3.6: Osmosis
- Page ID
- 284478
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Outcomes
- Define osmosis and diffusion.
- Distinguish among hypotonic, hypertonic, and isotonic solutions.
- Describe a semipermeable membrane.
- Predict behavior of blood cells in different solution types.
- Describe flow of solvent molecules across a membrane.
- Identify the polar and nonpolar regions of a cell membrane.
- Explain the components present in a phospholipid.
Fish cells, like all cells, have semipermeable membranes. Eventually, the concentration of "stuff" on either side of them will even out. A fish that lives in salt water will have somewhat salty water inside itself. Put it in freshwater, and the freshwater will, through osmosis, enter the fish, causing its cells to swell, and the fish will die. What will happen to a freshwater fish in the ocean?
Osmosis
Imagine you have a cup that has \(100 \: \text{mL}\) water, and you add \(15 \: \text{g}\) of table sugar to the water. The sugar dissolves and the mixture that is now in the cup is made up of a solute (the sugar) that is dissolved in the solvent (the water). The mixture of a solute in a solvent is called a solution.
Imagine now that you have a second cup with \(100 \: \text{mL}\) of water, and you add \(45 \: \text{g}\) of table sugar to the water. Just like the first cup, the sugar is the solute, and the water is the solvent. But now you have two mixtures of different solute concentrations. In comparing two solutions of unequal solute concentration, the solution with the higher solute concentration is hypertonic, and the solution with the lower solute concentration is hypotonic. Solutions of equal solute concentration are isotonic. The first sugar solution is hypotonic to the second solution. The second sugar solution is hypertonic to the first.
You now add the two solutions to a beaker that has been divided by a semipermeable membrane, with pores that are too small for the sugar molecules to pass through, but are big enough for the water molecules to pass through. The hypertonic solution is one side of the membrane and the hypotonic solution on the other. The hypertonic solution has a lower water concentration than the hypotonic solution, so a concentration gradient of water now exists across the membrane. Water molecules will move from the side of higher water concentration to the side of lower concentration until both solutions are isotonic. At this point, equilibrium is reached.
Red blood cells behave the same way (see figure below). When red blood cells are in a hypertonic (higher concentration) solution, water flows out of the cell faster than it comes in. This results in crenation (shriveling) of the blood cell. On the other extreme, a red blood cell that is hypotonic (lower concentration outside the cell) will result in more water flowing into the cell than out. This results in swelling of the cell and potential hemolysis (bursting) of the cell. In an isotonic solution, the flow of water in and out of the cell is happening at the same rate.
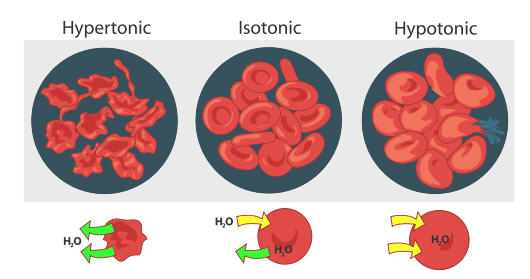
Osmosis is the diffusion of water molecules across a semipermeable membrane from an area of lower concentration solution (i.e., higher concentration of water) to an area of higher concentration solution (i.e., lower concentration of water). Water moves into and out of cells by osmosis.
- If a cell is in a hypertonic solution, the solution has a lower water concentration than the cell cytosol, and water moves out of the cell until both solutions are isotonic.
- Cells placed in a hypotonic solution will take in water across their membranes until both the external solution and the cytosol are isotonic.
A red blood cell will swell and undergo hemolysis (burst) when placed in a hypotonic solution. When placed in a hypertonic solution, a red blood cell will lose water and undergo crenation (shrivel). Animal cells tend to do best in an isotonic environment, where the flow of water in and out of the cell is occurring at equal rates.
Diffusion
Passive transport is a way that small molecules or ions move across the cell membrane without input of energy by the cell. The three main kinds of passive transport are diffusion (or simple diffusion), osmosis, and facilitated diffusion. Simple diffusion and osmosis do not involve transport proteins. Facilitated diffusion requires the assistance of proteins.
Diffusion is the movement of molecules from an area of high concentration of the molecules to an area with a lower concentration. For cell transport, diffusion is the movement of small molecules across the cell membrane. The difference in the concentrations of the molecules in the two areas is called the concentration gradient. The kinetic energy of the molecules results in random motion, causing diffusion. In simple diffusion, this process proceeds without the aid of a transport protein. It is the random motion of the molecules that causes them to move from an area of high concentration to an area with a lower concentration.
Diffusion will continue until the concentration gradient has been eliminated. Since diffusion moves materials from an area of higher concentration to the lower, it is described as moving solutes "down the concentration gradient". The end result is an equal concentration, or equilibrium, of molecules on both sides of the membrane. At equilibrium, movement of molecules does not stop. At equilibrium, there is equal movement of materials in both directions.
Not everything can make it into your cells. Your cells have a plasma membrane that helps to guard your cells from unwanted intruders.
The Plasma Membrane and Cytosol
If the outside environment of a cell is water-based, and the inside of the cell is also mostly water, something has to make sure the cell stays intact in this environment. What would happen if a cell dissolved in water, like sugar does? Obviously, the cell could not survive in such an environment. So something must protect the cell and allow it to survive in its water-based environment. All cells have a barrier around them that separates them from the environment and from other cells. This barrier is called the plasma membrane, or cell membrane.
