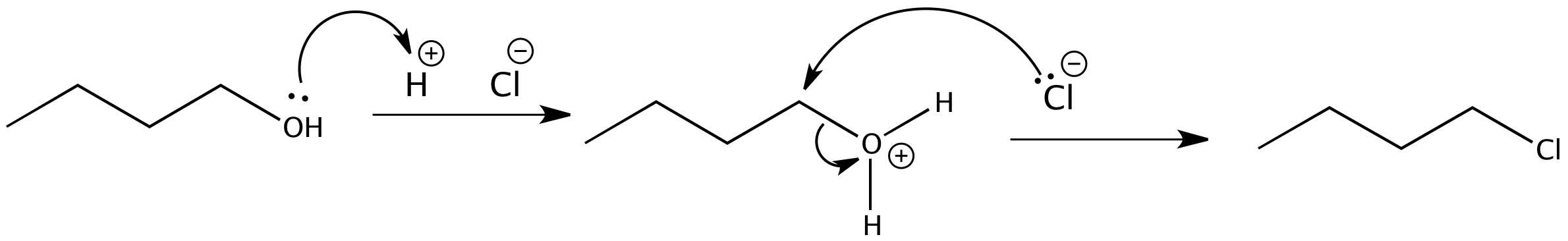
Again, acid is required. Although halide ions (particularly iodide and bromide ions) are strong nucleophiles, they are not strong enough to carry out substitution reactions with alcohols themselves. Direct displacement of the hydroxyl group does not occur because the leaving group would have to be a strongly basic hydroxide ion:
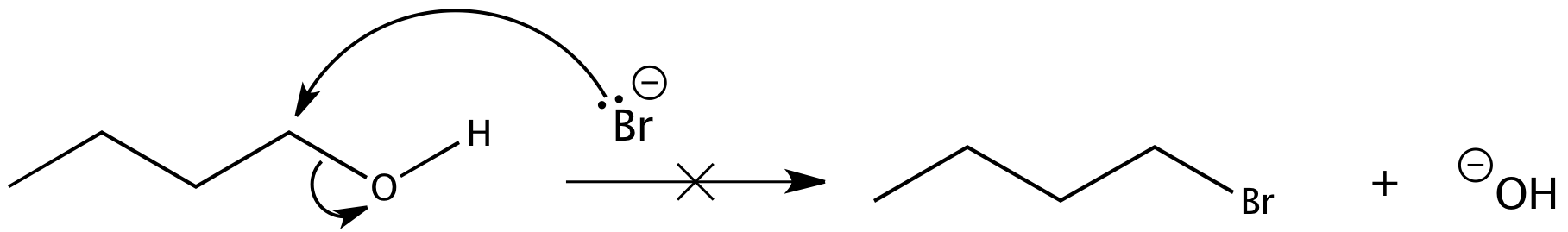
We can see now why the reactions of alcohols with hydrogen halides are acid-promoted.
Carbocation rearrangements are extremely common in organic chemistry reactions are are defined as the movement of a carbocation from an unstable state to a more stable state through the use of various structural reorganizational "shifts" within the molecule. Once the carbocation has shifted over to a different carbon, we can say that there is a structural isomer of the initial molecule. However, this phenomenon is not as simple as it sounds.
Carbocation Rearrangement in the SN1 Reaction
Whenever alcohols are subject to transformation into various carbocations, the carbocations are subject to a phenomenon known as carbocation rearrangement. A carbocation, in brief, holds the positive charge in the molecule that is attached to three other groups and bears a sextet rather than an octet. However, we do see carbocation rearrangements in reactions that do not contain alcohol as well. Those, on the other hand, require more difficult explanations than the two listed below. There are two types of rearrangements: hydride shift and alkyl shift. These rearrangements usualy occur in many types of carbocations. Once rearranged, the molecules can also undergo further unimolecular substitution (SN1) or unimolecular elimination (E1). Though, most of the time we see either a simple or complex mixture of products. We can expect two products before undergoing carbocation rearrangement, but once undergoing this phenomenon, we see the major product.
