3.5: Calorimetry
- Page ID
- 393503
- To use calorimetric data to calculate enthalpy changes.
Thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between objects or substances can be measured. Calorimetry describes a set of techniques employed to measure enthalpy changes in chemical processes using devices called calorimeters. To have any meaning, the quantity that is actually measured in a calorimetric experiment, the change in the temperature of the device, must be related to the heat evolved or consumed in a chemical reaction. We begin this section by explaining how the flow of thermal energy affects the temperature of an object.
Heat Capacity
We have seen that the temperature of an object changes when it absorbs or loses thermal energy. The magnitude of the temperature change depends on both the amount of thermal energy transferred (q) and the heat capacity of the object. Its heat capacity (C) is the amount of energy needed to raise the temperature of the object exactly 1°C; the units of C are joules per degree Celsius (J/°C). Note that a degree Celsius is exactly the same as a Kelvin, so the heat capacities can be expresses equally well, and perhaps a bit more correctly in SI, as joules per Kelvin, J/K
The change in temperature (\(ΔT\)) is
\[ \Delta T = \dfrac{q}{C} \label{3.5.1} \]
where q is the amount of heat (in joules), C is the heat capacity (in joules per degree Celsius), and \(ΔT\) is \(T_{final} − T_{initial}\) (in degrees Celsius). Note that \(ΔT\) is always written as the final temperature minus the initial temperature.
Since the scaling for Kelvin (K) and degrees Celsius (°C) are exactly the same, the DIFFERENCE
\[ΔT = T_{final} − T_{initial} \nonumber \]
is the same is either is used for temperature calculations, but make sure not to mix these two temperature units for BOTH \(T_{final}\) and \(T_{initial}\).
The value of \(C\) is intrinsically a positive number, but \(ΔT\) and \(q\) can be either positive or negative, and they both must have the same sign. If \(ΔT\) and \(q\) are positive, then heat flows from the surroundings into an object. If \(ΔT\) and \(q\) are negative, then heat flows from an object into its surroundings.
The heat capacity of an object depends on both its mass and its composition. For example, doubling the mass of an object doubles its heat capacity. Consequently, the amount of substance must be indicated when the heat capacity of the substance is reported. The molar heat capacity (\(c_p\)) is the amount of energy needed to increase the temperature of 1 mol of a substance by 1°C; the units of \(c_p\) for molar heat capacity are thus J/(mol°C).The subscript p indicates that the value was measured at constant pressure. The specific heat capacity (cp) is the amount of energy needed to increase the temperature of 1 g of a substance by 1°C; its units are thus J/(g°C).
We can relate the quantity of a substance, the amount of heat transferred, its specific heat, and the temperature change either via moles:
\[q = nc_pΔT \label{3.5.2} \]
where \(n\) is the number of moles of substance and \(c_p\) is the molar heat capacity or via mass:
\[q = mc_pΔT \label{3.5.3} \]
where \(m\) is the mass of substance in grams and \(c_p\) is the specific heat capacity.
The specific heat capacities of some common substances are given in Table \(\PageIndex{1}\). Note that the specific heat values of most solids are less than 1 J/(g°C), whereas those of most liquids are about 2 J/(g°C). Water in its solid and liquid states is an exception. The heat capacity of ice is twice as high as that of most solids; the heat capacity of liquid water, 4.18 J/(g°C), is one of the highest known. More values are given in the Reference Table T4.
| Compound | Specific Heat [J/(g°C)] | Compound | Specific Heat [J/(g°C)] |
|---|---|---|---|
| H2O (s) (-10oC) | 2.05 | Al(s) | 0.897 |
| H2O (l) | 4.18 | Fe(s) | 0.449 |
| H2O (g) | 2.062 | Cu(s) | 0.385 |
| CH3OH (methanol) | 2.531 | Au(s) | 0.129 |
| CH3CH2OH (ethanol) | 2.438 | Hg(l) | 0.140 |
| n-C6H14 (n-hexane) | 2.270 | NaCl(s) | 0.864 |
| C6H6 (benzene) | 1.741 | MgO(s) | 0.918 |
| C(s) (graphite) | 0.709 | SiO2(s) (quartz) | 0.742 |
| C(s) (diamond) | 0.509 | CaCO3(s) (calcite) | 0.915 |
The high specific heat of liquid water has important implications for life on Earth. A given mass of water releases more than five times as much heat for a 1°C temperature change as does the same mass of limestone or granite. Consequently, coastal regions of our planet tend to have less variable climates than regions in the center of a continent. After absorbing large amounts of thermal energy from the sun in summer, the water slowly releases the energy during the winter, thus keeping coastal areas warmer than otherwise would be expected. Water’s capacity to absorb large amounts of energy without undergoing a large increase in temperature also explains why swimming pools and waterbeds are usually heated. Heat must be applied to raise the temperature of the water to a comfortable level for swimming or sleeping and to maintain that level as heat is exchanged with the surroundings. Moreover, because the human body is about 70% water by mass, a great deal of energy is required to change its temperature by even 1 °C. Consequently, the mechanism for maintaining our body temperature at about 37 °C does not have to be as finely tuned as would be necessary if our bodies were primarily composed of a substance with a lower specific heat.