The Plasma Membrane
The plasma membrane (see figure below) is made of a double layer of special lipids, known as phospholipids. The phospholipid is a lipid molecule with a hydrophilic ("water-loving") head and two hydrophobic ("water-hating") tails. Because of the hydrophilic and hydrophobic nature of the phospholipid, the molecule must be arranged in a specific pattern as only certain parts of the molecule can physically be in contact with water. Remember that there is water outside the cell, and the cytoplasm inside the cell is mostly water as well. So the phospholipids are arranged in a double layer (a bilayer) to keep the cell separate from its environment. Lipids do not mix with water (recall that oil is a lipid), so the phospholipid bilayer of the cell membrane acts as a barrier, keeping water out of the cell, and keeping the cytoplasm inside the cell. The cell membrane allows the cell to stay structurally intact in its water-based environment.
The function of the plasma membrane is to control what goes in and out of the cell. Some molecules can go through the cell membrane to enter and leave the cell, but some cannot. The cell is therefore not completely permeable. "Permeable" means that anything can cross a barrier. An open door is completely permeable to anything that wants to enter or exit through the door. The plasma membrane is semipermeable, meaning that some things can enter the cell, and some things cannot.
Molecules that cannot easily pass through the bilayer include ions and small hydrophilic molecules, such as glucose, and macromolecules, including proteins and RNA. Examples of molecules that can easily diffuse across the plasma membrane include carbon dioxide and oxygen gas. These molecules diffuse freely in and out of the cell, along their concentration gradient. Though water is a polar molecule, it can also diffuse through the plasma membrane.

Cytosol
The inside of all cells also contain a jelly-like substance called cytosol. Cytosol is composed of water and other molecules, including enzymes, which are proteins that speed up the cell's chemical reactions. Everything in the cell sits in the cytosol, like fruit in a Jell-o mold. The term cytoplasm refers to the cytosol and all of the organelles, the specialized compartments of the cell. The cytoplasm does not include the nucleus. As a prokaryotic cell does not have a nucleus, the DNA is in the cytoplasm.
Supplemental Resources
- The Plasma Membrane: www.youtube.com/watch?v=moPJkCbKjBs
Concept Review Exercises
1. What are some of the features of a semipermeable membrane?
2. What do the prefixes hyper, hypo, and iso mean?
Answers
1. A semipermeable membrane allows some substances to pass through but not others.
2. hyper – higher; hypo – lower; iso - same
Key Takeaways
- Water moves into and out of cells by osmosis.
- Water (solvent) moves from an area of lower concentration solution (i.e., higher concentration of water) to an area of higher concentration solution (i.e., lower concentration of water).
EXERCISES
1. Two solutions are separated by a semipermeable membrane. Solution A contains 25.0 g of NaCl in 100.0 mL of water and solution B contains 35.0 g of NaCl in 100.0 mL of water.
- Which one has a higher concentration?
- Which way will water molecules flow?
- Which volume will increase?
- Which volume will decrease?
- What will happen to the concentration of solution A?
- What will happen to the concentration of solution B?
2. Two solutions with different concentrations and compositions are separated by a semipermeable membrane. The left-hand solution is a .50 M solution of MgSO4, while the right-hand solution contains CaCl2 at a concentration of .40 M. Determine the direction of the flow of solvent, left or right.
3. Given the following situations, wherein two tanks of different solutions are separated by a semipermeable membrane, determine the direction of the flow of solvent (water).
a. Solution A contains a 0.40 M concentration of CaCl2, while Solution B contains a 0.45 M concentration of KI
b. Solution A contains a 1.00 M concentration of NH4Cl, while Solution B contains a 1.00 M concentration of CH2O
4. Cells are placed in a solution and the cells then undergo hemolysis. What can be said about the relative concentrations of solute in the cell and the solution?
5. Describe the relative concentrations inside and outside a red blood cell when crenation occurs.
6. A saltwater fish is placed in a freshwater tank. What will happen to the fish? Describe the flow of water molecules to explain the outcome.
7. What makes up the "head" region of a phospholipid? Is it hydrophobic or hydrophilic?
8. What makes up the "tail" region of a phospholipid? Is it hydrophobic or hydrophilic?
Answers
1. Two solutions are separated by a semipermeable membrane. Solution A contains 25.0 g of NaCl in 100.0 mL of water and solution B contains 35.0 g of NaCl in 100.0 mL of water.
- Solution B
- A →→ B
- B
- A
- increase
- decrease
2. Water (solvent) flows from left to right.
3.
a. Water flows from Solution B to Solution A.
b. Water flows from Solution B to Solution A.
4. Cells contain fluid with higher concentration than solution outside the cell.
5. Cells contain fluid with a lower concentration than the solution outside the cell.
6. Water molecules will flow from the tank water into the fish because the fish has a higher concentration of salt. If the fish absorbs too much water, it will die.
7. The "head" region is a phosphate group and it is hydrophilic.
8. The "tail" is a hydrocarbon tail and it is hydrophobic.
Contributors
Allison Soult, Ph.D. (Department of Chemistry, University of Kentucky)