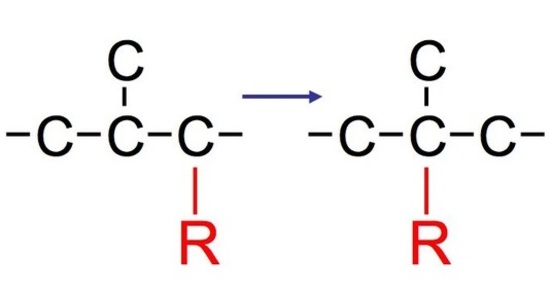
Hydride Shift
Whenever a nucleophile attacks some molecules, we typically see two products. However, in most cases, we normally see both a major product and a minor product. The major product is typically the rearranged product that is more substituted (aka more stable). The minor product, in contract, is typically the normal product that is less substituted (aka less stable).
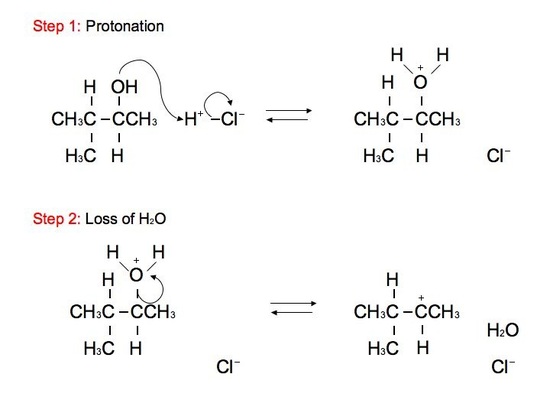
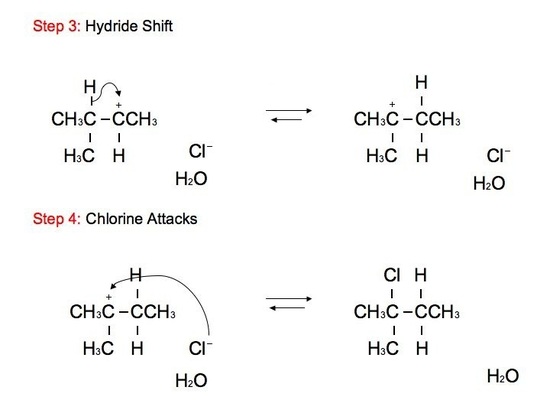
The reaction: We see that the formed carbocations can undergo rearrangements called hydride shift. This means that the two electron hydrogen from the unimolecular substitution moves over to the neighboring carbon. We see the phenomenon of hydride shift typically with the reaction of an alcohol and hydrogen halides, which include HBr, HCl, and HI. HF is typically not used because of its instability and its fast reactivity rate. Below is an example of a reaction between an alcohol and hydrogen chloride:
.jpg?revision=1&size=bestfit&width=410&height=306)
GREEN (Cl) = nucleophile BLUE (OH) = leaving group ORANGE (H) = hydride shift proton RED(H) = remaining proton
The alcohol portion (-OH) has been substituted with the nucleophilic Cl atom. However, it is not a direct substitution of the OH atom as seen in SN2 reactions. In this SN1 reaction, we see that the leaving group, -OH, forms a carbocation on Carbon #3 after receiving a proton from the nucleophile to produce an alkyloxonium ion. Before the Cl atom attacks, the hydrogen atom attached to the Carbon atom directly adjacent to the original Carbon (preferably the more stable Carbon), Carbon #2, can undergo hydride shift. The hydrogen and the carbocation formally switch positions. The Cl atom can now attack the carbocation, in which it forms the more stable structure because of hyperconjugation. The carbocation, in this case, is most stable because it attaches to the tertiary carbon (being attached to 3 different carbons). However, we can still see small amounts of the minor, unstable product. The mechanism for hydride shift occurs in multiple steps that includes various intermediates and transition states. Below is the mechanism for the given reaction above:
Contributors
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)
Jim Clark (Chemguide.co.uk)
- Layne A. Morsch (University of Illinois Springfield)