Fe Wars: Lactoferrin
- Page ID
- 97951
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Iron Wars
Wars can be started over metals like gold or silver, but the human body wages an internal war against pathogens on a daily basis over a seemingly insignificant metal: iron (see figure 1). While largely known for its rusting abilities, iron is also the catalyst for numerous metabolic processes, a binding agent in molecules like hemoglobin, and is even speculated to be a contributing ion to the early formation of macromolecules that eventually led to life forms.1 However, humans aren’t the only organisms that need this ion as a nutrient source; bacterial species depend on ions like iron to survive.1 As a result of this mutual need, iron wars break out between bacteria and the human body, and each side has used scientific methods to more efficiently acquire this resource.
 Figure 1: Cartoon depicting the “iron wars” between human and bacterial iron-sequestering proteins. Right: E. Faecalis siderophore, ferric hydroxamate. Left: human protein, lactoferrin.
Figure 1: Cartoon depicting the “iron wars” between human and bacterial iron-sequestering proteins. Right: E. Faecalis siderophore, ferric hydroxamate. Left: human protein, lactoferrin.
Enter to the scene lactoferrin, a iron-chelating protein that has an extremely high binding affinity for iron (III).2 Lactoferrin is a member of the transferrin family, a group of proteins that bind specifically to iron and perform various functions in the body (see figure 2).2 Transferrin, while in some