Experiment_729_Qualitative Testing of Amino Acids and Proteins 1_2
- Page ID
- 305612
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)|
Student Name |
|
Laboratory Date: Date Report Submitted: |
___________________________
|
|
Student ID |
|
Experiment Number and Title |
Experiment 729: Qualitative Analysis of Amino Acids and Proteins |
Experiment 729: Qualitative Analysis of Amino Acids and Proteins
Section 1: Purpose and Summary:
-
Develop an understanding of the structure and bonding in amino acids and proteins.
-
Identify different amino acids and an unknown by paper chromatography.
-
Observe how different amino acids and proteins react in different chemical tests.
What are proteins?
Proteins are polymers of amino acids linked together by peptide bonds. Proteins are important biological molecules with many functions. There are transport proteins (hemoglobin is an example), storage proteins, structural proteins, proteins for muscular contraction, and others. Proteins that catalyze biological reactions are called enzymes. All proteins are very large molecules with high molecular weights that range from 6500 to 205,000 or more grams per mole.
An amino acid is a molecule that contains an amino group and a carboxyl group in the same molecule. Amino acids found in proteins are α-amino acids. This means the amino group (NH2- or NH3+-) is attached to the alpha carbon--this is the carbon next to the carboxyl group. There are 20 amino acids that differ from each other only in the identity of the side chain attached to the alpha-carbon. The amino acid side chains can be classified based on whether they are nonpolar, polar, acidic, or basic. The general structure of an amino acid is shown below. In the following structure, R represents the side chain.
Since proteins are biological molecules, they are usually found in a neutral solution that is buffered between pH 7.0 and 7.4. In this situation, the acidic and basic groups on the amino acids will be ionized. Most amino acids exist as “zwitterions” (dipolar ions) at pH 7. The structure of a zwitterion is shown below. Because of the charges, amino acids are water-soluble.
Amino acids can be linked together – the amino group on one amino acid can react with the carboxyl group on another amino acid, forming an amide. Therefore, the bonds linking together the amino acid residues are amide bonds, but in proteins they are called peptide bonds. Shown on the next page is the structure of a dipeptide (the prefix "di" means two--there are two amino acids in a dipeptide). A typical protein molecule contains around 100 amino acids joined by peptide bonds.
alanylglycine, a dipeptide
There are several aspects to describe structure of a protein. The primary structure of a protein is the sequence of amino acids. This sequence determines the overall shape of the protein. The secondary structure consists of regular and repeating structures held together by hydrogen bonds between the C=O and the N-H groups along the backbone of the molecule. The alpha helix and beta sheet are examples of secondary structure. The tertiary structure refers to the overall folding of the entire polypeptide chain and is determined by interactions between the side chains off the amino acids. These interactions include hydrogen bonding, hydrophobic interactions, salt bridges, and disulfide bridges. Quaternary structure involves the same types of interactions as tertiary structure, but the interactions occur between the side chains on different polypeptide chains. These interactions hold together different subunits of a protein.
If the overall folding of a protein is changed, the protein is called denatured. A denatured protein is no longer biological active. Some common denaturing agents include heat, organic solvents, acids, bases, agitation, detergents, and heavy metal ions. It is important to note that denaturation affects the secondary, tertiary, and quaternary structure of a protein, but does not affect the primary structure. Sometimes the denaturation is reversible, and the protein can be renatured (which reestablishes its biological activity).
Paper Chromatography of Amino Acids
Chromatography is a technique of separation and identification. There are many types of chromatography, including but not limited to paper chromatography, thin layer chromatography, gas chromatography, liquid chromatography, and ion-exchange chromatography. In this experiment, paper chromatography will be used to identify amino acids.
For paper chromatography, the samples to be tested are spotted on one edge of a rectangular piece of filter paper. The paper is placed in a container that has a solvent that wets the bottom of the paper. The paper acts as a wick for the solvent. The solvent passed through the samples as it wicks up the paper, carrying the samples. The samples move at different rates: those that are more attracted to the solvent will travel faster, and those that are more attracted to the paper will move slower. After the solvent has moved about 3/4 of the way up the paper, the chromatography is complete. The paper is removed from the container and dried. The amino acid solutions are colorless, so a developer needs to be used to make the amino acids visible. For amino acids, it is sprayed with a developer called ninhydrin. This will react with amino acids and produce a purple color. Ninhydrin will also react with proteins.
A photo of the chromatography setup is below:
<< PHOTO WILL BE IN NEXT UPDATE >>
After the paper is developed, the distance that the solvent traveled and the distance that each amino acid has traveled are measured. This is called the retention factor, Rf, and has a different value for each amino acid is then calculated. By comparing Rf values of unknown amino acids to those of known values, the unknowns can be identified.