A home solar energy storage unit uses 400 L of water for storing thermal energy. On a sunny day, the initial temperature of the water is 22.0 °C. During the course of the day, the temperature of the water rises to 38.0 °C as it circulates through the water wall. How much energy has been stored in the water? (The density of water at 22.0 °C is 0.998 g/mL.)

Given: volume and density of water and initial and final temperatures
Asked for: amount of energy stored
Strategy:
- Use the density of water at 22.0 °C to obtain the mass of water (\(m\)) that corresponds to 400 L of water. Then compute \(ΔT\) for the water.
- Determine the amount of heat absorbed by substituting values for \(m\), \(c_p\), and \(ΔT\) into Equation \(\ref{3.5.3}\).
Solution:
A The mass of water is
\[ \begin{align*} \text{mass of } \ce{H2O} &= 400 \; \cancel{L}\left ( \dfrac{1000 \; \cancel{mL}}{1 \; \cancel{L}} \right ) \left ( \dfrac{0.998 \; g}{1 \; \cancel{mL}} \right ) \\[4pt] &= 3.99\times 10^{5}g\; \ce{H2O} \end{align*} \nonumber \]
The temperature change (\(ΔT\)) is
\[38.0 ^oC − 22.0 ^oC = +16.0^oC. \nonumber \]
B From Table \(\PageIndex{1}\), the specific heat of water is 4.18 J/(g°C). From Equation \(\ref{3.5.3}\), the heat absorbed by the water is thus
\[ \begin{align*} q &=mc_{p}\Delta T \\[4pt] &= \left ( 3.99 \times 10^{5} \; \cancel{g} \right )\left ( \dfrac{4.18 \; J}{\cancel{g}\cdot \cancel{^{o}C}} \right ) \left ( 16.0 \; \cancel{^{o}C} \right ) \\[4pt] &= 2.67 \times 10^{7}\,J = 2.67 \times 10^{4}\,kJ \end{align*} \nonumber \]
Both \(q\) and \(ΔT\) are positive, consistent with the fact that the water has absorbed energy.
Some solar energy devices used in homes circulate air over a bed of rocks that absorb thermal energy from the sun. If a house uses a solar heating system that contains 2500 kg of sandstone rocks, what amount of energy is stored if the temperature of the rocks increases from 20.0 °C to 34.5 °C during the day? Assume that the specific heat of sandstone is the same as that of quartz (\(\ce{SiO2}\)) in Table \(\PageIndex{1}\).
- Answer
-
\(2.7 \times 10^4 \,kJ\)
Even though the mass of sandstone is more than six times the mass of the water in Example \(\PageIndex{1}\), the amount of thermal energy stored is the same to two significant figures.
When two objects at different temperatures are placed in contact, heat flows from the warmer object to the cooler one until the temperature of both objects is the same. The law of conservation of energy says that the total energy cannot change during this process:
\[q_{cold} + q_{hot} = 0 \label{3.5.4} \]
The equation implies that the amount of heat that flows from a warmer object is the same as the amount of heat that flows into a cooler object. Because the direction of heat flow is opposite for the two objects, the sign of the heat flow values must be opposite:
\[q_{cold} = −q_{hot} \label{3.5.5} \]
Thus heat is conserved in any such process, consistent with the law of conservation of energy.
Equation \(\ref{3.5.5}\) argues that the amount of heat lost by a warmer object equals the amount of heat gained by a cooler object.