cases able to prevent bacterial infection, is used more efficiently as an iron transport protein. Transferrin has a lower binding affinity to iron than lactoferrin does, and a slight dip below biological pH can trigger the protein to release its ion.3 On the other hand, lactoferrin binds to iron with a much higher affinity, and the ion is not known to be released until pH drops to around 2-3.4 A pH of this acidity can be found in the body in places such as inflammation sites, cellular vesicles, or the stomach.4 The ability for lactoferrin to retain iron in a pH far below the biological level of 7.4 makes lactoferrin a more effective tool in the war over iron than transferrin, but less effective as an iron source for the body itself.
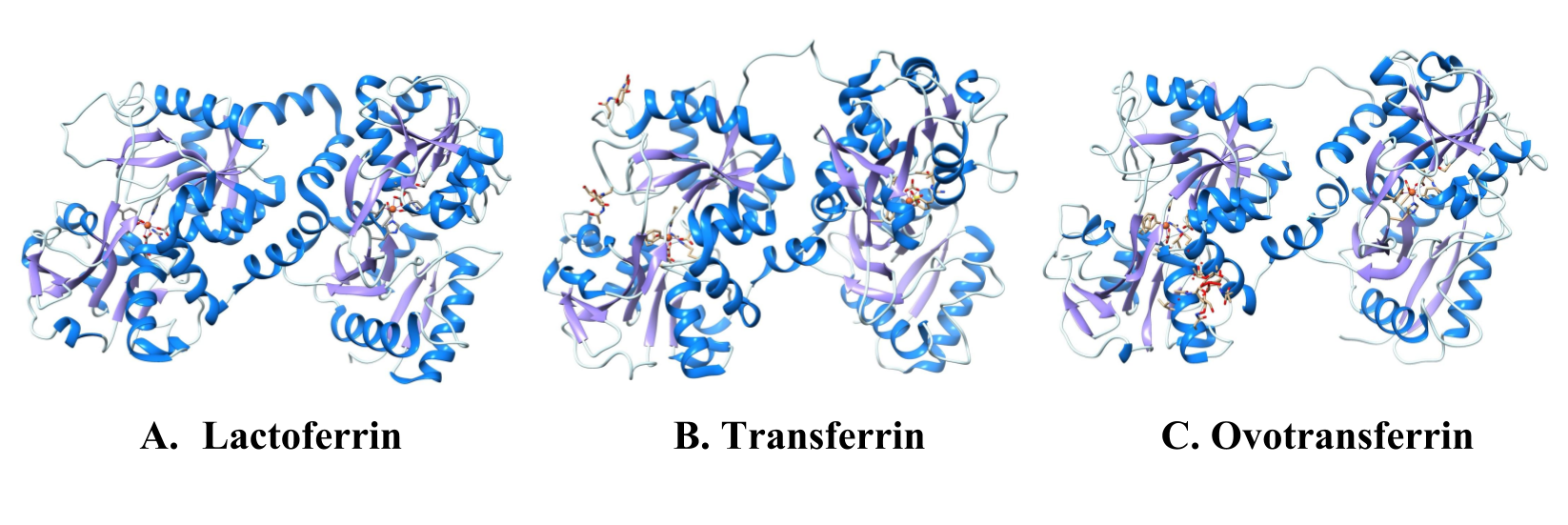 Figure 2: Comparison of the structures of three iron chelating proteins in the transferrin family: Lactoferrin, transferrin, and ovotransferrin. All transferrin proteins bind with high affinity to iron (III) ions, but lactoferrin has the highest binding affinity (KD ~ 10-20 M compared with KD ~ 10-9 M for transferrin). 6,10
Figure 2: Comparison of the structures of three iron chelating proteins in the transferrin family: Lactoferrin, transferrin, and ovotransferrin. All transferrin proteins bind with high affinity to iron (III) ions, but lactoferrin has the highest binding affinity (KD ~ 10-20 M compared with KD ~ 10-9 M for transferrin). 6,10
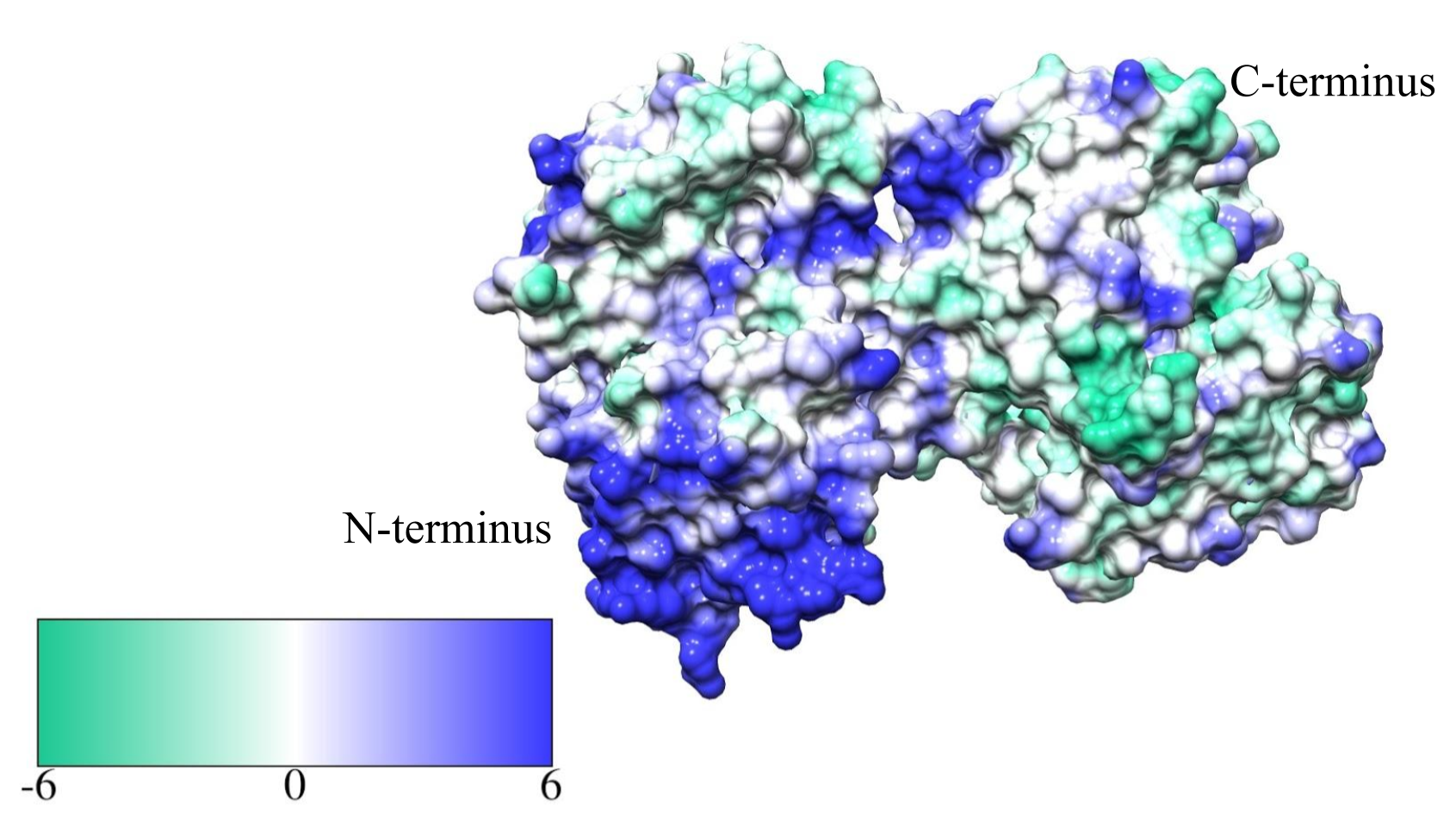