A sample calculation of Rf values is shown below.
\(R_{f}=\frac{\text { distance traveled by amino acid }}{\text { distance traveled by solvent }}\)
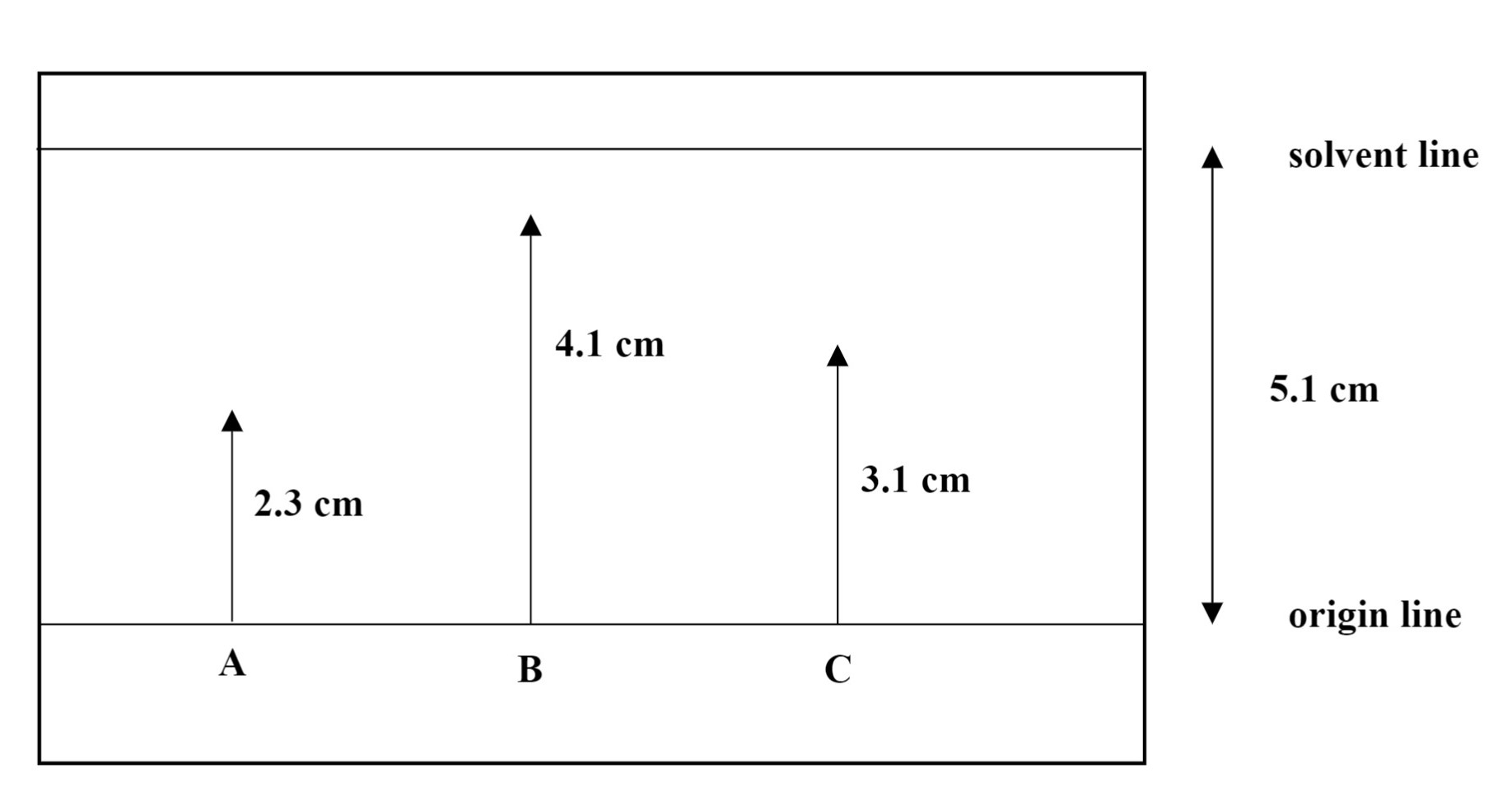
Spot A: \(R_{f}=\frac{2.3 \mathrm{~cm}}{5.1 \mathrm{~cm}}=0.45\) Spot B: \(R_{f}=\frac{4.1 \mathrm{~cm}}{5.1 \mathrm{~cm}}=0.80\)
Spot C: \(R_{f}=\frac{3.1 \mathrm{~cm}}{5.1 \mathrm{~cm}}=0.61\)
In this experiment, students will test several known amino acid solutions and one unknown. The results for the unknown should match one of the known amino acids.