Substituting for q from Equation \(\ref{3.5.3}\) into Equation \(\ref{3.5.5}\) gives
\[ \left [ mc_{p} \Delta T \right ] _{cold} + \left [ mc_{p} \Delta T \right ] _{hot}=0 \label{3.5.6} \]
which can be rearranged to give
\[ \left [ mc_{p} \Delta T \right ] _{cold} = - \left [ mc_{p} \Delta T \right ] _{hot} \label{3.5.7} \]
When two objects initially at different temperatures are placed in contact, we can use Equation \(\ref{3.5.7}\) to calculate the final temperature if we know the chemical composition and mass of the objects.
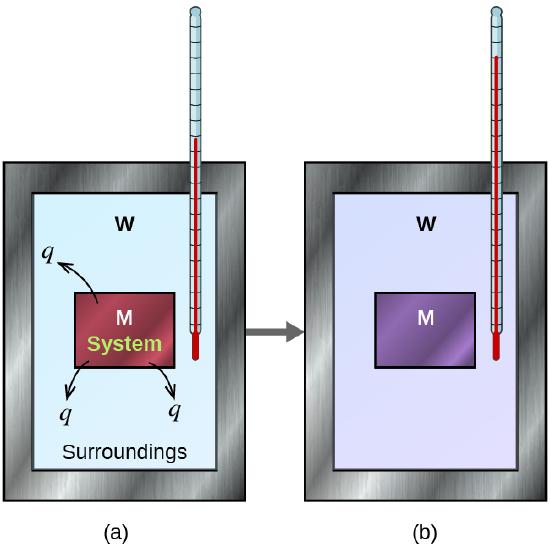
Figure \(\PageIndex{2}\): In a simple calorimetry process, (a) heat, q, is transferred from the hot metal, M, to the cool water, W, until (b) both are at the same temperature.
If a 30.0 g piece of copper pipe at 80.0 °C is placed in 100.0 g of water at 27.0 °C, what is the final temperature? Assume that no heat is transferred to the surroundings.
Given: mass and initial temperature of two objects
Asked for: final temperature
Strategy: Using Equation \(\ref{3.5.7}\) and writing \(ΔT\) as \(T_{final} − T_{initial}\) for both the copper and the water, substitute the appropriate values of \(m\), \(c_p\), and \(T_{initial}\) into the equation and solve for \(T_{final}\).
Solution
We can adapt Equation \(\ref{3.5.7}\) to solve this problem, remembering that \(ΔT\) is defined as \(T_{final} − T_{initial}\):\[ \left [ mc_{p} \left (T_{final} - T_{initial} \right ) \right ] _{Cu} + \left [ mc_{p} \left (T_{final} - T_{initial} \right ) \right ] _{H_{2}O} =0 \nonumber \]
Substituting the data provided in the problem and Table \(\PageIndex{1}\) gives
\[ \newcommand{\celsius}{\,^\circ\text{C}} \begin{align*} \left [ \left (30 \; g \right ) \left (0.385 \; J \right ) \left (T_{final} - 80.0 \celsius \right ) \right ] _{Cu} + \left [ (100\,g) (4.18\, J/\celsius) \left (T_{final} - 27.0 \celsius \right ) \right ] _{\ce{H2O}} &=0 \\[4pt] \left[T_{final}\left ( 11.6 \; J/ \celsius \right ) -924 \; J \right] + \left[ T_{final}\left ( 418.4 \; J/\celsius \right ) -11,300 \; J \right]&= 0 \\[4pt] T_{final}\left ( 430 \; J/\left ( g \cdot \celsius \right ) \right ) &= 12,224 \; J \\[4pt] T_{final} &= 28.4 \; \celsius \end{align*} \nonumber \]
It is expected that the water will increase in temperature since the added copper pipe was hotter. However, the amount of the increase is not great and that is because there is more mass of water (three-fold), but mostly because the greater specific heat of water (order of magnitude) vs. copper.
If a 14.0 g chunk of gold at 20.0°C is dropped into 25.0 g of water at 80.0 °C, what is the final temperature if no heat is transferred to the surroundings?
- Answer
-
80.0°C
A 28.0 g chunk of aluminum is dropped into 100.0 g of water with an initial temperature of 20.0 °C. If the final temperature of the water is 24.0 °C, what was the initial temperature of the aluminum? (Assume that no heat is transferred to the surroundings.)