In addition to its antibacterial defenses, lactoferrin performs other functions in the body. The overall structure includes a unique cationic region on its N-terminal (See figure 3).2 This basic region has been found to bind to lipopolysaccharides and glycosaminoglycans.2 This binding feature allows lactoferrin to more directly attack bacteria because lipopolysaccharides and glycosaminoglycans are commonly found in the cell membrane of bacteria, and lactoferrin has been found to bind to the surfaces of bacteria and prevent iron uptake.2 Additionally, this cationic region can bind to DNA, and thus serves as a defense against viruses by binding to viral DNA and preventing its spread through the body.2 Recent research has taken particular interest in using lactoferrin as an oral cancer treatment, because when it is digested in the stomach, it is broken down to the peptide lactoferricin and absorbed into the bloodstream.2 Lactoferricin has been found to prevent cellular growth by inhibiting the function of proteins involved in cellular reproduction.5
Figure 3: The cationic region of lactoferrin is highlighted in blue. This site on the N-terminus provides lactoferrin with unique properties that aid in its antimicrobial defense system.2,5 It is able to bind directly to the surfaces of bacteria, the free DNA of viruses, and even inhibit overproduction of cancer cells.2,5
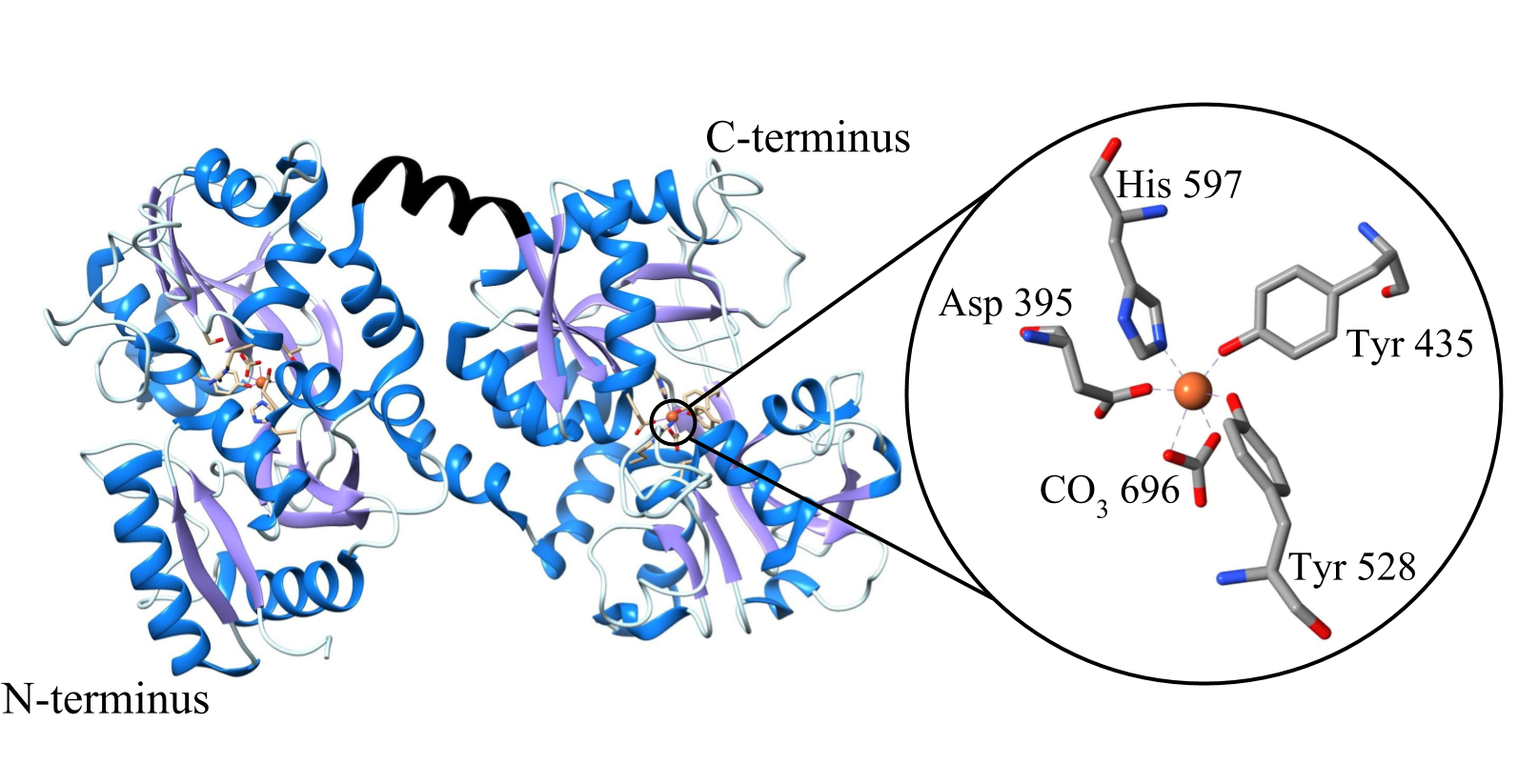
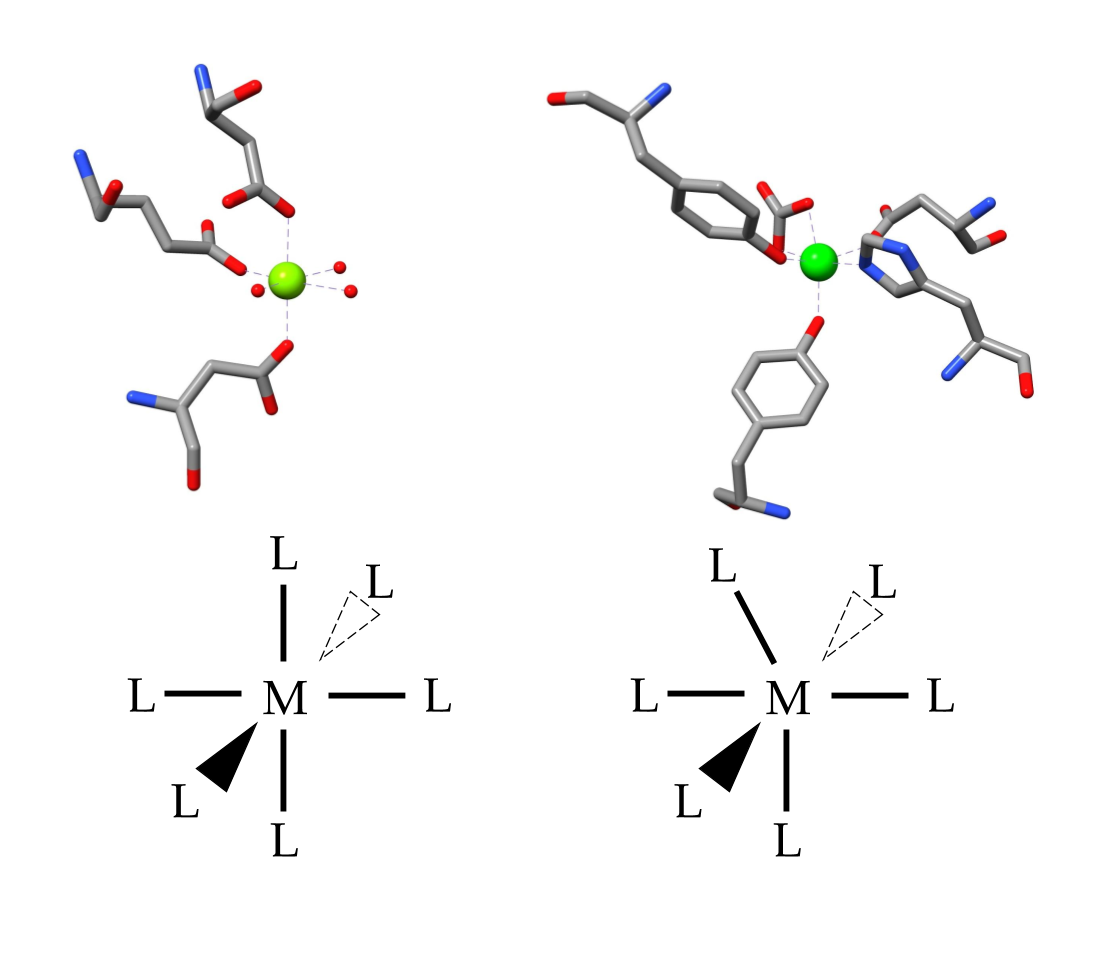