Reactions of Amino Acids and Proteins
Hydrolysis
Proteins can be hydrolyzed either partially or completely in the presence of acids, bases, or digestive enzymes. When a protein is hydrolyzed, some or all of the peptide bonds are broken. The products obtained depend on how long the hydrolysis is allowed to take place. The products of partial hydrolysis are peptides and the products of complete hydrolysis are amino acids.
Biuret Test
Compounds that contain two or more peptide bonds will react with Cu2+ in a basic solution to form a violet-pink complex. The original Cu2+ solution is blue, so if the solution remains blue, the compound being tested could be an amino acid or a dipeptide or neither.
Xanthoproteic Test
The aromatic rings on tyrosine and tryptophan react with nitric acid. In this reaction, the aromatic rings become nitrated. When nitric acid is added to a sample and the mixture is heated, a yellow solution will result if the sample contains tyrosine or tryptophan. When this yellow solution is treated with a strong base (such as NaOH), it turns orange. Since most proteins contain one or both amino acids, most proteins will show a positive reaction in this test.
Ninhydrin Test
Free amino groups will react with the ninhydrin reagent to yield a purple solution. Almost all amino acids contain a free amino group (except proline and hydroxyproline). Some proteins also give a positive test with ninhydrin.
Sulfur Test
Sulfur-containing amino acids include cysteine and methionine. If a sample that contains one or both of these amino acids is acidified, the gas H2S is produced, which smells like rotten eggs. When a piece of moistened lead (II) acetate paper is held over the solution as the H2S is being produced, the H2S reacts with the lead ion, forming a black or gray coating of PbS (lead (II) sulfide). Appearance of this black color is taken as a positive test for a sulfur-containing amino acid.
Denaturation
There are many ways to denature a protein. Recall that denaturation refers to a disruption of the secondary, tertiary, and quaternary structures – denaturation destroys the normal folding of the protein, making it inactive. When a protein is denatured, it often coagulates, forming a visible solid. Some denaturing agents include heat, organic solvents, agitation, acid or base, and heavy metal ions.
Section 2: Safety Precautions and Waste Disposal
Safety Precautions:
Wear your safety goggles.
Nitric acid is very corrosive – avoid skin contact with it. If skin contact occurs, flood the area with plenty of running water for 10 minutes.
Heavy metal ions such as Pb2+ are poisonous. Avoid skin contact with solutions containing these ions and with the lead acetate paper. Wash your hands thoroughly before leaving the lab.
Ninhydrin spray causes stains.
Waste Disposal:
At the end of the experiment, all liquid wastes go into the inorganic waste container. Solid wastes go into the solid waste container.
Section 3: Procedure
Part I. Paper Chromatography
|
|
|
|
|
Identification code for the unknown amino acid:
Unknown # __________________ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amino Acid |
Distance traveled by amino acid (cm) |
Distance traveled by solvent (cm) |
Rf value |
|
Phenylalanine |
|
|
|
|
Alanine |
|
|
|
|
Glutamic acid |
|
|
|
|
Serine |
|
|
|
|
Lysine |
|
|
|
|
Aspartic acid |
|
|
|
|
Unknown amino acid |
|
|
|
Identification of Unknown
|
Unknown number |
Identity of Unknown |
Reasoning |
|
|
|
|
Part 2. Chemical Tests for Amino Acids
|
Note: Be sure to shake the protein solutions before obtaining your sample because some of the proteins tend to settle out.
Note: Several tests require a hot water bath. Prepare the water bath early so it is available when you need it.
