12.5: Transitions between Phases
- Page ID
- 476614
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Identify the names of the phase changes between solid, liquid, and gas phases.
We have introduced the different physical states of matter and mentioned that transitions between these physical states are a type of physical change. We will explore these changes in more detail in this section.
Evaporation
A puddle of water left undisturbed eventually disappears. The liquid molecules escape into the gas phase, becoming water vapor. Vaporization is the process in which a liquid is converted to a gas. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. If the water is instead kept in a closed container, the water vapor molecules do not have a chance to escape into the surroundings, and so the water level does not change. As some water molecules become vapor, an equal number of water vapor molecules condense back into the liquid state. Condensation is the change of state from a gas to a liquid.
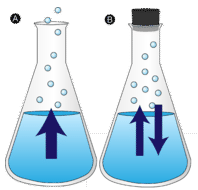
In some parts of the world, people are able to cool off using a device known as a "swamp cooler". This piece of equipment traces its origin back to the ancient Egyptians, who hung wet blankets across the doors of their homes. As the warm air passed through the blankets, water would evaporate and cool the air. The royalty went one step further, and had servants fan wet cloths over jugs of water to get more evaporation and cooling. The origin of the term "swamp cooler" is not known; they certainly do not work in a swamp. The best conditions for cooling include a high temperature (over \(80^\text{o} \text{F}\)) and a low humidity (preferably less than \(30\%\)). These coolers work well in desert areas, but do not provide any cooling in the humid areas of the country.
Boiling Point
As a liquid is heated, the average kinetic energy of its particles increases. The rate of evaporation increases as more and more molecules are able to escape the liquid's surface into the vapor phase. Eventually a point is reached when the molecules all throughout the liquid have enough kinetic energy to vaporize. At this point, the liquid begins to boil. The boiling point is the temperature at which the vapor pressure of a liquid is equal to the external pressure. Figure \(\PageIndex{2}\) illustrates the boiling of liquid.
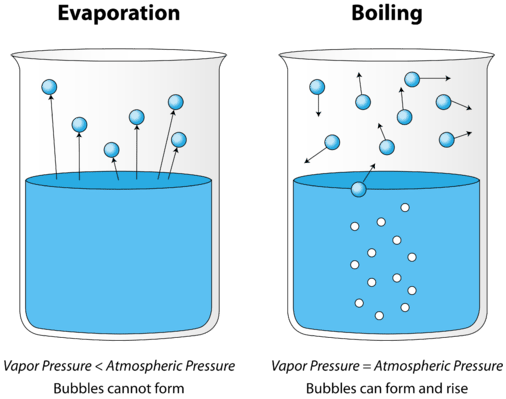
In the picture on the left, the liquid is below its boiling point, yet some of the liquid evaporates. On the right, the temperature has been increased until bubbles begin to form in the body of the liquid. When the vapor pressure inside the bubbles is equal to the external atmospheric pressure, the bubbles rise to the surface of the liquid and burst. The temperature at which this process occurs is the boiling point of the liquid.
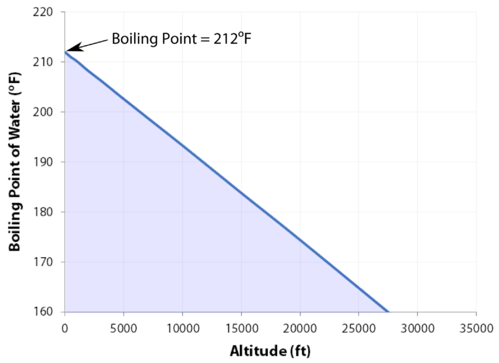
The normal boiling point is the temperature at which the vapor pressure of the liquid is equal to standard pressure. Because atmospheric pressure can change based on location, the boiling point of a liquid changes with the external pressure. The normal boiling point is a constant because it is defined relative to the standard atmospheric pressure of \(760 \: \text{mm} \: \ce{Hg}\) (or \(1 \: \text{atm}\) or \(101.3 \: \text{kPa}\)).
Melting Point
Solids are similar to liquids in that both are condensed states, with particles that are far closer together than those of a gas. However, while liquids are fluid, solids are not. The particles of most solids are packed tightly together in an orderly arrangement. The motion of individual atoms, ions, or molecules in a solid is restricted to vibrational motion about a fixed point. Solids are almost completely incompressible and are the densest of the three states of matter.