The structure of lactoferrin is essential for its high binding affinity. Lactoferrin has two hemispheres; the N-terminal, and the C-terminal, which are connected by an alpha helix (see figure 4a).2 Each terminal is divided into two lobes, with a “cleft” between them where the metal binding site is located.6 The amino acids found at the binding site are the same in all transferrin proteins, and consists of two phenolate oxygens from two tyrosines, a histidine, an aspartic acid, and a carbonate ion bound to an arginine (see figure 4b).2,4
Figure 4 a-b: a) Molecule lactoferrin pictured, the alpha helix connecting N-terminal to C-terminal is pictured in black. In other transferrin proteins, this connection is a flexible beta sheet. It is predicted that this rigid helix contributes to the conformational changes experienced by lactoferrin that allow it to retain iron ions at a lower pH.6 b) The active binding site of lactoferrin on the C-terminus, but identical amino acids are on the N-terminal. Consists of four amino acids, and a carbonate ion with an iron (III) ion in the center.
It is predicted that the carbonate is the first to bind to the iron ion, followed by the tyrosine groups.6 Then lactoferrin undergoes a conformational change, wherein the N-terminus closes like a hinge, to lock the iron into place.6 The resulting structure brings the iron closer to the histidine and the aspartic acid found at the binding site, and the resulting complex displays an octahedral coordination geometry (see figure 5).6 This final structure is very stable and allows the lactoferrin protein to retain the iron ions in a pH as low as 2-3.2
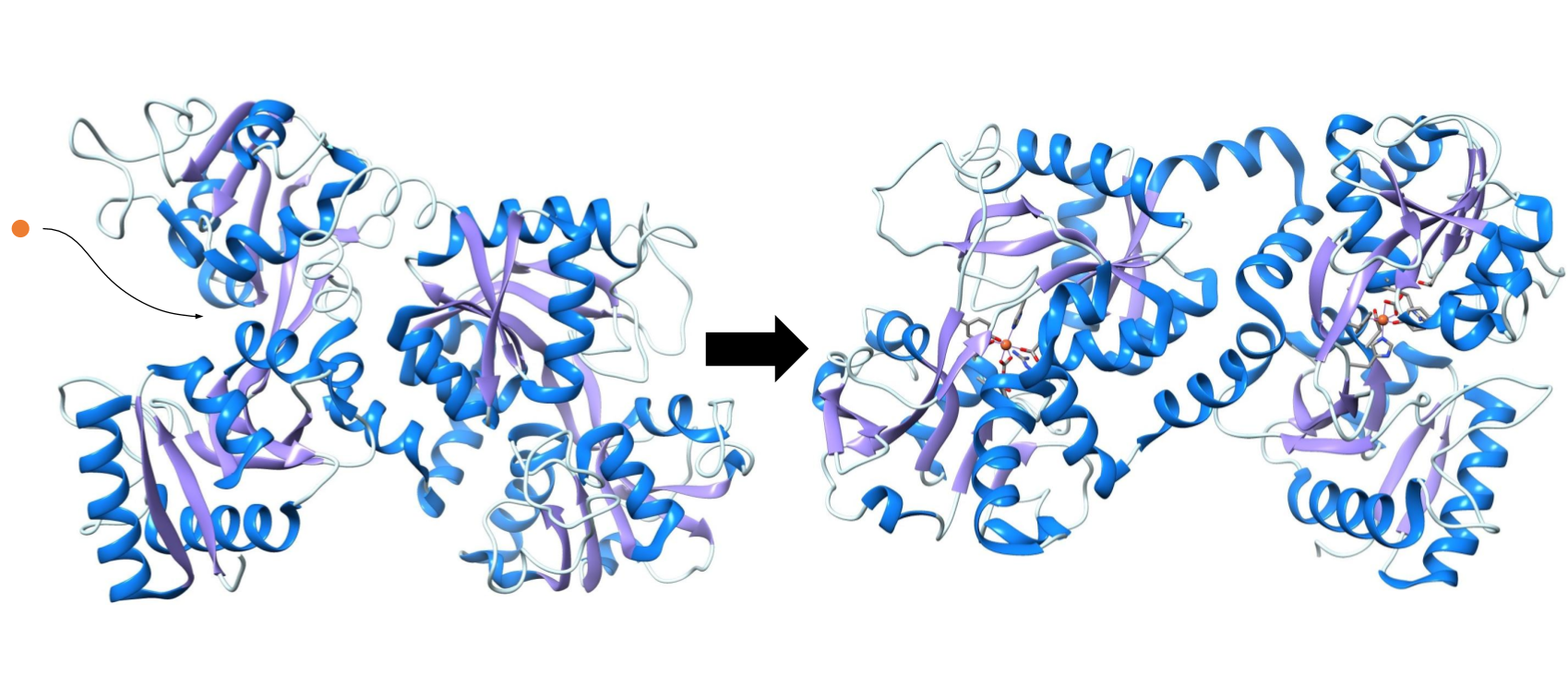