Obtain solutions of glutamic acid, lysine, and serine. One by one, stir the amino acid sample with a glass stirring rod and then touch it to a piece of pH paper (not litmus paper). Note and record the pH (by comparing the color of the spot to the pH scale on the box of pH paper - be sure to look at both sides of the box). Make sure to rinse and wipe off the stirring rod between solutions so that you don’t accidentally contaminate them. |
Observations:
|
||||||||
|
2. Biuret Test In this part, you will test solutions of egg albumin, gelatin, casein, glycine, and proline. Put 2 mL of each solution to be tested in its own test tube. Add 3 mL of 10% NaOH to each tube and mix well. (Remember, if you are using a stirring rod, rinse it and wipe it off between solutions so that you don’t contaminate the solutions.) Add 2 drops of 2% copper sulfate (CuSO4) solution (which is blue) to each tube and mix well. Record your observations on the data table below. |
|
||||||||
|
3. Xanthoproteic Test You will test solutions of egg albumin and tyrosine. Put about 1 mL of each solution to be tested in its own test tube. In the hood, carefully add 5 drops of concentrated nitric acid (HNO3). Caution: nitric acid must be kept under the hood at all times. Avoid all skin contact with nitric acid. Mix each test tube carefully and place the tubes in a boiling water bath for two minutes. Observe and record the color of the solutions. Remove them from the water bath and let them cool. When they are cool, add 10% NaOH to each tube dropwise until the solutions are basic. (You can do this by adding one drop of NaOH, mixing well with a stirring rod, and touching the stirring rod to a piece of red litmus paper. If the litmus paper does not turn blue, add another drop of NaOH to the solution, mix with the stirring rod, and again touch it to a piece of red litmus paper. Repeat this sequence until the litmus paper turns blue – then your solution is basic. You can use the same piece of litmus paper, if you touch the stirring rod to a different dry section each time.) Observe and record any color changes. |
|
||||||||
|
4. Ninhydrin Test You will test solutions of glycine, proline, and egg albumin. Put 2 mL of each solution to be tested in separate test tubes. Add 1 mL of 1 % ninhydrin solution to each tube. Heat the tubes for several minutes in a boiling water bath. Record your observations. |
|
||||||||
|
5. Sulfur Test Test solid egg albumin and solid cysteine. Weigh out about 0.2 g of each of these solids and place the samples in separate test tubes. Add 10 mL of 3 M NaOH to each tube and place the tubes in a boiling water bath for 15 minutes. If the solutions start to foam, remove them from the water bath briefly. After 15 minutes of heating, remove the tubes from the water bath and let them cool to room temperature. Add about 10 mL of 3 M HCl to each tube to acidify the contents. Moisten two pieces of lead acetate paper with deionized water and place a piece over the top of each tube. Put the tubes in the boiling water bath again. Do not let the lead acetate paper fall into the solutions. Record your observations. Wash your hands after handling the lead acetate paper, as lead compounds are toxic. |
|
||||||||
|
6. Denaturation Tests a. Put 3 mL of egg albumin solution in a test tube and place the test tube in a boiling water bath. Observe the appearance of the solution before and after heating.
b. Put 3 mL of egg albumin solution in a test tube and add 7 mL of 95 % ethanol. Mix well and record your observations.
c. Put 3 mL of egg albumin solution in a test tube and add 5-8 drops of FeCl3 solution. Mix well and record your observations.
d. Put 3 mL of egg albumin solution in a test tube and add 5-8 drops of 0.2 M ZnCl2 solution. Mix well and record your observations. Put the waste in the inorganic waste container in the hood. |
|
Observations
Biuret Test
|
Substance |
Observations |
Is this a positive or a negative reaction? |
|
Egg albumin |
|
|
|
Gelatin |
|
|
|
Casein |
|
|
|
Glycine |
|
|
|
Proline |
|
|
Xanthoproteic Test
|
Substance |
Initial Color |
Color after acid is added |
Color after base is added |
|
Egg albumin |
|
|
|
|
Tyrosine |
|
|
|
Ninhydrin Test
|
Substance |
Observations |
|
Glycine |
|
|
Proline |
|
|
Egg albumin |
|
Sulfur Test
|
Substance |
Observations |
Conclusion |
|
Solid egg albumin |
|
|
|
Solid cysteine |
|
|
Denaturation Tests
|
Substance |
Initial appearance |
Appearance after denaturation |
|
Egg albumin + heat |
|
|
|
Egg albumin + 95% ethanol |
|
|
|
Egg albumin + FeCl3 |
|
|
|
Egg albumin + Hg2Cl2 |
|
|
Post Lab Questions:
1. According to the results of the paper chromatography, which amino acids were most attracted to the solvent? Which amino acids were most attracted to the paper? Explain.
2. According to your answer to question #1, which do you think is more polar, the chromatography solvent or the paper? Explain.
3. If you had completely hydrolyzed the egg albumin before doing the Biuret test on that sample, what results would you expect for the Biuret test? Explain why. Include what color you would expect to see, whether it is a positive or negative test, and what that means.
4. Suggest a reason why alcohol or other disinfectants are often applied to a person’s skin before an injection is given (based on something you learned in this lab).
5. Draw the structure of phenylalanine in its regular form and in its zwitterion form.
6. What substances react in the Biuret test?
7. What substances react in the Xanthoproteic test?
8. Suggest a reason why milk is used as an antidote for lead poisoning. Hint: milk contains a lot of protein.
9. Predict the results of each of the following reactions. Include the observed colors and whether it corresponds to a positive or a negative test.
-
The Biuret test on proline
-
The Biuret test on egg albumin
-
The ninhydrin test on lysine
-
The xanthoproteic test on egg albumin
-
The sulfur test on cysteine