As a solid is heated, its particles vibrate more rapidly as it absorbs kinetic energy. Eventually, the organization of the particles within the solid structure begins to break down, and the solid starts to melt. The melting point is the temperature at which a solid changes into a liquid. At its melting point, the disruptive vibrations of the particles of the solid overcome the attractive forces operating within the solid. As with boiling points, the melting point of a solid is dependent on the strength of those attractive forces. Sodium chloride \(\left( \ce{NaCl} \right)\) is an ionic compound that consists of a multitude of strong ionic bonds. Sodium chloride melts at \(801^\text{o} \text{C}\). Ice (solid \(\ce{H_2O}\)) is a molecular compound whose molecules are held together by hydrogen bonds. Though hydrogen bonds are the strongest of the intermolecular forces, the strength of hydrogen bonds is much less than that of ionic bonds. The melting point of ice is 0 °C.
The melting point of a solid is the same as the freezing point of the liquid. At that temperature, the solid and liquid states of the substance are in equilibrium. For water, this equilibrium occurs at \(0^\text{o} \text{C}\).
\[\ce{H_2O} \left( s \right) \rightleftharpoons \ce{H_2O} \left( l \right)\nonumber \]
We tend to think of solids as those materials that are solid at room temperature. However, all materials have melting points of some sort. Gases become solids at extremely low temperatures, and liquids will also become solid if the temperature is low enough.
The table below gives the melting points of some common materials.
| Materials | Melting Point (°C) |
|---|---|
| Hydrogen | -259 |
| Oxygen | -219 |
| Diethyl ether | -116 |
| Ethanol | -114 |
| Water | 0 |
| Pure silver | 961 |
| Pure gold | 1063 |
| Iron | 1538 |
Sublimation
Solids also have a vapor pressure, though it is generally much less than that of a liquid. A snow bank will gradually disappear even if the temperature stays below \(0^\text{o} \text{C}\). The snow does not melt, but instead passes directly from the solid state to the vapor state. Sublimation is the change of state from a solid to a gas, without passing through the liquid state.
"Dry ice", or solid carbon dioxide, is a substance that sublimes at atmospheric pressures. Dry ice is very cold \(\left( -78^\text{o} \text{C} \right)\) and so is used as a coolant for goods, such as ice cream, that must remain frozen during shipment. Because the dry ice sublimes rather than melts, there is no liquid mess associated with its change of state as it warms. As you may have seen in demonstrations, dry ice merely transforms from a solid to a cold, steamy looking gas. (The steam you see is actual water that has condensed around the very cold carbon dioxide gas that is forming as the dry ice sublimes.)
Summary of State Changes
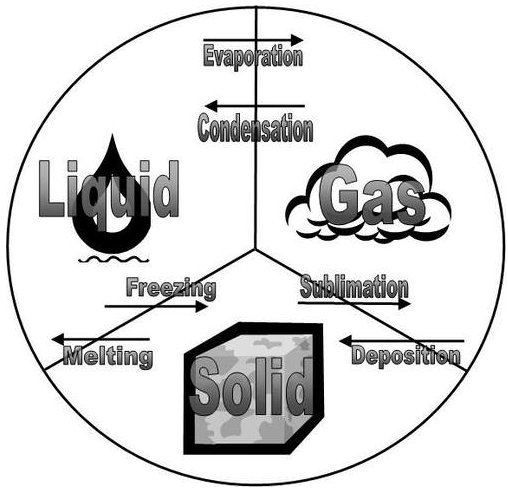
All of the changes of state that occur between solid, liquid, and gas are summarized in the diagram in the figure below. Freezing is the opposite of melting, and both represent the equilibrium between the solid and liquid states. Evaporation occurs when a liquid turns to a gas. Condensation is the opposite of vaporization, and both represent the equilibrium between the liquid and gas states. Deposition is the opposite of sublimation, and both represent the equilibrium between the solid and gas states.
Section Summary
- The boiling point is the temperature at which the vapor pressure of a liquid is equal to the external pressure.
- As the altitude increases, the boiling point decreases.
- Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid.
- Condensation is the change of state from a gas to a liquid.
- As the temperature increases, the rate of evaporation increases.
- The melting point is the temperature at which a solid changes into a liquid.
- Intermolecular forces have a strong influence on melting point.
- Sublimation is the change of state from a solid to a gas, without passing through the liquid state.
- Freezing is the opposite of melting.
- Deposition is the opposite of sublimation.
Glossary
- vaporization
- The process in which a liquid is converted to a gas.
- evaporation
- The conversion of a liquid to its vapor below the boiling temperature of the liquid.
- condensation
- The change of state from a gas to a liquid.
- boiling point
- The temperature at which the vapor pressure of a liquid is equal to the external pressure.
- melting point
- The temperature at which a solid changes into a liquid.
- sublimation
- The change of state from a solid to a gas, without passing through the liquid state.
- freezing
- the change of state from a liquid to a solid.
- deposition
- the change of state from a gas to a solid, without passing through the liquid state.