Lactoferrin has high specificity for iron (III) ions. This is likely due to the combination of several different factors, some of which contribute to general stability of the metal complex, and some that lead to the iron (III) preference.
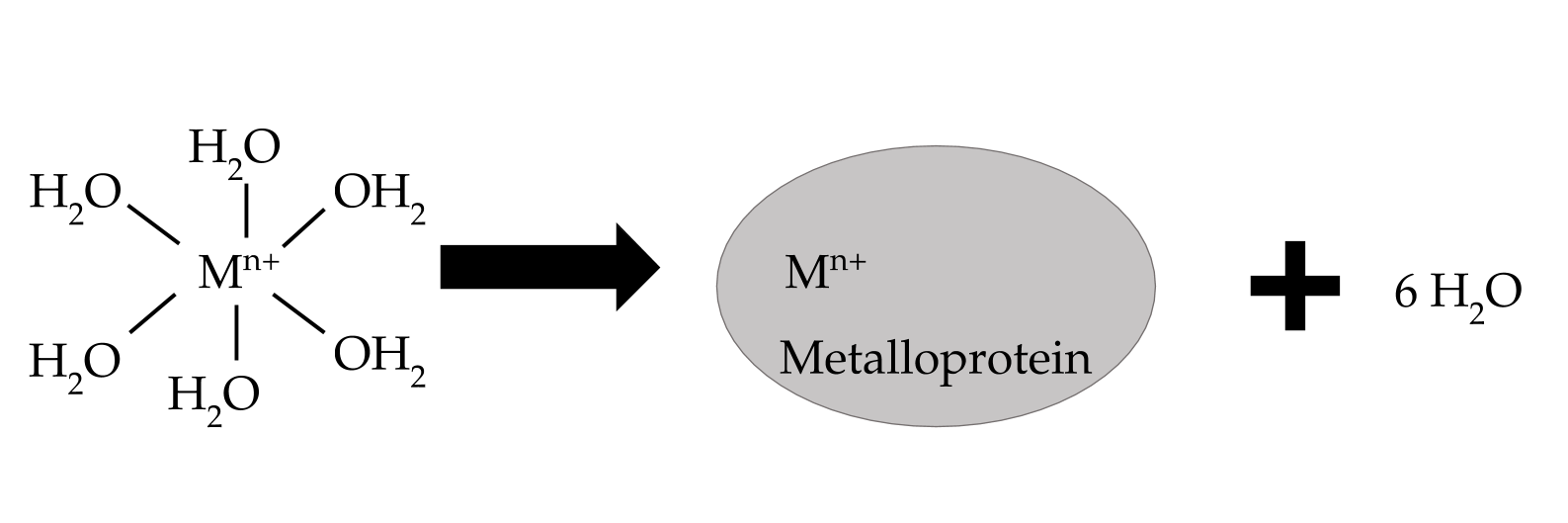
The chelate effect is a thermodynamic factor that contributes to general metal complex stability, because it explains that stability increases as entropy increases. That is to say that as the number of individual molecules in a system increases, so does the stability of a complex. For example, when an ion (Mn+) is not bound to a system or protein in the body, it is likely bound to six water molecules, since this complex is more stable than a charged ion, and there is plenty of water in the body. So, when that ion binds to a single complex with multiple binding atoms, the chelate effect contributes to high stability of the metal-protein complex, because the number of free molecules goes from two to seven (see figure 6). For this reason, it is entropically favorable for iron to form a bond with lactoferrin because it replaces six individual molecules with a multidentate ligand.
Figure 6: When an iron (III) is “free” in the human body, it usually forms bonds to water molecules. The chelate effect explains that increasing the number of free individual molecules is a favorable reaction, so when that ion binds to a protein with multiple binding atoms, it forms a single metal-protein complex and six free water molecules. Thus, the total number of molecules in a system goes from two to seven, which increases the entropy, or chaos.