- Answer
-
90.6°C
In Example \(\PageIndex{1}\), radiant energy from the sun was used to raise the temperature of water. A calorimetric experiment uses essentially the same procedure, except that the thermal energy change accompanying a chemical reaction is responsible for the change in temperature that takes place in a calorimeter. If the reaction releases heat (\(q_{rxn} < 0\)), then heat is absorbed by the calorimeter (\(q_{calorimeter} > 0\)) and its temperature increases. Conversely, if the reaction absorbs heat (\(q_{rxn} > 0\)), then heat is transferred from the calorimeter to the system (\(q_{calorimeter} < 0\)) and the temperature of the calorimeter decreases. In both cases, the amount of heat absorbed or released by the calorimeter is equal in magnitude and opposite in sign to the amount of heat produced or consumed by the reaction. The heat capacity of the calorimeter or of the reaction mixture may be used to calculate the amount of heat released or absorbed by the chemical reaction. The amount of heat released or absorbed per gram or mole of reactant can then be calculated from the mass of the reactants.
Conservation of Energy: The Movement of Heat between Substances: Conservation of Energy: The Movement of Heat between Substances, YouTube(opens in new window) [youtu.be]
Constant-Pressure Calorimetry
Because \(ΔH\) is defined as the heat flow at constant pressure, measurements made using a constant-pressure calorimeter (a device used to measure enthalpy changes in chemical processes at constant pressure) give \(ΔH\) values directly. This device is particularly well suited to studying reactions carried out in solution at a constant atmospheric pressure. A “student” version, called a coffee-cup calorimeter (Figure \(\PageIndex{2}\)), is often encountered in general chemistry laboratories. Commercial calorimeters operate on the same principle, but they can be used with smaller volumes of solution, have better thermal insulation, and can detect a change in temperature as small as several millionths of a degree (10−6 °C). Because the heat released or absorbed at constant pressure is equal to \(ΔH\), the relationship between heat and \(ΔH_{rxn}\) is
\[ \Delta H_{rxn}=q_{rxn}=-q_{calorimeter}=-mc_{p} \Delta T \label{3.5.8} \]
The use of a constant-pressure calorimeter is illustrated in Example \(\PageIndex{3}\).

When 5.03 g of solid potassium hydroxide are dissolved in 100.0 mL of distilled water in a coffee-cup calorimeter, the temperature of the liquid increases from 23.0 °C to 34.7 °C. The density of water in this temperature range averages 0.9969 g/cm3. What is \(ΔH_{soln}\) (in kilojoules per mole)? Assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of the solution is the same as the specific heat of pure water.
Given: mass of substance, volume of solvent, and initial and final temperatures
Asked for: ΔHsoln
Strategy:
- Calculate the mass of the solution from its volume and density and calculate the temperature change of the solution.
- Find the heat flow that accompanies the dissolution reaction by substituting the appropriate values into Equation \ref{3.5.8}.
- Use the molar mass of \(\ce{KOH}\) to calculate \(ΔH_{soln}\).
Solution:
A To calculate ΔHsoln, we must first determine the amount of heat released in the calorimetry experiment. The mass of the solution is
\[ \left (100.0 \; \cancel{mL}\; \ce{H2O} \right ) \left ( 0.9969 \; g/ \cancel{mL} \right )+ 5.03 \; g \; \ce{KOH}=104.72 \; g \nonumber \]
The temperature change is therefore
\[(34.7\, ^oC − 23.0 \,^oC) = +11.7\, ^oC. \nonumber \]
B Because the solution is not very concentrated (approximately 0.9 M), we assume that the specific heat of the solution is the same as that of water. The heat flow that accompanies dissolution is thus
\[ \begin{align*} q_{calorimeter}&=mc_{p} \Delta T \\[4pt] &=\left ( 104.72 \; \cancel{g} \right ) \left ( \dfrac{4.184 \; J}{\cancel{g}\cdot \cancel{^{o}C}} \right )\left ( 11.7 \; ^{o}C \right ) \\[4pt] &=5130 \; J = 5.13 \; kJ \end{align*} \nonumber \]
The temperature of the solution increased because heat was absorbed by the solution (\(q > 0\)). Where did this heat come from? It was released by \(\ce{KOH}\) dissolving in water. From Equation \(\ref{3.5.8}\), we see that
\[ΔH_{rxn} = −q_{calorimeter} = −5.13\, kJ \nonumber \]
This experiment tells us that dissolving 5.03 g of \(\ce{KOH}\) in water is accompanied by the release of 5.13 kJ of energy. Because the temperature of the solution increased, the dissolution of \(\ce{KOH}\) in water must be exothermic.
