8.5 Drawing Lewis Dot Structures
- Page ID
- 169773
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
Lewis Dot Structures, Electron and Molecular Geometry
Exercise \(\PageIndex{1}\)
Draw the best Lewis Dot Structure for each of the following species. Give the name of the electronic arrangement and the name for the molecular geometry for each of the species.
BeF2, BCl3, CCl4, PBr5, SI6, BH2–, NI3, ClF4+, SF5–
- Answer
-
Species Name Lewis Dot Structure Electron Geometry Molecular Geometry BeF2 linear linear BCl3 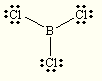
trigonal planar trigonal planar CCl4 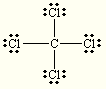
tetrahedral tetrahedral PBr5 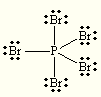
trigonal bipyramidal trigonal bipyramidal SI6 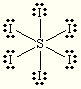
octahedral octahedral BH2– 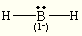
trigonal planar bent NI3 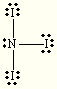
tetrahedral trigonal pyramidal ClF4+ trigonal bipyramidal see saw SF5– octahedral square pyramidal