The chelate effect is a thermodynamic explanation for the stability of the formation of a metalloprotein complex. Hard soft acid-base (HSAB) theory contributes to the iron selectivity of lactoferrin. HSAB theory is a thermodynamic concept that can predict the stability of metal-ligand interactions. Metals are acids, ligands are bases, and generally hard acids interact with hard bases, and soft acids interact with soft bases. Factors such as charge density and characteristics of an interaction (electrostatic vs covalent) contribute to whether a metal or ligand is considered hard or soft. All of the amino acids found in the binding site, with the exception of histidine, prefer hard acids. Since like binds with like, and iron (III) is a hard acid, the site is favorable for iron. However, several other metals are hard acids as well, including magnesium (II), chromium (III), and calcium (II). These other metals are also known to be able to bind to lactoferrin, but are not nearly as common as iron (III), and don’t usually remain bound lower than a pH of 4-5.7
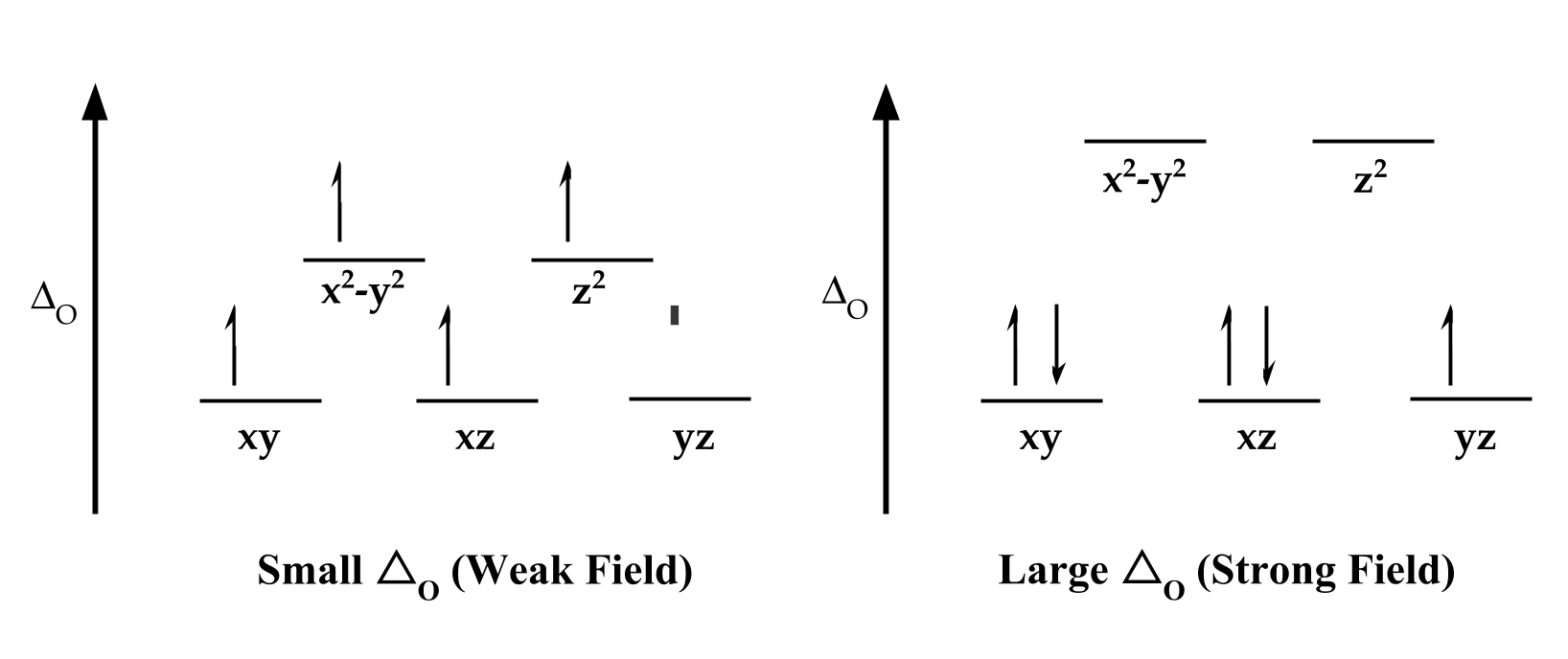
Once lactoferrin has bound to the metal ion, the iron(III)-protein complex that forms is extremely stable, as it is with all transferrin proteins. An important factor in this stability is the geometry of the binding site. The d-orbitals of metals have a large influence on geometry. When a metal binds to a ligand, it can form bonding and antibonding orbitals for electrons to occupy. The difference between the orbitals can influence electron spin and configuration (see figure 7). The energy difference between these d-orbitals can be called the field splitting (△O). When △O is small, the system is said to have a weak field and the electrons are high spin, which means they will occupy the higher energy orbitals before pairing up. On the other hand, when △O is large, the system has a strong field and are low spin, so they will pair up before occupying a higher orbital.
Figure 7: The difference between d-orbitals in metal ions is △o, which influences their electron spin. If △o is small, then it requires less energy for the electrons to move up into a higher energy orbital than it does to pair up, but when △o is large, the jump into the next orbital would be too high. Note that this only impacts d4-d7 electrons.