C The last step is to use the molar mass of \(\ce{KOH}\) to calculate \(ΔH_{soln}\), i.e., the heat released when dissolving 1 mol of \(\ce{KOH}\):
\[ \begin{align*} \Delta H_{soln} &= \left ( \dfrac{5.13 \; kJ}{5.03 \; \cancel{g}} \right )\left ( \dfrac{56.11 \; \cancel{g}}{1 \; mol} \right ) \\[4pt] &=-57.2 \; kJ/mol \end{align*} \nonumber \]
A coffee-cup calorimeter contains 50.0 mL of distilled water at 22.7 °C. Solid ammonium bromide (3.14 g) is added and the solution is stirred, giving a final temperature of 20.3°C. Using the same assumptions as in Example \(\PageIndex{3}\), find \(ΔH_{soln}\) for \(\ce{NH4Br }\) (in kilojoules per mole).
- Answer
-
16.6 kJ/mol
Conservation of Energy: Coffee Cup Calorimetry: Conservation of Energy: Coffee Cup Calorimetry, YouTube(opens in new window) [youtu.be] (opens in new window)
Heat Produced by an Exothermic Reaction
When 50.0 mL of 1.00 M HCl(aq) and 50.0 mL of 1.00 M NaOH(aq), both at 22.0 °C, are added to a coffee cup calorimeter, the temperature of the mixture reaches a maximum of 28.9 °C. What is the approximate amount of heat produced by this reaction?
HCl(aq)+NaOH(aq) ⟶ NaCl(aq)+H2O(l)
Solution
To visualize what is going on, imagine that you could combine the two solutions so quickly that no reaction took place while they mixed; then after mixing, the reaction took place. At the instant of mixing, you have 100.0 mL of a mixture of HCl and NaOH at 22.0 °C. The HCl and NaOH then react until the solution temperature reaches 28.9 °C.
The heat given off by the reaction is equal to that taken in by the solution. Therefore:
\(q_{reaction} = -q_{solution}\)
(It is important to remember that this relationship only holds if the calorimeter does not absorb any heat from the reaction, and there is no heat exchange between the calorimeter and the outside environment.)
Next, we know that the heat absorbed by the solution depends on its specific heat, mass, and temperature change:
\(q_{solution} = (c_{p}×m×ΔT)_{solution}\)
To proceed with this calculation, we need to make a few more reasonable assumptions or approximations. Since the solution is aqueous, we can proceed as if it were water in terms of its specific heat and mass values. The density of water is approximately 1.0 g/mL, so 100.0 mL has a mass of about 1.0 × 102 g (two significant figures). The specific heat of water is approximately 4.184 J/g °C, so we use that for the specific heat of the solution. Substituting these values gives:
\(q_{solution} = (4.184J/g °C)(1.0×102g)(28.9°C−22.0°C)=2.9\times 10^{3}J\)
Finally, since we are trying to find the heat of the reaction, we have:
\(q_{reaction} = -q_{solution}=−2.9\times 10^{3}J\)
The negative sign indicates that the reaction is exothermic. It produces 2.9 kJ of heat
Thermochemistry of Hand Warmers
When working or playing outdoors on a cold day, you might use a hand warmer to warm your hands. A common reusable hand warmer contains a supersaturated solution of NaC2H3O2 (sodium acetate) and a metal disc. Bending the disk creates nucleation sites around which the metastable NaC2H3O2 quickly crystallizes (a later chapter on solutions will investigate saturation and supersaturation in more detail).
The process NaC2H3O2(aq)⟶NaC2H3O2(s) is exothermic, and the heat produced by this process is absorbed by your hands, thereby warming them (at least for a while). If the hand warmer is reheated, the NaC2H3O2 redissolves and can be reused.