Some of the factors that contribute to △O include the formal charge of the ion, the location of the ion on the periodic table, and the types of ligands in the system. All of the donor atoms in the binding site of lactoferrin (except histidine) are sigma and pi donors. Sigma and pi donors are typically involved in weak field interactions, which is a smaller distance between d-orbitals (△O). In addition, iron (III) has a location in the fourth row of the periodic table, and △O decreases going up the periodic table. These factors indicate that iron (III) in lactoferrin has a weak field. Since Iron (III) is a d5 electron, a weak field means that iron’s d-electrons will occupy the higher energy orbitals, and won’t pair up.
Ligand field stabilization energy (LFSE) can suggest the preferred geometry of an ion. The LFSE is calculated using the equation LFSE = -0.4x + 0.6y, where x is equal to the number of electrons in lower energy orbitals, and y is equal to the number of electrons in higher energy orbitals. Iron (III) has a LFSE of zero, which means there is no other ligand position that would provide a lower LFSE, and so iron is able to form stable bonds in any geometry configuration. Usually d5 metal ions form an octahedral complex when bound to six ligands (See figure 8). However, in the case of lactoferrin, the carbonate ion causes a slight change in the geometry which results in a distorted octahedral configuration.
Figure 8: Comparison of the molecular geometries of a standard octahedron and the distorted octahedron of lactoferrin. The carbonate ion found in the binding site of lactoferrin causes the ligand position to shift.
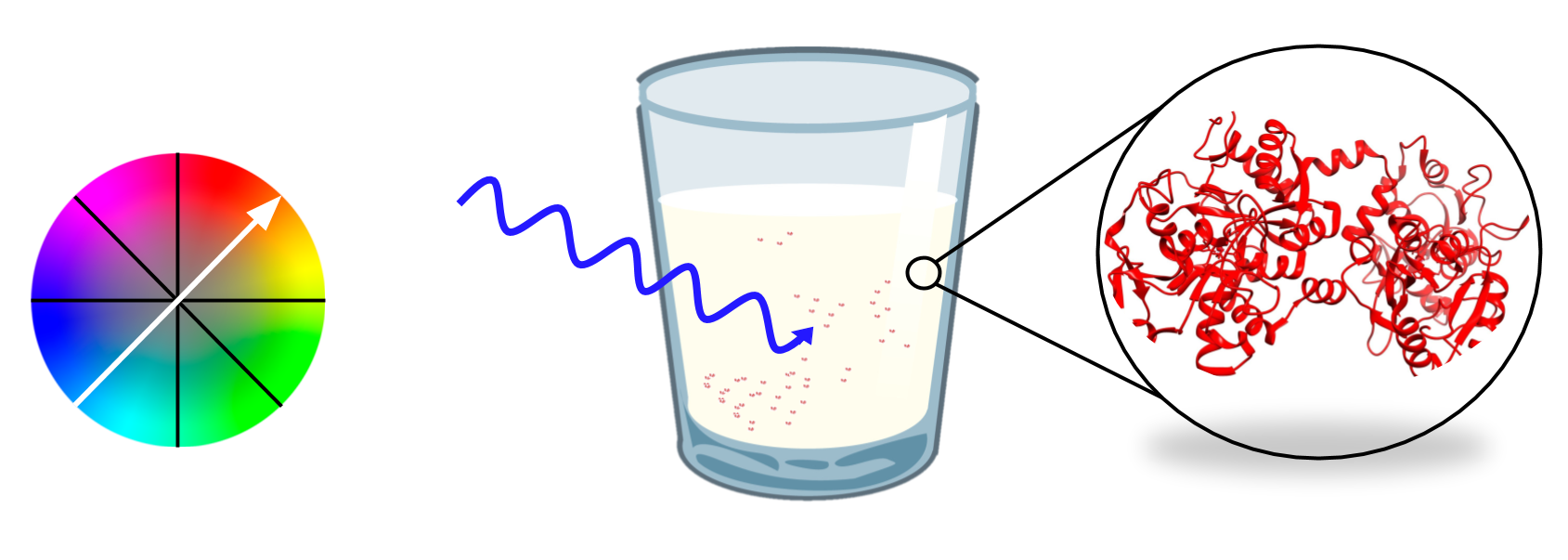
Understanding this concept of crystal field theory provides some insight to how scientists are able to “see” when iron is bound to lactoferrin. The size of △O can be indicative of whether an ion will undergo a d-d transition or a charge transfer transition. When light waves hit a metal ion and the electrons are excited into a higher d-orbital, the ion is undergoing a d-d transition. Charge transfer, on the other hand, can occur with an exchange of electrons from either the metal to the ligand (MLCT) or from the ligand to the metal (LMCT). In the case of lactoferrin, the majority of the ligands are pi-donors, which is indicative of a ligand to metal charge transfer because the ligand has remaining pi-electrons in its orbitals. The lightwaves excite these electrons into a higher binding orbital of the metal, which produce vibrant colors and strong spectroscopic bands.
Research found the extinction coefficient for the system to be rather large, around 2,500 M-1 at a wavelength of 464 nm, which is highly indicative of a charge transfer transition. In addition, the light absorption for lactoferrin is found to be λ=465 nm, which corresponds to the wavelength of blue light. Since colors appear in color as the opposite of the wavelength they absorb (see figure 9), lactoferrin appears as a red-orange color, which is in conjunction with observations initially made of lactoferrin describing it as a red substance found in milk.2,8
Lactoferrin’s ability to sequester iron from its environment makes it useful as a defense protein in the innate immune system. After both lobes have taken up an iron ion, it can take a pH as low as 2-3 to remove it. Since bacteria are often not this acidic, but still require iron to perform biological functions, lactoferrin essentially starves bacteria from their nutrition source. Most bacteria are defeated by this tactic, with some notable exceptions.
Some bacteria, such as enterococci, have developed tactics to fight against iron sequestering proteins, including lactoferrin.9 These tactics include the use of siderophores, ferric reduction, and proteases that can cleave lactoferrin. Siderophores are essentially bacteria’s version of lactoferrin, and are also able to bind to iron (III) with high affinity by using the chelate effect the same way lactoferrin does. The method of iron reduction, on the other hand, utilizes HSAB theory, and changes the hard acid iron (III) into an intermediate, which would make the ion less stable in its environment. Finally, proteases that can cleave proteins like lactoferrin are able to change the binding site, which ultimately impacts the ligands bound to the iron. This would cause a change in the geometry of the binding site and cause the iron to “pop out” of place.9
While bacteria use these defenses to regain some iron ions, these methods might shed some light on how the human body regains iron ions from lactoferrin. To date, this remains elusive, and it is unclear exactly how the body removes ferric iron from lactoferrin. Unlike transferrin, which has a role that involves sequestering iron ions as well as transporting them for further use,10 lactoferrin exists almost exclusively in secreted fluids, and is not known to be used for any other reasons than lowering iron concentration. Since transferrin needs to be able to both obtain and release iron fairly easily, it makes sense that iron ions are released around a pH of 5.5, and even a slight dip in pH has been known to trigger the release mechanism.6,3, 10 However, since lactoferrin is used far more efficiently as an antibacterial agent, then its ability to remain bound to iron in a much lower pH is better at preventing bacteria from obtaining the iron, but makes it a poor source of iron for the body. It is known that lactoferrin can be a source of iron for infants under six months,11 and it is speculated that lactoferrin must undergo a conformational change to release bound ions. However, further research is needed to understand what this change entails.6 Since lactoferrin is largely found in secreted fluids, it could be a possibility that it is not intended to release iron ions, since the body is disposing of the protein.
Sources
1. Rosa, L.; Cutone, A.; Lepanto, M.; Paesano, R.; Valenti, P. Lactoferrin: A Natural Glycoprotein Involved In Iron And Inflammatory Homeostasis. International Journal of Molecular Sciences 2017, 18, 1985.
2. Vogel, H. Lactoferrin, A Bird’S Eye View. Biochemistry and Cell Biology 2012, 90, 233-244.
3.Carmona, F.; González, A.; Sánchez, M.; Gálvez, N.; Cuesta, R.; Capdevila, M.; Dominguez-Vera, J. Varying Iron Release From Transferrin And Lactoferrin Proteins. A Laboratory Experiment. Biochemistry and Molecular Biology Education 2017, 45, 521-527.
4. Sill, C.; Biehl, R.; Hoffmann, B.; Radulescu, A.; Appavou, M.; Farago, B.; Merkel, R.; Richter, D. Structure And Domain Dynamics Of Human Lactoferrin In Solution And The Influence Of Fe(III)-Ion Ligand Binding. BMC Biophysics 2016, 9.
5. Gibbons, J. Lactoferrin And Cancer In Different Cancer Models. Frontiers in Bioscience 2011, S3, 1080.
6. Baker, H.; Baker, E. Lactoferrin And Iron: Structural And Dynamic Aspects Of Binding And Release. BioMetals 2004, 17, 209-216.
7. Ainscough, E.; Brodie, A.; Plowman, J. The Chromium, Manganese, Cobalt, And Copper Complexes Of Human Lactoferrin. Inorganica Chimica Acta 1979, 37, 282.
8. Carmona, F.; Muñoz-Robles, V.; Cuesta, R.; Gálvez, N.; Capdevila, M.; Maréchal, J.; Dominguez-Vera, J. Monitoring Lactoferrin Iron Levels By Fluorescence Resonance Energy Transfer: A Combined Chemical And Computational Study. JBIC Journal of Biological Inorganic Chemistry 2014, 19, 439-447.
9. Lisiecki, P. Transferrin And Lactoferrin – Human Iron Sources For Enterococci. Polish Journal of Microbiology 2017, 66, 419-425.
10. Dautry-Varsat, A.; Ciechanover, A.; Lodish, H. Ph And The Recycling Of Transferrin During Receptor-Mediated Endocytosis. Proceedings of the National Academy of Sciences1983, 80, 2258-2262.
11. Lönnerdal, B. Nutritional Roles Of Lactoferrin. Current Opinion in Clinical Nutrition and Metabolic Care 2009, 12, 293-297.
Contributed by
This work was originally written by Chloe Griggs, Spring 2018: Chloe is currently (as of 2018) a sophomore chemistry major at Saint Mary's College in Notre Dame, IN.
This work was originally edited by Dr. Kathryn Haas (Assistant Professor), Madison Sendzik (Teaching and Research Assistant), and Dr. Dorothy Feigl (Professor) at Saint Mary's College.