Another common hand warmer produces heat when it is ripped open, exposing iron and water in the hand warmer to oxygen in the air. One simplified version of this exothermic reaction is 2Fe(s)+32O2(g)⟶Fe2O3(s). Salt in the hand warmer catalyzes the reaction, so it produces heat more rapidly; cellulose, vermiculite, and activated carbon help distribute the heat evenly. Other types of hand warmers use lighter fluid (a platinum catalyst helps lighter fluid oxidize exothermically), charcoal (charcoal oxidizes in a special case), or electrical units that produce heat by passing an electrical current from a battery through resistive wires.
Heat Flow in an Instant Ice Pack
When solid ammonium nitrate dissolves in water, the solution becomes cold. This is the basis for an “instant cold pack”. When 3.21 g of solid NH4NO3 dissolves in 50.0 g of water at 24.9 °C in a calorimeter, the temperature decreases to 20.3 °C. Calculate the value of q for this reaction and explain the meaning of its arithmetic sign. State any assumptions that you made.

- Answer
-
We assume that the calorimeter prevents heat transfer between the solution and its external environment (including the calorimeter itself), therefore:
\(q_{reaction} = -q_{solution}\)
We also assume that the specific heat capacity of the solution is the same as water. so \(c_{p,solution} = 4.184 J/g^{o}C\).
\(q_{reaction} = -q_{solution} = (-c_{p,solution}×m_{solution}×ΔT)_{solution}\)
\(q_{reaction} = -[(4.184 J/g^{o}C) (53.2 g) (20.3 ^{o}C - (-4.6 ^{o}C))]\)
\(q_{reaction} = 1.0 \times 10^{3}J\)
Constant-Volume Calorimetry
Constant-pressure calorimeters are not very well suited for studying reactions in which one or more of the reactants is a gas, such as a combustion reaction. The enthalpy changes that accompany combustion reactions are therefore measured using a constant-volume calorimeter, such as the bomb calorimeter(A device used to measure energy changes in chemical processes. shown schematically in Figure \(\PageIndex{3}\)). The reactant is placed in a steel cup inside a steel vessel with a fixed volume (the “bomb”). The bomb is then sealed, filled with excess oxygen gas, and placed inside an insulated container that holds a known amount of water. Because combustion reactions are exothermic, the temperature of the bath and the calorimeter increases during combustion. If the heat capacity of the bomb and the mass of water are known, the heat released can be calculated.

Because the volume of the system (the inside of the bomb) is fixed, the combustion reaction occurs under conditions in which the volume, but not the pressure, is constant. The heat released by a reaction carried out at constant volume is identical to the change in internal energy (\(ΔU\)) rather than the enthalpy change (\(ΔH\)); \(ΔU\) is related to \(ΔH\) by an expression that depends on the change in the number of moles of gas during the reaction. The difference between the heat flow measured at constant volume and the enthalpy change is usually quite small, however (on the order of a few percent). Assuming that \(ΔU < ΔH\), the relationship between the measured temperature change and \(ΔH_{comb}\) is given in Equation \(\ref{3.5.9}\), where cbomb is the total heat capacity of the steel bomb and the water surrounding it:
\[ \Delta H_{comb} < q_{comb} = q_{calorimeter} = c_{bomb} \Delta T \label{3.5.9} \]
To measure the heat capacity of the calorimeter, we first burn a carefully weighed mass of a standard compound whose enthalpy of combustion is accurately known. Benzoic acid (\(\ce{C6H5CO2H}\)) is often used for this purpose because it is a crystalline solid that can be obtained in high purity. The combustion of benzoic acid in a bomb calorimeter releases 26.38 kJ of heat per gram (i.e., its \(ΔH_{comb} = −26.38\, kJ/g\)). This value and the measured increase in temperature of the calorimeter can be used in Equation \(\ref{3.5.9}\) to determine \(c_{bomb}\). The use of a bomb calorimeter to measure the \(ΔH_{comb}\) of a substance is illustrated in Example \(\PageIndex{4}\).
The combustion of 0.579 g of benzoic acid in a bomb calorimeter caused a 2.08 °C increase in the temperature of the calorimeter. The chamber was then emptied and recharged with 1.732 g of glucose and excess oxygen. Ignition of the glucose resulted in a temperature increase of 3.64°C. What is the \(ΔH_{comb}\) of glucose?
Given: mass and \(ΔT\) for combustion of standard and sample
Asked for: \(ΔH_{comb}\) of glucose
Strategy:
- Calculate the value of \(q_{rxn}\) for benzoic acid by multiplying the mass of benzoic acid by its \(ΔH_{comb}\). Then use Equation \(\ref{3.5.9}\) to determine the heat capacity of the calorimeter (\(c_{bomb}\)) from \(q_{comb}\) and \(ΔT\).
- Calculate the amount of heat released during the combustion of glucose by multiplying the heat capacity of the bomb by the temperature change. Determine the ΔHcomb of glucose by multiplying the amount of heat released per gram by the molar mass of glucose.
Solution:
The first step is to use Equation \(\ref{3.5.9}\) and the information obtained from the combustion of benzoic acid to calculate cbomb. We are given \(ΔT\), and we can calculate qcomb from the mass of benzoic acid:
\[ \begin{align*} q_{comb} &= \left ( 0.579 \; \cancel{g} \right )\left ( -26.38 \; kJ/\cancel{g} \right ) \\[4pt] &= - 15.3 \; kJ \end{align*} \nonumber \]
From Equation \(\ref{3.5.9}\),
\[ \begin{align*} -c_{bomb} &= \dfrac{q_{comb}}{\Delta T} \\[4pt] &= \dfrac{-15.3 \; kJ}{2.08 \; ^{o}C} \\[4pt] &=- 7.34 \; kJ/^{o}C \end{align*} \nonumber \]
B According to the strategy, we can now use the heat capacity of the bomb to calculate the amount of heat released during the combustion of glucose:
\[ \begin{align*} q_{comb} &=-c_{bomb}\Delta T \\[4pt] &= \left ( -7.34 \; kJ/^{o}C \right )\left ( 3.64 \; ^{o}C \right ) \\[4pt] &=- 26.7 \; kJ \end{align*} \nonumber \]
Because the combustion of 1.732 g of glucose released 26.7 kJ of energy, the ΔHcomb of glucose is
\[ \begin{align*} \Delta H_{comb} &=\left ( \dfrac{-26.7 \; kJ}{1.732 \; \cancel{g}} \right )\left ( \dfrac{180.16 \; \cancel{g}}{mol} \right ) \\[4pt] &= -2780 \; kJ/mol \\[4pt] &=2.78 \times 10^{3} \; J/mol \end{align*} \nonumber \]
This result is in good agreement (< 1% error) with the value of \(ΔH_{comb} = −2803\, kJ/mol\) that calculated using enthalpies of formation.
When 2.123 g of benzoic acid is ignited in a bomb calorimeter, a temperature increase of 4.75 °C is observed. When 1.932 g of methylhydrazine (CH3NHNH2) is ignited in the same calorimeter, the temperature increase is 4.64 °C. Calculate the ΔHcomb of methylhydrazine, the fuel used in the maneuvering jets of the US space shuttle.
- Answer
-
−1.30 × 103 kJ/mol
Conservation of Energy: Bomb Calorimetry: Conservation of Energy: Bomb Calorimetry, YouTube(opens in new window) [youtu.be]
Summary
Enthalpy is a state function used to measure the heat transferred from a system to its surroundings or vice versa at constant pressure. Only the change in enthalpy (ΔH) can be measured. A negative ΔH means that heat flows from a system to its surroundings; a positive ΔH means that heat flows into a system from its surroundings. Calorimetry measures enthalpy changes during chemical processes, where the magnitude of the temperature change depends on the amount of heat released or absorbed and on the heat capacity of the system. Calorimetry is the set of techniques used to measure enthalpy changes during chemical processes. It uses devices called calorimeters, which measure the change in temperature when a chemical reaction is carried out. The magnitude of the temperature change depends on the amount of heat released or absorbed and on the heat capacity of the system. The heat capacity (C) of an object is the amount of energy needed to raise its temperature by 1°C; its units are joules per degree Celsius. The specific heat (cp) of a substance is the amount of energy needed to raise the temperature of 1 g of the substance by 1°C, and the molar heat capacity (cp, molar) is the amount of energy needed to raise the temperature of 1 mol of a substance by 1°C. Liquid water has one of the highest specific heats known. Heat flow measurements can be made with either a constant-pressure calorimeter, which gives ΔH values directly, or a bomb calorimeter, which operates at constant volume and is particularly useful for measuring enthalpies of combustion.